TECHNETIUM TC99M MERTIATIDE Kit for the preparation Ref.[10977] Active ingredients: Technetium ⁹⁹ᵐTc mertiatide
Source: FDA, National Drug Code (US) Revision Year: 2020
Product description
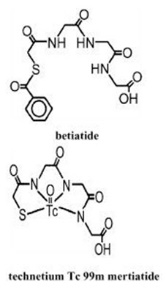
Kit for the Preparation of Technetium Tc99m Mertiatide is used for the preparation of technetium Tc 99m mertiatide, a diagnostic radiopharmaceutical. It is supplied as a sterile, nonpyrogenic, lyophilized powder. Each vial contains betiatide (N-[N-[N-[(benzoylthio) acetyl]glycyl]glycyl]glycine). After reconstitution with sterile sodium pertechnetate Tc 99m injection, the technetium Tc 99m mertiatide (disodium[N[N-[N-(mercaptoacetyl) glycyl]glycyl] glycinato (2-) - N,N′,N″,S′]oxotechnetate (2-)) which is formed is suitable for intravenous administration.
Each 10 milliliter vial contains 1 milligram betiatide, 0.05 milligram (minimum) stannous chloride dihydrate (SnCl2∙2H2O) and 0.2 milligram (maximum) total tin expressed as stannous chloride dihydrate (SnCl2∙2H2O), 40 milligrams sodium tartrate dihydrate (Na2C4H2O6∙2H2O), and 20 milligrams lactose monohydrate. Prior to lyophilization, sodium hydroxide or hydrochloric acid may be added for pH adjustment. The pH of the reconstituted drug is between 5.0 and 6.0. No bacteriostatic preservative is present. The contents are sealed under argon. Betiatide is light sensitive and must be protected from light.
Betiatide and technetium Tc 99m mertiatide have the following structural formulas:
PHYSICAL CHARACTERISTICS
Technetium Tc 99m decays by isomeric transition with a physical half-life of 6.02 hours. The principal photon that is useful for detection and imaging is listed in Table 1.
Table 1. Principal Radiation Emission Data*:
| Radiation M | Mean % per Disintegration | Energy (keV) |
|---|---|---|
| Gamma-2 | 89.07 | 140.5 |
* Kocher, David C., “Radioactive Decay Tables”, DOE/TIC-11026, 108 (1981).
The specific gamma ray constant for Technetium Tc 99m is 0.78 R/mCi-hr at 1 cm. The first half-value thickness of lead (Pb) for Technetium Tc 99m is 0.017 cm. A range of values for the relative attenuation of the radiation emitted by this radionuclide that results from interposition of various thicknesses of Pb is shown in Table 2. For example, the use of 0.25 cm of Pb will decrease the external radiation exposure by a factor of about 1000.
Table 2. Radiation Attenuation by Lead Shielding:
| Shield Thickness (Pb) cm | Coefficient of Attenuation |
|---|---|
| 0.017 | 0.5 |
| 0.08 | 10-1 |
| 0.16 | 10-2 |
| 0.25 | 10-3 |
| 0.33 | 10-4 |
To correct for physical decay of the radionuclide, the fractions that remain at selected time intervals after the time of calibration are shown in Table 3.
Table 3. Physical Decay Chart: Technetium Tc 99m, Half-life 6.02 Hours:
| Hours | Fraction Remaining | Hours | Fraction Remaining |
|---|---|---|---|
| 0* | 1.000 | 7 | 0.447 |
| 1 | 0.891 | 8 | 0.398 |
| 2 | 0.794 | 9 | 0.355 |
| 3 | 0.708 | 10 | 0.316 |
| 4 | 0.631 | 11 | 0.282 |
| 5 | 0.562 | 12 | 0.251 |
| 6 | 0.501 |
* Calibration Time
| How Supplied |
|---|
|
Kit for the Preparation of Technetium Tc 99m Mertiatide is supplied as a lyophilized powder packaged in vials. Each reaction vial contains 1 mg betiatide, 0.05 mg (minimum) stannous chloride dihydrate (SnCl2∙2H2O), 0.2 mg (maximum) total tin expressed as stannous chloride dihydrate (SnCl2∙2H2O), 40 mg sodium tartrate dihydrate (Na2C4H2O6∙2H2O), and 20 mg lactose monohydrate. The pH of the reconstituted drug is between 5.0 and 6.0. No bacteriostatic preservative is present. Packages containing 5 reaction vials (NDC 45567-0655-1) are available. Sep-Pak is a trademark of Waters Technologies Corporation. Manufactured by: Sun Pharmaceutical Industries, Inc., Billerica, MA 01821 USA |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.