INLYTA Film-coated tablet Ref.[6318] Active ingredients: Axitinib
Source: European Medicines Agency (EU) Revision Year: 2019 Publisher: Pfizer Europe MA EEIG, Boulevard de la Plaine 17, 1050, Bruxelles, Belgium
Contraindications
Hypersensitivity to axitinib or to any of the excipients listed in section 6.1.
Special warnings and precautions for use
Specific safety events should be monitored before initiation of, and periodically throughout, treatment with axitinib as described below.
Cardiac failure events
In clinical studies with axitinib for the treatment of patients with RCC, cardiac failure events (including cardiac failure, cardiac failure congestive, cardiopulmonary failure, left ventricular dysfunction, ejection fraction decreased, and right ventricular failure) were reported (see section 4.8).
Signs or symptoms of cardiac failure should periodically be monitored throughout treatment with axitinib. Management of cardiac failure events may require temporary interruption or permanent discontinuation and/or dose reduction of axitinib therapy.
Hypertension
In clinical studies with axitinib for the treatment of patients with RCC, hypertension was very commonly reported (see section 4.8).
In a controlled clinical study, the median onset time for hypertension (systolic blood pressure >150 mmHg or diastolic blood pressure > 100 mmHg) was within the first month of the start of axitinib treatment and blood pressure increases have been observed as early as 4 days after starting axitinib.
Blood pressure should be well-controlled prior to initiating axitinib. Patients should be monitored for hypertension and treated as needed with standard antihypertensive therapy. In the case of persistent hypertension, despite use of antihypertensive medicinal products, the axitinib dose should be reduced. For patients who develop severe hypertension, temporarily interrupt axitinib and restart at a lower dose once the patient is normotensive. If axitinib is interrupted, patients receiving antihypertensive medicinal products should be monitored for hypotension (see section 4.2).
In case of severe or persistent arterial hypertension and symptoms suggestive of posterior reversible encephalopathy syndrome (PRES) (see below), a diagnostic brain magnetic resonance image (MRI) should be considered.
Thyroid dysfunction
In clinical studies with axitinib for the treatment of patients with RCC, events of hypothyroidism and, to a lesser extent, hyperthyroidism, were reported (see section 4.8).
Thyroid function should be monitored before initiation of, and periodically throughout, treatment with axitinib. Hypothyroidism or hyperthyroidism should be treated according to standard medical practice to maintain euthyroid state.
Arterial embolic and thrombotic events
In clinical studies with axitinib, arterial embolic and thrombotic events (including transient ischemic attack, myocardial infarction, cerebrovascular accident and retinal artery occlusion) were reported (see section 4.8).
Axitinib should be used with caution in patients who are at risk for, or who have a history of, these events. Axitinib has not been studied in patients who had an arterial embolic or thrombotic event within the previous 12 months.
Venous embolic and thrombotic events
In clinical studies with axitinib, venous embolic and thrombotic events (including pulmonary embolism, deep vein thrombosis, and retinal vein occlusion/thrombosis) were reported (see section 4.8).
Axitinib should be used with caution in patients who are at risk for, or who have a history of, these events. Axitinib has not been studied in patients who had a venous embolic or thrombotic event within the previous 6 months.
Elevation of haemoglobin or haematocrit
Increases in haemoglobin or haematocrit, reflective of increases in red blood cell mass, may occur during treatment with axitinib (see section 4.8, polycythaemia). An increase in red blood cell mass may increase the risk of embolic and thrombotic events.
Haemoglobin or haematocrit should be monitored before initiation of, and periodically throughout, treatment with axitinib. If haemoglobin or haematocrit becomes elevated above the normal level, patients should be treated according to standard medical practice to decrease haemoglobin or haematocrit to an acceptable level.
Haemorrhage
In clinical studies with axitinib, haemorrhagic events were reported (see section 4.8).
Axitinib has not been studied in patients who have evidence of untreated brain metastasis or recent active gastrointestinal bleeding, and should not be used in those patients. If any bleeding requires medical intervention, temporarily interrupt the axitinib dose.
Aneurysms and artery dissections
The use of VEGF pathway inhibitors in patients with or without hypertension may promote the formation of aneurysms and/or artery dissections. Before initiating Inlyta, this risk should be carefully considered in patients with risk factors such as hypertension or history of aneurysm.
