AUCATZYL Dispersion for infusion Ref.[115571] Active ingredients: Obecabtagene autoleucel
Source: European Medicines Agency (EU) Revision Year: 2025 Publisher: Autolus GmbH, Im Schwarzenbach 4, 79576 Weil am Rhein, Germany
4.1. Therapeutic indications
Aucatzyl is indicated for the treatment of adult patients 26 years of age and above with relapsed or refractory (r/r) B cell precursor acute lymphoblastic leukaemia (B ALL).
4.2. Posology and method of administration
Aucatzyl must be administered in a qualified treatment centre by a physician with experience in the treatment of haematological malignancies and trained for administration and management of patients treated with the medicinal product.
In the event of cytokine release syndrome (CRS), at least one dose of tocilizumab, and emergency equipment, must be available prior to infusion. The treatment centre must have access to additional doses of tocilizumab within 24 hours (see section 4.4). In the exceptional case where tocilizumab is not available (e.g., due to a shortage that is listed in the European Medicines Agency shortage catalogue), suitable alternative anti-interleukin (IL)-6 therapy (e.g., siltuximab) to treat CRS instead of tocilizumab must be available prior to infusion.
Posology
Aucatzyl is intended for autologous and intravenous use only (see section 4.4).
The target dose is 410 × 106 CD19 CAR-positive viable T cells (range: 308-513 × 106 CAR-positive viable T cells) supplied in 3 or more infusion bags.
The treatment regimen consists of a split dose to be administered on day 1 and day 10 (± 2 days). The dose regimen will be determined by the tumour burden assessed by bone marrow (BM) blast percentage from a sample obtained within 7 days prior to the start of lymphodepletion (Figure 1).
The RfIC and Dose Schedule Planner (Annex IIIA), located inside the lid of the cryoshipper, must be followed for the actual cell counts and volumes to be infused and to guide the appropriate dose regimen.
Bone marrow assessment
A BM assessment must be available from a biopsy and/or aspirate sample obtained within 7 days prior to the commencement of the lymphodepleting chemotherapy. The BM assessment will be used to determine the Aucatzyl dose regimen: High Tumour Burden Regimen if blast percentage is >20% or Low Tumour Burden Regimen if blast percentage is ≤20% (see Figure 1).
If BM assessment results are inconclusive, the biopsy or aspirate must be repeated (but only once). A repeat biopsy or aspirate should only be taken prior to lymphodepleting chemotherapy.
If results remain inconclusive, the High Tumour Burden Regimen should be administered (i.e., administration of the 10 × 106 dose on day 1 per Figure 1).
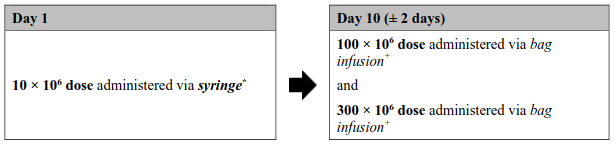
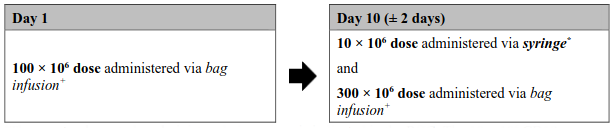
Figure 1. Aucatzyl tumour burden adjusted split dose regimen:
High tumour burden dose regimen
(Bone marrow blast >20% or inconclusive)
Low tumour burden dose regimen
(Bone marrow blast ≤20%)
* The exact volume to be administered via syringe is indicated in the RfIC. The 10 × 106 CD19 CAR-positive viable T cell bag configuration contains an overfill, and therefore it is important to withdraw only the volume specified.
+ The 100 × 106 and 300 × 106 doses will be suspended in one or more infusion bags with no overfill.
Bridging therapy
Bridging therapy can be considered according to the prescriber's choice prior to infusion to reduce tumour burden or stabilise the disease (see section 5.1).
Pretreatment (lymphodepleting chemotherapy)
The lymphodepleting chemotherapy regimen must be administered before infusion of Aucatzyl: fludarabine (FLU) 30 mg/m2/day intravenously and cyclophosphamide (CY) 500 mg/m²/day intravenously on days -6 and -5, followed by fludarabine on days -4 and -3 (total dose: FLU 120 mg/m²; CY 1 000 mg/m²). For dose modifications of cyclophosphamide and fludarabine, see corresponding Summaries of Product Characteristics of cyclophosphamide and fludarabine.
