Source: European Medicines Agency (EU) Revision Year: 2018 Publisher: Aspen Pharma Trading Limited, 3016 Lake Drive Citywest Business Campus, Dublin 24, Ireland
Fondaparinux is intended for subcutaneous use only. Do not administer intramuscularly.
Fondaparinux should be used with caution in patients who have an increased risk of haemorrhage, such as those with congenital or acquired bleeding disorders (e.g. platelet count <50,000/mm³), active ulcerative gastrointestinal disease and recent intracranial haemorrhage or shortly after brain, spinal or ophthalmic surgery and in special patient groups as outlined below.
For prevention of VTE: Agents that may enhance the risk of haemorrhage should not be administered concomitantly with fondaparinux. These agents include desirudin, fibrinolytic agents, GP IIb/IIIa receptor antagonists, heparin, heparinoids, or Low Molecular Weight Heparin (LMWH). When required, concomitant therapy with vitamin K antagonist should be administered in accordance with the information of Section 4.5. Other antiplatelet medicinal products (acetylsalicylic acid, dipyridamole, sulfinpyrazone, ticlopidine or clopidogrel), and NSAIDs should be used with caution. If co-administration is essential, close monitoring is necessary.
For treatment of superficial-vein thrombosis: Fondaparinux should be used with caution in patients who are being treated concomitantly with other medicinal products that increase the risk of haemorrhage.
Presence of superficial-vein thrombosis greater than 3 cm from the sapheno-femoral junction should be confirmed and concomitant DVT should be excluded by compression ultrasound or objective methods prior to initiating treatment with fondaparinux. There are no data regarding the use of fondaparinux 2.5 mg in superficial-vein thrombosis patients with concomitant DVT or with superficial-vein thrombosis within 3 cm of the sapheno-femoral junction (see section 4.2 and 5.1).
The safety and efficacy of fondaparinux 2.5 mg has not been studied in the following groups: patients with superficial-vein thrombosis following sclerotherapy or resulting as a complication of an intravenous line, patients with history of superficial-vein thrombosis within the previous 3 months, patients with history of venous thromboembolic disease within the previous 6 months, or patients with active cancer (see section 4.2 and 5.1).
In patients undergoing major orthopaedic surgery, epidural or spinal haematomas that may result in long-term or permanent paralysis cannot be excluded with the concurrent use of fondaparinux and spinal/epidural anaesthesia or spinal puncture. The risk of these rare events may be higher with postoperative use of indwelling epidural catheters or the concomitant use of other medicinal products affecting haemostasis.
The elderly population is at increased risk of bleeding. As renal function is generally decreasing with age, elderly patients may show reduced elimination and increased exposure of fondaparinux (see section 5.2). Fondaparinux should be used with caution in elderly patients (see section 4.2).
Prevention of VTE: Patients with body weight <50 kg are at increased risk of bleeding. Elimination of fondaparinux decreases with weight. Fondaparinux should be used with caution in these patients (see section 4.2).
Treatment of superficial-vein thrombosis: There are no clinical data available for the use of fondaparinux for the treatment of superficial-vein thrombosis in patients with body weight less than 50kg. Therefore, fondaparinux is not recommended for treatment of superficial-vein thrombosis in these patients (see section 4.2).
*Prevention of VTE:*Fondaparinux is known to be mainly excreted by the kidney. Patients with creatinine clearance <50 ml/min are at increased risk of bleeding and VTE and should be treated with caution (see sections 4.2, 4.3 and 5.2). There are limited clinical data available from patients with creatinine clearance less than 30 ml/min.
Treatment of superficial-vein thrombosis: Fondaparinux should not be used in patients with creatinine clearance <20 ml/min (see section 4.3). The dose should be reduced to 1.5 mg once daily in patients with creatinine clearance in the range of 20 to 50 ml/min (see sections 4.2 and 5.2). The safety and efficacy of 1.5 mg has not been studied.
Prevention of VTE: Dosing adjustment of fondaparinux is not necessary. However, the use of fondaparinux should be considered with caution because of an increased risk of bleeding due to a deficiency of coagulation factors in patients with severe hepatic impairment (see section 4.2).
Treatment of superficial-vein thrombosis: There are no clinical data available for the use of fondaparinux for the treatment of superficial-vein thrombosis in patients with severe hepatic impairment. Therefore, fondaparinux is not recommended for the treatment of superficial-vein thrombosis in these patients (see section 4.2).
Fondaparinux should be used with caution in patients with a history of HIT. The efficacy and safety of fondaparinux have not been formally studied in patients with HIT type II. Fondaparinux does not bind to platelet factor 4 and does not usually cross-react with sera from patients with Heparin Induced Thrombocytopenia (HIT) type II. However, rare spontaneous reports of HIT in patients treated with fondaparinux have been received.
The needle shield of the pre-filled syringe contains dry natural latex rubber that has the potential to cause allergic reactions in latex sensitive individuals.