Gastrointestinal perforation and fistula formation
In clinical studies with axitinib, events of gastrointestinal perforation and fistulas were reported (see section 4.8).
Symptoms of gastrointestinal perforation or fistula should be periodically monitored for throughout treatment with axitinib.
Wound healing complications
No formal studies of the effect of axitinib on wound healing have been conducted.
Treatment with axitinib should be stopped at least 24 hours prior to scheduled surgery. The decision to resume axitinib therapy after surgery should be based on clinical judgment of adequate wound healing.
Posterior reversible encephalopathy syndrome (PRES)
In clinical studies with axitinib, events of PRES were reported (see section 4.8).
PRES is a neurological disorder which can present with headache, seizure, lethargy, confusion, blindness and other visual and neurologic disturbances. Mild to severe hypertension may be present. Magnetic resonance imaging is necessary to confirm the diagnosis of PRES. In patients with signs or symptoms of PRES, temporarily interrupt or permanently discontinue axitinib treatment. The safety of reinitiating axitinib therapy in patients previously experiencing PRES is not known.
Proteinuria
In clinical studies with axitinib, proteinuria, including that of Grade 3 and 4 severity, was reported (see section 4.8).
Monitoring for proteinuria before initiation of, and periodically throughout, treatment with axitinib is recommended. For patients who develop moderate to severe proteinuria, reduce the dose or temporarily interrupt axitinib treatment (see section 4.2). Axitinib should be discontinued if the patient develops nephrotic syndrome.
Liver-related adverse reactions
In a controlled clinical study with axitinib for the treatment of patients with RCC, liver-related adverse reactions were reported. The most commonly reported liver-related adverse reactions included increases in alanine aminotransferase (ALT), aspartate aminotransferase (AST), and blood bilirubin (see section 4.8). No concurrent elevations of ALT (>3 times the upper limit of normal [ULN]) and bilirubin (>2 times the ULN) were observed.
In a clinical dose-finding study, concurrent elevations of ALT (12 times the ULN) and bilirubin (2.3 times the ULN), considered to be drug-related hepatotoxicity, were observed in 1 patient who received axitinib at a starting dose of 20 mg twice daily (4 times the recommended starting dose).
Liver function tests should be monitored before initiation of, and periodically throughout, treatment with axitinib.
Hepatic impairment
In clinical studies with axitinib, the systemic exposure to axitinib was approximately two-fold higher in subjects with moderate hepatic impairment (Child-Pugh class B) compared to subjects with normal hepatic function. A dose decrease is recommended when administering axitinib to patients with moderate hepatic impairment (Child-Pugh class B) (see section 4.2).
Axitinib has not been studied in patients with severe hepatic impairment (Child-Pugh class C) and should not be used in this population.
Elderly (≥65 years) and race
In a controlled clinical study with axitinib for the treatment of patients with RCC, 34% of patients treated with axitinib were ≥65 years of age. The majority of patients were White (77%) or Asian (21%). Although greater sensitivity to develop adverse reactions in some older patients and Asian patients cannot be ruled out, overall, no major differences were observed in the safety and effectiveness of axitinib between patients who were ≥65 years of age and non-elderly, and between White patients and patients of other races.
No dosage adjustment is required on the basis of patient age or race (see sections 4.2 and 5.2).
Lactose
This medicinal product contains lactose. Patients with rare hereditary problems of galactose intolerance, Lapp lactase deficiency or glucose-galactose malabsorption should not take this medicinal product.
Interaction with other medicinal products and other forms of interaction
In vitro data indicate that axitinib is metabolised primarily by CYP3A4/5 and, to a lesser extent, CYP1A2, CYP2C19, and uridine diphosphate-glucuronosyltransferase (UGT) 1A1.
CYP3A4/5 inhibitors
Ketoconazole, a strong inhibitor of CYP3A4/5, administered at a dose of 400 mg once daily for 7 days, increased the mean area under the curve (AUC) 2-fold and Cmax 1.5-fold of a single 5-mg oral dose of axitinib in healthy volunteers. Co-administration of axitinib with strong CYP3A4/5 inhibitors (e.g. ketoconazole, itraconazole, clarithromycin, erythromycin, atazanavir, indinavir, nefazodone, nelfinavir, ritonavir, saquinavir, and telithromycin) may increase axitinib plasma concentrations. Grapefruit may also increase axitinib plasma concentrations. Selection of concomitant medicinal products with no or minimal CYP3A4/5 inhibition potential is recommended. If a strong CYP3A4/5 inhibitor must be co-administered, a dose adjustment of axitinib is recommended (see section 4.2).
