Acute lymphoblastic leukaemia (ALL)
Active Ingredient: Mercaptopurine
Indication for Mercaptopurine
Mercaptopurine is indicated for the treatment of acute lymphoblastic leukaemia (ALL) in adults, adolescents and children.
For this indication, competent medicine agencies globally authorize below treatments:
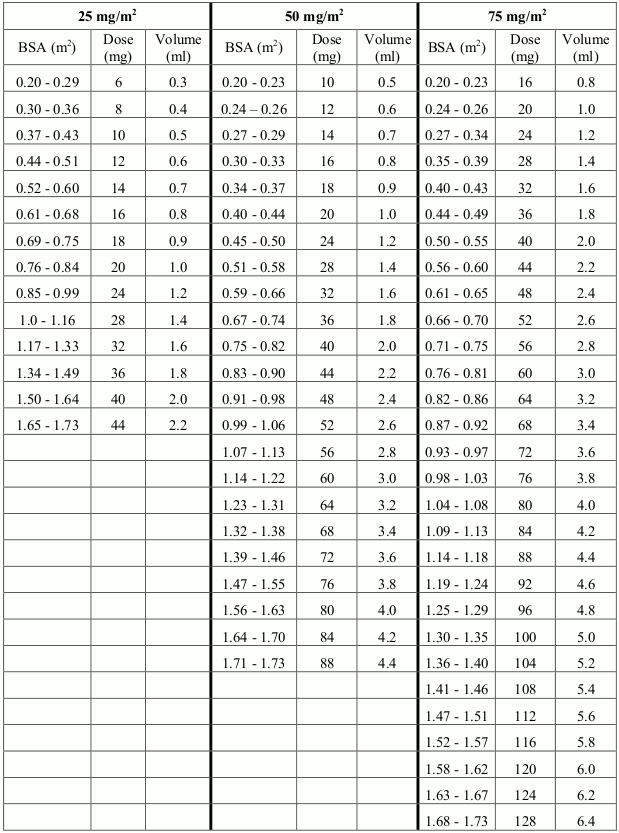
25-75 mg/m² once a day
Route of admnistration
Oral
Defined daily dose
25 - 75 mg per m² of body surface area (BSA)
Dosage regimen
From 25 To 75 mg per m² of body surface area (BSA) once every day
Detailed description
The dose is governed by cautiously monitored haematotoxicity and the dose should be carefully adjusted to suit the individual patient in accordance with the employed treatment protocol. Depending on phase of treatment, starting or target doses generally vary between 25-75 mg/m² body surface area (BSA) per day, but should be lower in patients with reduced or absent Thiopurine Methyl Transferase (TPMT) enzyme activity.
6-mercaptopurine is metabolised by the polymorphic TPMT enzyme. Patients with little or no inherited TPMT activity are at increased risk for severe toxicity from conventional doses of mercaptopurine and generally require substantial dose reduction. TPMT genotyping or phenotyping can be used to identify patients with absent or reduced TPMT activity. TPMT testing cannot substitute for haematological monitoring in patients receiving mercaptopurine. The optimal starting dose for homozygous deficient patients has not been established.
Dosage considerations
Mercaptopurine may be taken with food or on an empty stomach, but patients should standardise the method of administration. The dose should not be taken with milk or dairy products. Mercaptopurine should be taken at least 1 hour before or 2 hours after milk or dairy products.
6-mercaptopurine displays diurnal variation in pharmacokinetics and efficacy. Administration in the evening compared to morning administration may lower the risk of relapse. Therefore the daily dose of mercaptopurine should be taken in the evening.
Liability Disclaimer : RxReasoner has utilized reasonable care in providing content and services that are accurate, complete and up to date. However, RxReasoner does not accept any responsibility or liability about it. The content and services of RxReasoner are for informational purposes only and they are not intended to be a substitute for the knowledge, expertise, skill, and judgment of physicians, pharmacists, nurses, or other healthcare professionals involved in patient care. RxReasoner offers no medical advice. Users are responsible for the use of the provided content. A shown indication or treatment should not be construed to indicate that the medication is safe, appropriate, or effective in any given patient or under any particular circumstances. The absence of an indication or treatment should not roule out the existence of other appropriate medications. Always seek the advice of a physician or other qualified health provider with any questions you may have regarding a medical condition or medicament. RxReasoner is not liable for any damages allegedly sustained arising out of the use of its content and services.