Retreatment with lymphodepleting chemotherapy, in patients who could not receive the Aucatzyl dose on day 1 as planned, could be considered if there is an Aucatzyl dose delay of more than 10 days. Lymphodepleting chemotherapy should not be repeated after the first dose of Aucatzyl is administered.
Aucatzyl is infused 3 days (± 1 day) after completion of lymphodepleting chemotherapy (day 1), allowing a minimum 48-hour washout.
Treatment with Aucatzyl should be delayed in some patient groups at risk (see section 4.4). A delay to the second split dose may be required to manage toxicities.
Premedication
To minimise the risk of an infusion reaction, it is recommended that patients be premedicated with paracetamol (1 000 mg orally) and diphenhydramine 12.5 to 25 mg intravenously or orally (or equivalent medicinal products) approximately 30 minutes prior to Aucatzyl infusion.
Prophylactic use of systemic corticosteroids is not recommended.
Reasons to delay treatment
Delay Aucatzyl treatment if there are unresolved serious adverse reactions from preceding chemotherapies, if the patient is experiencing severe intercurrent infection, or has active graft-versus-host disease. If the patient requires supplementary oxygen, Aucatzyl should only be infused, if considered appropriate, based on the treating physician's benefit/risk assessment.
Reasons to delay the second split dose
Dose delays or discontinuation of treatment may be required after the first split dose to manage adverse reactions as described in Table 1.
Table 1. Dose delay or discontinuation - guidance intended to reduce the risk of adverse reactions:
| Adverse reaction | Gradea | Actions |
|---|---|---|
| Second split dose Day 10 (± 2 days) | ||
| Cytokine release syndrome (CRS) following the first split dose | Grade 2 | Consider postponing Aucatzyl infusion up to day 21 to allow for the CRS to resolve to grade 1 or less. If the CRS persist beyond day 21, do not administer the second dose. |
| Grade ≥3 | Discontinue treatment. | |
| Immune effector cell-associated neurotoxicity syndrome (ICANS) following the first split dose (see section 4.4) | Grade 1 | Consider postponing Aucatzyl infusion up to day 21 to allow for the ICANS to completely resolve. If the ICANS persist beyond day 21, do not administer the second dose. |
| Grade ≥2 | Discontinue treatment. | |
| Pulmonary or cardiac toxicities following the first split doseb,c | Grade ≥3 | Discontinue treatment. |
| Severe intercurrent infection at the time of Aucatzyl infusion (may affect first and second dose) (see section 4.4)b | Grade ≥3 | Consider postponing Aucatzyl infusion up to day 21 until the severe intercurrent infection is considered controlled. If the severe intercurrent infection persists beyond day 21, do not administer the second dose. |
| Requirement for supplementary oxygen (may affect first and second dose)b,c | Grade ≥3 | Consider postponing Aucatzyl treatment up to day 21 only if CRS has resolved to grade 1 or less and ICANS has completely resolved. If the adverse reaction persists beyond day 21, do not administer the second dose. |
| Other clinically relevant adverse reactions following the first split doseb | Grade ≥3 | Consider postponing Aucatzyl infusion up to day 21 only if CRS has resolved to grade 1 or less and ICANS has completely resolved. If the adverse reaction persists beyond day 21, do not administer the second dose. |
a Based on the Common Terminology Criteria for Adverse Events (CTCAE) v5.0. Grade 1 is mild, grade 2 is moderate, grade 3 is severe, and grade 4 is life-threatening. Adapted from National Comprehensive Cancer Network v2.2024 Treatment Guidelines "Management of CAR T-Cell-Related Toxicities" and ASTCT/ASBMT Consensus.
b No dose postponement of second dose for grade 1 or grade 2 events.
c If O2 saturation is less than 92% due to medical conditions.
Monitoring
Patients should be monitored daily for 14 days after the first infusion for signs and symptoms of potential CRS, immune effector cell-associated neurotoxicity syndrome (ICANS) and other toxicities (see section 4.4).