CYP1A2 and CYP2C19 inhibitors
CYP1A2 and CYP2C19 constitute minor (<10%) pathways in axitinib metabolism. The effect of strong inhibitors of these isozymes on axitinib pharmacokinetics has not been studied. Caution should be exercised due to the risk of increased axitinib plasma concentrations in patients taking strong inhibitors of these isozymes.
CYP3A4/5 inducers
Rifampicin, a strong inducer of CYP3A4/5, administered at a dose of 600 mg once daily for 9 days, reduced the mean AUC by 79% and Cmax by 71% of a single 5 mg dose of axitinib in healthy volunteers.
Co-administration of axitinib with strong CYP3A4/5 inducers (e.g. rifampicin, dexamethasone, phenytoin, carbamazepine, rifabutin, rifapentin, phenobarbital, and Hypericum perforatum [St. John's wort]) may decrease axitinib plasma concentrations. Selection of concomitant medicinal products with no or minimal CYP3A4/5 induction potential is recommended. If a strong CYP3A4/5 inducer must be co-administered, a dose adjustment of axitinib is recommended (see section 4.2).
In vitro studies of CYP and UGT inhibition and induction
In vitro studies indicated that axitinib does not inhibit CYP2A6, CYP2C9, CYP2C19, CYP2D6, CYP2E1, CYP3A4/5, or UGT1A1 at therapeutic plasma concentrations.
In vitro studies indicated that axitinib has a potential to inhibit CYP1A2. Therefore, co- administration of axitinib with CYP1A2 substrates may result in increased plasma concentrations of CYP1A2 substrates (e.g. theophylline).
In vitro studies also indicated that axitinib has the potential to inhibit CYP2C8. However, co-administration of axitinib with paclitaxel, a known CYP2C8 substrate, did not result in increased plasma concentrations of paclitaxel in patients with advanced cancer, indicating lack of clinical CYP2C8 inhibition.
In vitro studies in human hepatocytes also indicated that axitinib does not induce CYP1A1, CYP1A2, or CYP3A4/5. Therefore co-administration of axitinib is not expected to reduce the plasma concentration of co-administered CYP1A1, CYP1A2, or CYP3A4/5 substrates in vivo.
In vitro studies with P-glycoprotein
In vitro studies indicated that axitinib inhibits P-glycoprotein. However, axitinib is not expected to inhibit P-glycoprotein at therapeutic plasma concentrations. Therefore, co-administration of axitinib is not expected to increase the plasma concentration of digoxin, or other P-glycoprotein substrates, in vivo.
Fertility, pregnancy and lactation
Pregnancy
There are no data regarding the use of axitinib in pregnant women. Based on the pharmacological properties of axitinib, it may cause foetal harm when administered to a pregnant woman. Studies in animals have shown reproductive toxicity including malformations (see section 5.3). Axitinib should not be used during pregnancy unless the clinical condition of the woman requires treatment with this medicinal product.
Women of childbearing potential must use effective contraception during and up to 1 week after treatment.
Breast-feeding
It is unknown whether axitinib is excreted in human milk. A risk to the suckling child cannot be excluded. Axitinib should not be used during breast-feeding.
Fertility
Based on non-clinical findings, axitinib has the potential to impair reproductive function and fertility in humans (see section 5.3).
Effects on ability to drive and use machines
Axitinib has minor influence on the ability to drive and use machines. Patients should be advised that they may experience events such as dizziness and/or fatigue during treatment with axitinib.
Undesirable effects
Summary of the safety profile
The following risks, including appropriate action to be taken, are discussed in greater detail in section 4.4:cardiac failure events, hypertension, thyroid dysfunction, arterial thromboembolic events, venous thromboembolic events, elevation of haemoglobin or haematocrit, haemorrhage, gastrointestinal perforation and fistula formation, wound healing complications, PRES, proteinuria, and elevation of liver enzymes.