Bleeding risk is increased with concomitant administration of fondaparinux and agents that may enhance the risk of haemorrhage (see section 4.4).
Oral anticoagulants (warfarin), platelet inhibitors (acetylsalicylic acid), NSAIDs (piroxicam) and digoxin did not interact with the pharmacokinetics of fondaparinux. The fondaparinux dose (10 mg) in the interaction studies was higher than the dose recommended for the present indications. Fondaparinux neither influenced the INR activity of warfarin, nor the bleeding time under acetylsalicylic acid or piroxicam treatment, nor the pharmacokinetics of digoxin at steady state.
Follow-up therapy with another anticoagulant medicinal product
If follow-up treatment is to be initiated with heparin or LMWH, the first injection should, as a general rule, be given one day after the last fondaparinux injection.
If follow up treatment with a Vitamin K antagonist is required, treatment with fondaparinux should be continued until the target INR value has been reached.
There are no adequate data from the use of fondaparinux in pregnant women. Animal studies are insufficient with respect to effects on pregnancy, embryo/foetal development, parturition and postnatal development because of limited exposure. Fondaparinux should not be prescribed to pregnant women unless clearly necessary.
Fondaparinux is excreted in rat milk but it is not known whether fondaparinux is excreted in human milk. Breast-feeding is not recommended during treatment with fondaparinux. Oral absorption by the child is however unlikely.
There are no data available on the effect of fondaparinux on human fertility. Animal studies do not show any effect on fertility.
No studies on the effect on the ability to drive and to use machines have been performed.
The most commonly reported serious adverse reactions reported with fondaparinux are bleeding complications (various sites including rare cases of intracranial/intracerebral and retroperitoneal bleedings) and anaemia. Fondaparinux should be used with caution in patients who have an increased risk of haemorrhage (see section 4.4).
The safety of fondaparinux 2.5 mg has been evaluated in 3,595 patients undergoing major orthopaedic surgery of the lower limbs treated up to 9 days, in 327 patients undergoing hip fracture surgery treated for 3 weeks following an initial prophylaxis of 1 week, 1,407 patients undergoing abdominal surgery treated up to 9 days, and in 425 medical patients who are at risk for thromboembolic complications treated up to 14 days.
The adverse reactions reported by the investigator as at least possibly related to fondaparinux are presented within each frequency grouping (very common ≥1/10; common: ≥1/100 to <1/10; uncommon: ≥1/1,000 to <1/100; rare: ≥1/10,000 to <1/1,000; very rare <1/10,000) and system organ class by decreasing order of seriousness; these adverse reactions should be interpreted within the surgical and medical context.
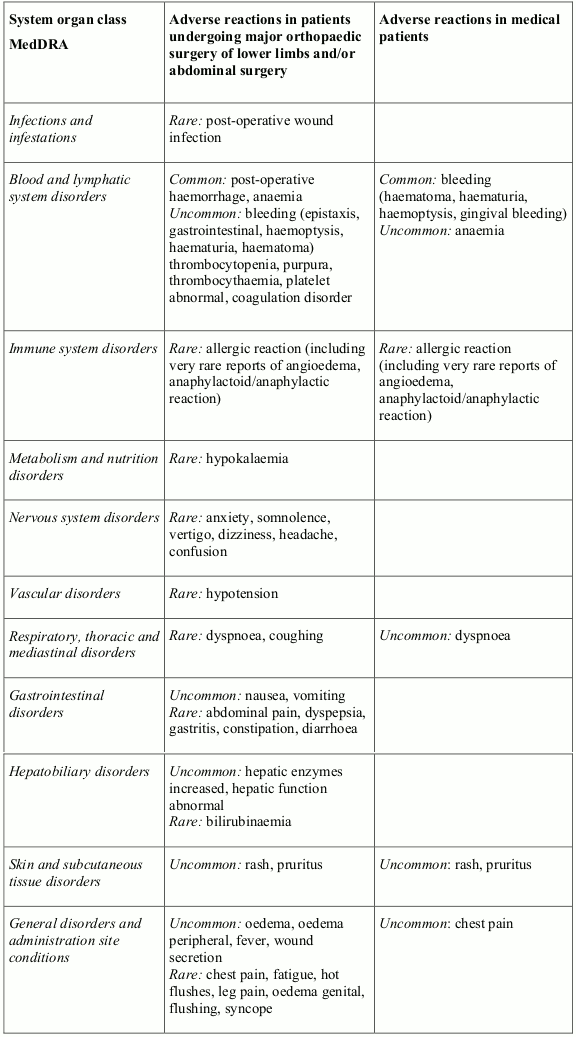
In other studies or in post-marketing experience, rare cases of intracranial/intracerebral and retroperitoneal bleedings have been reported.
Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the national reporting system listed in Appendix V.
In the absence of compatibility studies, this medicinal product must not be mixed with other medicinal products.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.