ANNOVERA Vaginal system (Ring) Ref.[10392] Active ingredients: 17 alpha-Ethinylestradiol
Source: FDA, National Drug Code (US) Revision Year: 2020
12.1. Mechanism of Action
CHCs lower the risk of becoming pregnant primarily by suppressing ovulation.
12.2. Pharmacodynamics
Cardiac Electrophysiology
The effect of SA on the QTc interval was evaluated in a Phase 1 randomized, placebo and positive controlled, double-blind, single-dose, three-period, crossover thorough QTc study in 44 healthy adult female subjects. At the single intravenous bolus dose which produces 4.5-fold the therapeutic serum concentrations of SA achieved with ANNOVERA, SA did not prolong the QTc interval to any clinically relevant extent.
12.3. Pharmacokinetics
Absorption
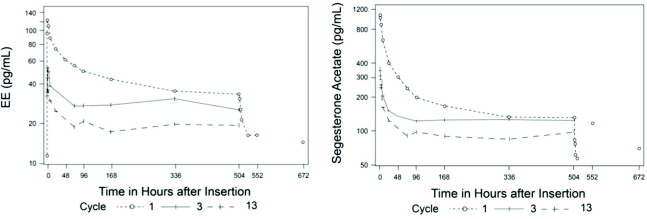
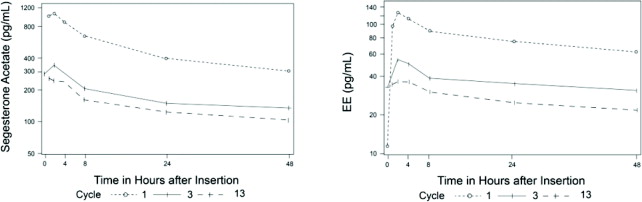
The pharmacokinetics (PK) of ANNOVERA were determined in 39 women who used ANNOVERA for up to 13 cycles. Following vaginal administration, SA and EE were absorbed into systemic circulation with median tmax of about 2 hours in Cycle 1, Cycle 3, and Cycle 13. Concentrations of both components declined after tmax and became more constant after 96 hours post-dose. Over subsequent cycles of use, the peak serum concentrations of SA and EE declined. Serum concentration-time profiles of SA and EE for Cycles 1, 3, and 13 of ANNOVERA use are provided in Figure 2 and Figure 3 with PK parameters summarized in Table 5 and Table 6.
Figure 2. Mean SA and EE Serum Concentrations Delivered by ANNOVERA Over 21 Days of Dosing for Cycles 1, 3, and 13:
Figure 3. Mean SA and EE Serum Concentrations Delivered by ANNOVERA Over the First 48 Hours of Dosing for Cycles 1, 3, and 13:
Table 5. Mean (SD) PK Parameters for SA following ANNOVERA Administration:
| Cycle | AUC0-21 day (ng*hr/mL) | AUC0-1 day (ng*hr/mL) | Cmax (pg/mL) | Cavg (pg/mL) |
|---|---|---|---|---|
| 1 | 96.2 (16.9) | 15 (3.2) | 1,147 (315) | 191 (34) |
| 3 | 65.9 (14.8) | 5 (1.6) | 363 (133) | 131 (29) |
| 13 | 47.2 (10.1) | 3.9 (1.4) | 294 (116) | 94 (20) |
Table 6. Mean (SD) PK Parameters for EE following ANNOVERA Administration:
| Cycle | AUC0-21 day (ng*hr/mL) | AUC0-1 day (ng*hr/mL) | Cmax (pg/mL) | Cavg (pg/mL) |
|---|---|---|---|---|
| 1 | 22.2 (9.8) | 2.1 (0.7) | 129 (39) | 44 (19) |
| 3 | 14.7 (4.7) | 0.9 (0.4) | 60 (32) | 29 (9) |
| 13 | 9.6 (4.1) | 0.7 (0.3) | 39 (16) | 19 (8) |
Distribution
The volume of distribution of SA is 19.6 L/kg. SA is approximately 95% bound to human serum proteins and has negligible binding affinity for sex hormone-binding globulin (SHBG). EE is highly protein bound but not specifically bound to serum albumin (98.5%) and induces an increase in the serum concentrations of SHBG.
Metabolism
In vitro data show that both SA and EE are metabolized by the cytochrome P450 (CYP) 3A4 isoenzyme. In human serum, two oxidative metabolites (5α-dihydro- and 17α-hydroxy-5α-dihydro metabolites) constitute 50% of exposure relative to SA. Both metabolites are not considered as active metabolites with EC50 to progesterone receptor 10-fold higher than that of SA. EE is primarily metabolized by aromatic hydroxylation, but a wide variety of hydroxylated and methylated metabolites are formed. These are present as free metabolites and as sulfate and glucuronide conjugates. The hydroxylated EE metabolites have weak estrogenic activity.