The most common (≥20%) adverse reactions observed following treatment with axitinib were diarrhoea, hypertension, fatigue, decreased appetite, nausea, weight decreased, dysphonia, palmar-plantar erythrodysaesthesia (hand-foot) syndrome, haemorrhage, hypothyroidism, vomiting, proteinuria, cough, and constipation.
Tabulated list of adverse reactions
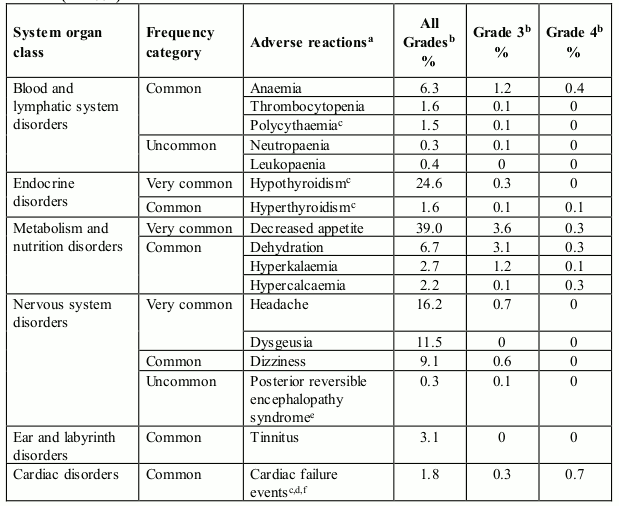
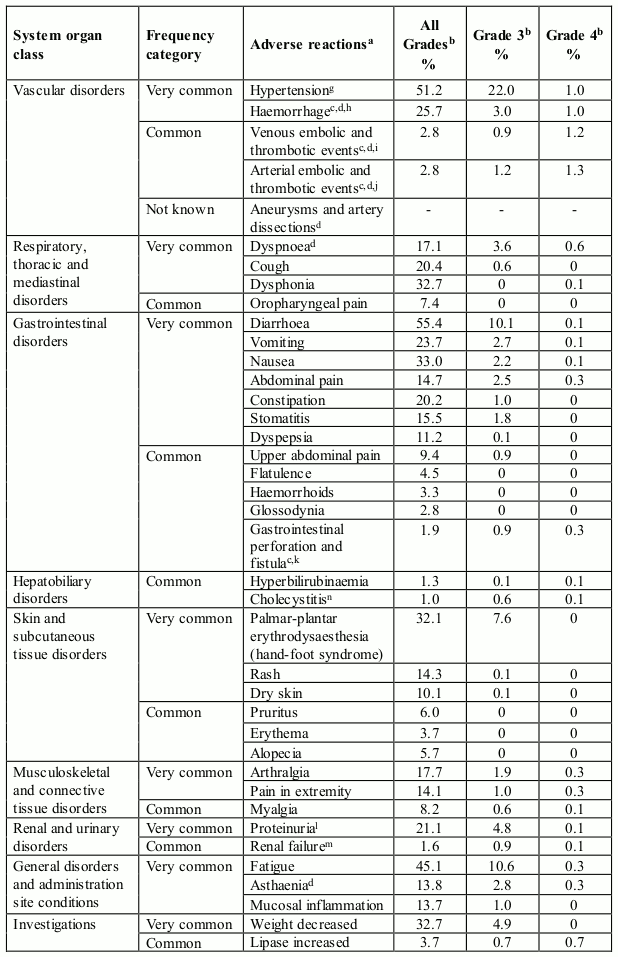
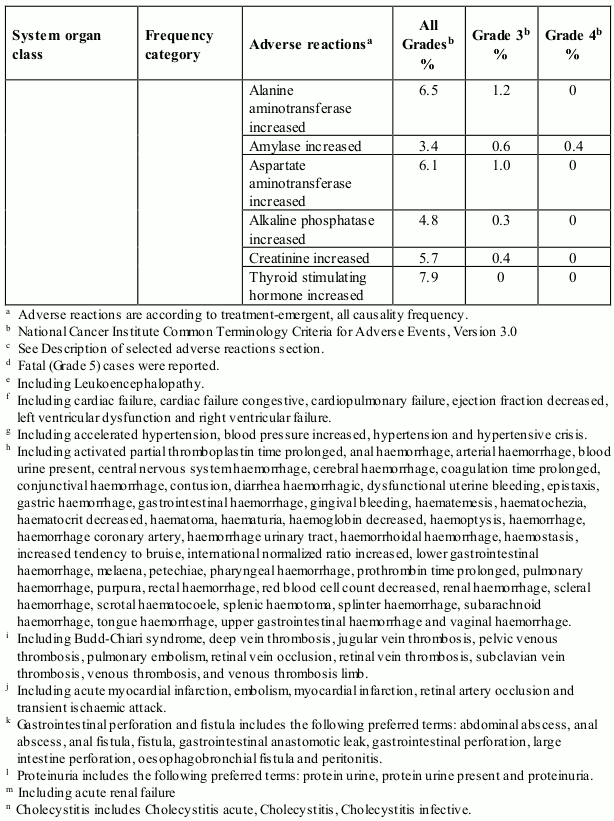
Table 1 presents adverse reactions reported in a pooled dataset of 672 patients who received axitinib in clinical studies for the treatment of patients with RCC (see section 5.1). Post-marketing adverse reactions identified in clinical studies are also included.
