B cell precursor acute lymphoblastic leukaemia (B ALL)
Active Ingredient: Obecabtagene autoleucel
Indication for Obecabtagene autoleucel
Obecabtagene autoleucel is indicated for the treatment of adult patients 26 years of age and above with relapsed or refractory (r/r) B cell precursor acute lymphoblastic leukaemia (B ALL).
For this indication, competent medicine agencies globally authorize below treatments:
410 × 10⁶ CD19 CAR-positive viable T cells administered as 2 split doses on Days 1 and 10 (± 2 days)
For:
Dosage regimens
Regimen A, in case that there is high tumour mutational burden
Intravenous, 10,000,000 cells obecabtagene autoleucel, one dose, over the duration of 9 days. Afterwards, intravenous, 100,000,000 cells obecabtagene autoleucel, one dose, over the duration of 2 days. Afterwards, intravenous, 300,000,000 cells obecabtagene autoleucel, one dose.
Regimen B, in case that there is low tumor mutational burden
Intravenous, 100,000,000 cells obecabtagene autoleucel, one dose, over the duration of 9 days. Afterwards, intravenous, 10,000,000 cells obecabtagene autoleucel, one dose, over the duration of 2 days. Afterwards, intravenous, 300,000,000 cells obecabtagene autoleucel, one dose.
Detailed description
The target dose is 410 × 106 CD19 CAR-positive viable T cells (range: 308-513 × 106 CAR-positive viable T cells) supplied in 3 or more infusion bags.
The treatment regimen consists of a split dose to be administered on day 1 and day 10 (± 2 days). The dose regimen will be determined by the tumour burden assessed by bone marrow (BM) blast percentage from a sample obtained within 7 days prior to the start of lymphodepletion (Figure 1).
The RfIC and Dose Schedule Planner (Annex IIIA), located inside the lid of the cryoshipper, must be followed for the actual cell counts and volumes to be infused and to guide the appropriate dose regimen.
Bone marrow assessment
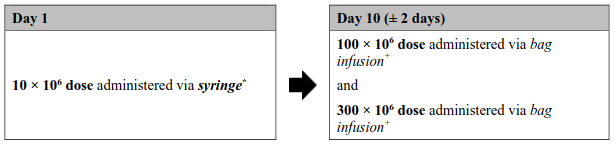
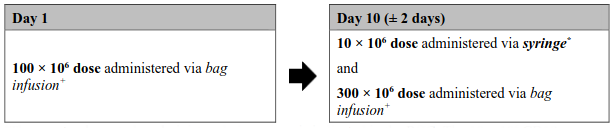
A BM assessment must be available from a biopsy and/or aspirate sample obtained within 7 days prior to the commencement of the lymphodepleting chemotherapy. The BM assessment will be used to determine the Aucatzyl dose regimen: High Tumour Burden Regimen if blast percentage is >20% or Low Tumour Burden Regimen if blast percentage is ≤20% (see Figure 1).
If BM assessment results are inconclusive, the biopsy or aspirate must be repeated (but only once). A repeat biopsy or aspirate should only be taken prior to lymphodepleting chemotherapy.
If results remain inconclusive, the High Tumour Burden Regimen should be administered (i.e., administration of the 10 × 106 dose on day 1 per Figure 1).
Figure 1. Aucatzyl tumour burden adjusted split dose regimen:
High tumour burden dose regimen
(Bone marrow blast >20% or inconclusive)
Low tumour burden dose regimen
(Bone marrow blast ≤20%)
* The exact volume to be administered via syringe is indicated in the RfIC. The 10 × 106 CD19 CAR-positive viable T cell bag configuration contains an overfill, and therefore it is important to withdraw only the volume specified.
+ The 100 × 106 and 300 × 106 doses will be suspended in one or more infusion bags with no overfill.
Bridging therapy
Bridging therapy can be considered according to the prescriber's choice prior to infusion to reduce tumour burden or stabilise the disease.
Pretreatment (lymphodepleting chemotherapy)
The lymphodepleting chemotherapy regimen must be administered before infusion of Aucatzyl: fludarabine (FLU) 30 mg/m²/day intravenously and cyclophosphamide (CY) 500 mg/m²/day intravenously on days -6 and -5, followed by fludarabine on days -4 and -3 (total dose: FLU 120 mg/m²; CY 1 000 mg/m²). For dose modifications of cyclophosphamide and fludarabine, see corresponding Summaries of Product Characteristics of cyclophosphamide and fludarabine.
Retreatment with lymphodepleting chemotherapy, in patients who could not receive the Aucatzyl dose on day 1 as planned, could be considered if there is an Aucatzyl dose delay of more than 10 days. Lymphodepleting chemotherapy should not be repeated after the first dose of Aucatzyl is administered.
Aucatzyl is infused 3 days (± 1 day) after completion of lymphodepleting chemotherapy (day 1), allowing a minimum 48-hour washout.
Treatment with Aucatzyl should be delayed in some patient groups at risk. A delay to the second split dose may be required to manage toxicities.
Premedication
To minimise the risk of an infusion reaction, it is recommended that patients be premedicated with paracetamol (1 000 mg orally) and diphenhydramine 12.5 to 25 mg intravenously or orally (or equivalent medicinal products) approximately 30 minutes prior to Aucatzyl infusion.
Prophylactic use of systemic corticosteroids is not recommended.
Reasons to delay treatment
Delay Aucatzyl treatment if there are unresolved serious adverse reactions from preceding chemotherapies, if the patient is experiencing severe intercurrent infection, or has active graft-versus-host disease. If the patient requires supplementary oxygen, Aucatzyl should only be infused, if considered appropriate, based on the treating physician's benefit/risk assessment.
Reasons to delay the second split dose
Dose delays or discontinuation of treatment may be required after the first split dose to manage adverse reactions.
Monitoring
Patients should be monitored daily for 14 days after the first infusion for signs and symptoms of potential CRS, immune effector cell-associated neurotoxicity syndrome (ICANS) and other toxicities.
Frequency of monitoring after the first 14 days should be carried out at the physician's discretion and should be continued for at least 4 weeks after.
Patients should be instructed to remain within close proximity of the qualified treatment centre (within 2 hours of travel) for at least 4 weeks following the first infusion.
Dosage considerations
Obecabtagene autoleucel is intended for autologous and intravenous use only.
Liability Disclaimer : RxReasoner has utilized reasonable care in providing content and services that are accurate, complete and up to date. However, RxReasoner does not accept any responsibility or liability about it. The content and services of RxReasoner are for informational purposes only and they are not intended to be a substitute for the knowledge, expertise, skill, and judgment of physicians, pharmacists, nurses, or other healthcare professionals involved in patient care. RxReasoner offers no medical advice. Users are responsible for the use of the provided content. A shown indication or treatment should not be construed to indicate that the medication is safe, appropriate, or effective in any given patient or under any particular circumstances. The absence of an indication or treatment should not roule out the existence of other appropriate medications. Always seek the advice of a physician or other qualified health provider with any questions you may have regarding a medical condition or medicament. RxReasoner is not liable for any damages allegedly sustained arising out of the use of its content and services.