Frequency of monitoring after the first 14 days should be carried out at the physician's discretion and should be continued for at least 4 weeks after.
Patients should be instructed to remain within close proximity of the qualified treatment centre (within 2 hours of travel) for at least 4 weeks following the first infusion.
Special populations
Elderly
No dose adjustment is required in patients over 65 years of age.
Paediatric population
The safety and efficacy of Aucatzyl in children and adolescents below 18 years of age have not yet been established. No data are available.
Method of administration
Aucatzyl is for autologous and intravenous use only.
The Administration instructions are to be strictly followed to minimise dosing errors.
- A leukodepleting filter must not be used. The product must not be irradiated.
- The Aucatzyl Dose Schedule Planner (completed with the patient's BM blast percentage and data from the RfIC) is provided with the RfIC and assists the determination of the appropriate dose regimen to be administered on day 1 and day 10 (± 2 days). The RfIC and Dose Schedule Planner are located inside the lid of the cryoshipper.
- The timing of Aucatzyl thaw, transfer and infusion time must be coordinated.
- It must be confirmed that the patient's identity matches with the patient identifiers on the infusion bag and the RfIC. Do not infuse if the information on the patient-specific label does not match.
- The total number of infusion bags to be administered must also be confirmed with the patient-specific information on the RfIC, see section 6.6.
- The volume to be administered for the 10 × 106 dose is specified on the RfIC. Use the smallest Luer-lock tip syringe necessary depending on the dosing volume specified on the RfIC.
- If more than one bag is needed, subsequent bags must be thawed only after the previous bag is fully administered.
- The entire content of the Aucatzyl infusion bag (100 × 106 and 300 × 106) must be infused at room temperature within 60 minutes post-thaw (infusion rate within 0.1 to 27 mL/minute) using a gravity or peristaltic pump.
Dose administration for 10 × 106 CD19 CAR-positive viable T cells (syringe-based infusion)
The 10 × 106 cell dose should be administered via syringe as this is the only way to deliver the volume specified on the RfIC. Withdrawal of the 10 × 106 cell dose into the syringe should be carried out as follows:
- Prepare and administer Aucatzyl using aseptic technique wearing gloves, protective clothing and eye protection to avoid potential transmission of infectious diseases.
- Gently mix the contents of the bag to disperse clumps of cellular material.
- The volume to be administered for the 10 × 106 dose is specified on the RfIC.
- Use the smallest Luer-lock tip syringe necessary (1, 3, 5, or 10 mL) with a Luer-lock bag spike (or equivalent) to draw up the volume specified on the RfIC.
- Do NOT use a leukodepleting filter.
- Do NOT use the syringe to mix the cells.
- Prime the tubing with sodium chloride 9 mg/mL (0.9%) solution for injection prior to infusion.
- Once Aucatzyl has been drawn into the syringe, verify the volume and administer as an intravenous infusion (as a slow push approximately 0.5 mL/minute) through a central venous line (or large peripheral venous access line appropriate for blood products).
- Complete infusion at room temperature within 60 minutes post-thaw and flush the tubing line with 60 mL of sodium chloride 9 mg/mL (0.9%) solution for injection.
- Dispose of any unused portion of Aucatzyl according to local guidelines.
Dose administration for 100 × 106 and/or 300 × 106 CD19 CAR-positive viable T cells
- Refer to the RfIC for the following details:
- The volume and total CD19 CAR-positive viable T cell number contained in each infusion bag.
- Depending on the infusion bag configuration and disease burden of the patient, the dose may be suspended in one or more infusion bags to be administered on day 1 or day 10. Refer to the RfIC and Dose Schedule Planner for the dose to be administered on the given dosing day and the number of bags required to deliver the specified CD19 CAR-positive viable T cell dose. If more than one bag is needed, thaw subsequent bag only after the previous bag is fully administered.
- Prime the tubing with sodium chloride 9 mg/mL (0.9%) solution for injection prior to infusion.
- Administer Aucatzyl via a gravity or peristaltic pump-assisted intravenous infusion through a central venous line (or large peripheral venous access line appropriate for blood products).
- Do NOT use a leukodepleting filter.