The adverse reactions are listed by system organ class, frequency category and grade of severity. Frequency categories are defined as: very common (≥1/10), common (≥1/100 to <1/10), uncommon (≥1/1,000 to <1/100), rare (≥1/10,000 to <1/1,000), very rare (<1/10,000), and not known (cannot be estimated from the available data). The current safety database for axitinib is too small to detect rare and very rare adverse reactions.
Categories have been assigned based on absolute frequencies in the pooled clinical studies data. Within each system organ class, adverse reactions with the same frequency are presented in order of decreasing seriousness.
Table 1. Adverse reactions reported in RCC studies in patients who received axitinib (N=672):
Description of selected adverse reactions
Cardiac failure events (see section 4.4)
In a controlled clinical study with axitinib (N=359) for the treatment of patients with RCC, cardiac failure events were reported in 1.7% patients receiving axitinib, including cardiac failure (0.6%), cardiopulmonary failure (0.6%), left ventricular dysfunction (0.3%), and right ventricular failure (0.3%). Grade 4 cardiac failure adverse reactions were reported in 0.6 % of patients receiving axitinib. Fatal cardiac failure was reported in 0.6 % of patients receiving axitinib.
In monotherapy studies with axitinib (N=672) for the treatment of patients with RCC, cardiac failure events (including cardiac failure, cardiac failure congestive, cardiopulmonary failure, left ventricular dysfunction, ejection fraction decreased, and right ventricular failure) were reported in 1.8% patients receiving axitinib. Grade ¾ cardiac failure events were reported in 1.0% patients and fatal cardiac failure events were reported in 0.3% patients receiving axitinib.
Thyroid dysfunction (see section 4.4)
In a controlled clinical study with axitinib for the treatment of patients with RCC, hypothyroidism was reported in 20.9% of patients and hyperthyroidism was reported in 1.1% of patients. Thyroid stimulating hormone (TSH) increased was reported as an adverse reaction in 5.3% of patients receiving axitinib. During routine laboratory assessments, in patients who had TSH <5 μU/mL before treatment, elevations of TSH to ≥10 μU/mL occurred in 32.2% of patients receiving axitinib.
In pooled clinical studies with axitinib (N=672) for the treatment of patients with RCC, hypothyroidism was reported in 24.6% of patients receiving axitinib. Hyperthyroidism was reported in 1.6% of patients receiving axitinib.
Venous embolic and thrombotic events (see section 4.4)
In a controlled clinical study with axitinib for the treatment of patients with RCC, venous embolic and thrombotic adverse reactions were reported in 3.9% of patients receiving axitinib, including pulmonary embolism (2.2%), retinal vein occlusion/thrombosis (0.6%) and deep vein thrombosis (0.6%). Grade ¾ venous embolic and thrombotic adverse reactions were reported in 3.1% of patients receiving axitinib. Fatal pulmonary embolism was reported in one patient (0.3%) receiving axitinib.