Excretion
The mean (SD) half-life of SA is 4.5 (3.4) hours. EE is known to be excreted in the urine and feces as glucuronide and sulfate conjugates, and it undergoes enterohepatic recirculation. The mean (SD) half-life of EE is 15.1 (7.5) hours.
Specific Populations
Body Mass Index (BMI)
Higher body weight associates with lower systemic exposure of SA and EE. In a PK study conducted in 18 females with BMI <25 (16.89-24.34) kg/m2 and 21 females with BMI >25 (25.15-37.46) kg/m2, up to 16% and 33% decreases in the systemic exposure (AUC0-21day) of SA and EE, respectively, were observed between the two BMI groups.
Interaction with Vaginal Medications
A clinical drug-drug interaction (DDI) study was conducted to evaluate the effect of vaginal antimycotic medication (miconazole nitrate) on the PK of SA and EE in 29 females using ANNOVERA. The results showed that a single-dose vaginal administration of 1,200 mg miconazole suppository on Day 8 of ANNOVERA use increased the systemic exposure of EE (AUCDay8-21) by approximately 67%. A similar trend was observed with SA with AUCDay8-9, AUCDay8-10, and AUCDay8-21 increasing by approximately 30%, 32%, and 19%, respectively. When 200 mg miconazole vaginal suppositories were administered on Day 8, Day 9, and Day 10 of ANNOVERA use, EE AUCDay8-11 and AUCDay8-21 were increased by 9% and 42%, respectively. SA AUCDay8-11 and AUCDay8-21 were increased by 28% and 27%, respectively. Water-based vaginal miconazole cream had no effect on ANNOVERA [see Drug Interactions (7.3)].
The in vitro studies suggest that SA is unlikely to inhibit or induce CYP enzymes at the therapeutic dose.
13.1. Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
In a 2-year carcinogenicity study in rats with subdermal implants releasing 40, 100, and 200 mcg segesterone acetate per day (approximately 17-86 times the daily dose of segesterone acetate in females using ANNOVERA, based on body surface area), no drug-related increase in tumor incidence was observed. In a 2-year intravaginal carcinogenicity study in mice, segesterone acetate gel produced an increased incidence of adenocarcinoma and lobular hyperplasia in the breast at a dose of 30 mg/kg/day, approximately 10 times the systemic exposure of segesterone acetate per day in females using ANNOVERA, based on AUC. A dose of 10 mg/kg/day in the mouse, approximately 3 times the systemic exposure of segesterone acetate per day based on AUC, did not result in carcinogenic findings.
Long-term continuous administration of natural and synthetic estrogens in certain animal species increases the frequency of carcinomas of the breast, uterus, cervix, vagina, testis, and liver.
Mutagenesis
Segesterone acetate was neither mutagenic nor clastogenic in the Ames/Salmonella reverse mutation assay, the chromosomal aberration assay in Chinese hamster ovary cells, or in the in vivo mouse micronucleus test.
Impairment of Fertility
A return to fertility study was conducted with segesterone acetate in rats, using subdermal implants releasing a dose approximately 25 times the anticipated daily vaginal human dose (based on body surface area). Three months of treatment with segesterone acetate suppressed fertility, but 7 weeks after cessation of treatment, there were no adverse effects on ovulation or resulting litter parameters.
14. Clinical Studies
The efficacy of ANNOVERA was evaluated in two 1-year multicenter trials enrolling 2,265 females, age 18-40 years, who were healthy and sexually active with regular menstrual cycles. The trials were conducted in the U.S., Dominican Republic, Brazil, Chile, Finland, Hungary, Sweden, and Australia, with 67.1% of females from the U.S. The racial/ethnic distribution was Caucasian (71.2%), African-American (14.1%), Asian (3.5%), other/multiple races (11.2%); 28.7% of the study population was Hispanic. The mean age was 26.7 years and the mean (range) BMI was 24.1 (16.0, 41.5) kg/m2. At approximately 50% enrollment, women with BMI >29.0 kg/m2 were no longer enrolled in the two trials and all women with a BMI >29.0 kg/m2 were discontinued from the trials.
Based on pooled data from the two trials, 2,111 females ≤35 years of age completed 17,427 evaluable 28-day cycles (cycles in which no back-up contraception was used). The pooled pregnancy rate, evaluated by the Pearl Index (PI), was 2.98 (95% Confidence Interval [2.13, 4.06]) per 100 woman-years of ANNOVERA use.
Return to fertility was assessed in 290 of the subjects in the two trials who either desired pregnancy or switched to a nonhormonal method after the trials, and all 290 subjects reported a return to fertility during the 6-month follow-up period (defined as a return of menses or pregnancy).
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.