In pooled clinical studies with axitinib (N=672) for the treatment of patients with RCC, venous embolic and thrombotic events were reported in 2.8% of patients receiving axitinib. Grade 3 venous embolic and thrombotic events were reported in 0.9% of patients. Grade 4 venous embolic and thrombotic events were reported in 1.2% of patients. Fatal venous embolic and thrombotic events were reported 0.1% patients receiving axitinib.
Arterial embolic and thrombotic events (see section 4.4)
In a controlled clinical study with axitinib for the treatment of patients with RCC, arterial embolic and thrombotic adverse reactions were reported in 4.7% of patients receiving axitinib, including myocardial infarction (1.4%), transient ischemic attack (0.8%) and cerebrovascular accident (0.6%). Grade ¾ arterial embolic and thrombotic adverse reactions were reported in 3.3% of patients receiving axitinib. A fatal acute myocardial infarction and cerebrovascular accident was reported in one patient each (0.3%). In monotherapy studies with axitinib (N=850), arterial embolic and thrombotic adverse reactions (including transient ischemic attack, myocardial infarction, and cerebrovascular accident) were reported in 5.3% of patients receiving axitinib.
In pooled clinical studies with axitinib (N=672) for the treatment of patients with RCC, arterial embolic and thrombotic events were reported in 2.8% of patients receiving axitinib. Grade 3 arterial embolic and thrombotic events were reported in 1.2% of patients. Grade 4 arterial embolic and thrombotic events were reported in 1.3% of patients. Fatal arterial embolic and thrombotic events were reported in 0.3% patients receiving axitinib.
Polycythaemia (see Elevation of haemoglobin or haematocrit in section 4.4)
In a controlled clinical study with axitinib for the treatment of patients with RCC, polycythaemia was reported in 1.4% of patients receiving axitinib. Routine laboratory assessments detected elevated haemoglobin above ULN in 9.7% of patients receiving axitinib. In four clinical studies with axitinib for the treatment of patients with RCC (N=537), elevated haemoglobin above ULN was observed in 13.6% receiving axitinib.
In pooled clinical studies with axitinib (N=672) for the treatment of patients with RCC, polycythaemia was reported in 1.5% of patients receiving axitinib.
Haemorrhage (see section 4.4)
In a controlled clinical study with axitinib for the treatment of patients with RCC that excluded patients with untreated brain metastasis, haemorrhagic adverse reactions were reported in 21.4% of patients receiving axitinib. The haemorrhagic adverse reactions in patients treated with axitinib included epistaxis (7.8%), haematuria (3.6%), haemoptysis (2.5%), rectal haemorrhage (2.2%), gingival bleeding (1.1%), gastric haemorrhage (0.6%), cerebral haemorrhage (0.3%) and lower gastrointestinal haemorrhage (0.3%). Grade >3 haemorrhagic adverse reactions were reported in 3.1% of patients receiving axitinib (including cerebral haemorrhage, gastric haemorrhage, lower gastrointestinal haemorrhage and haemoptysis). Fatal haemorrhage was reported in one patient (0.3%) receiving axitinib (gastric haemorrhage). In monotherapy studies with axitinib (N=850), haemoptysis was reported in 3.9% of patients; Grade >3 haemoptysis was reported in 0.5% of patients.
In pooled clinical studies with axitinib (N=672) for the treatment of patients with RCC, haemorrhagic events were reported in 25.7% of patients receiving axitinib. Grade 3 haemorrhagic adverse reactions were reported in 3% of patients. Grade 4 haemorrhagic adverse reactions were reported in 1% of patients and fatal haemorrhage were reported in 0.4% of patients receiving axitinib.
Gastrointestinal perforation and fistula formation (see section 4.4)
In a controlled clinical study with axitinib for the treatment of patients with RCC, gastrointestinal perforation-type events were reported in 1.7% of patients receiving axitinib, including anal fistula (0.6%), fistula (0.3%) and gastrointestinal perforation (0.3%).In monotherapy studies with axitinib (N=850), gastrointestinal perforation-type events were reported in 1.9% of patients and fatal gastrointestinal perforation was reported in one patient (0.1%).
In pooled clinical studies with axitinib (N=672) for the treatment of patients with RCC, gastrointestinal perforation and fistula were reported in 1.9% of patients receiving axitinib.
Reporting of suspected adverse reactions
Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the national reporting system listed in Appendix V.
Incompatibilities
Not applicable.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.