- Aseptic techniques must be applied when performing a venepuncture (if applicable), spiking the ports, and through cell administration process.
- Gently mix the contents of the bag during Aucatzyl infusion to disperse cell clumps.
- Infuse the entire content of the Aucatzyl infusion bag at room temperature within 60 minutes post-thaw using a gravity or peristaltic pump.
- After the entire contents of the infusion bag is infused, rinse the bag with 30 mL of sodium chloride 9 mg/mL (0.9%) solution for injection, then flush the tubing line with 60 mL of sodium chloride 9 mg/mL (0.9%) solution for injection.
- Repeat steps 1-3 for any additional infusion bags required on the given dosing day. Do NOT initiate thaw of the next bag until infusion of the previous bag is complete.
For instructions on planning, preparation, administration, measures to take in case of accidental exposure and disposal of Aucatzyl, see section 6.6.
4.9. Overdose
During clinical studies, occurrences of overdose were observed at the administration of the first dose in 3.9% of patients. All 5 patients had high tumour burden and should have received a 10 × 106 first dose but received a higher dose between 68 and 103 × 106 CAR T cells. CRS, ICANS and HLH, including severe events, were observed in patients who experienced overdose. In the event of a suspected overdose, any adverse reactions are to be treated in accordance with guidance provided (see section 4.4).
6.3. Shelf life
6 months at ≤ –150°C.
Once thawed: 1 hour at room temperature.
6.4. Special precautions for storage
Aucatzyl must be stored in the vapour phase of liquid nitrogen (≤ −150°C) and must remain frozen until the patient is ready for treatment to ensure viable cells are available for patient administration. Thawed medicinal product must not be refrozen. The product must not be irradiated before or during use as this could lead to inactivation of the product.
For storage conditions and duration after thawing of the medicinal product, see section 6.3.
6.5. Nature and contents of container
Ethylene vinyl acetate infusion bag(s) with a sealed filling tube and 2 available spike ports, containing either 10–20 mL (50 mL bags) or 30–70 mL (250 mL bags) cell dispersion. One individual treatment regimen includes 3 or more infusion bags for the total dose of 410 × 106 CD19 CAR-positive viable T cells. Each infusion bag is individually packed within an overwrap in a metal cassette. Metal cassettes are packed into a ModPak Modular Cryogenic Packout Kit. Up to 4 cassettes can fit in a single ModPak. Two ModPak may be required to transport the maximum number of 7 cassettes.
6.6. Special precautions for disposal and other handling
The product must not be irradiated before or during use as this could lead to inactivation of the product.
Receipt and storage of Aucatzyl
- Aucatzyl is supplied in the vapour phase of a liquid nitrogen cryoshipper (≤ -150°C)
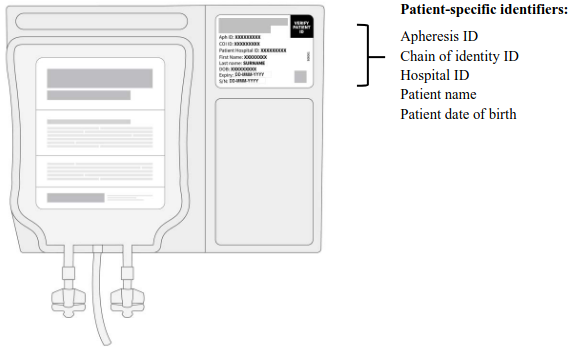
- Confirm the patient's identity. Open the cryoshipper, retrieve the RfIC and metal cassette(s). Open the metal cassette(s) to retrieve the infusion bags in their clear overwrap. The patient's identity must match the patient identifiers on the Aucatzyl RfIC located inside the cryoshipper and on the labels of the infusion bags (see Figure 2).
- Time out of the vapour phase liquid nitrogen environment should be kept to an absolute minimum to avoid premature product thaw (it is recommended not to exceed 90 seconds).
- When the patient's identity does not match the RfIC or label: Do not infuse the product. Contact Autolus at 00800 0825 0829 if there are any discrepancies between the labels and the patient identifiers.
- Keep the infusion bag(s) in the metal cassette(s), transfer Aucatzyl to the on-site controlled-access vapour phase of liquid nitrogen for storage ≤ -150°C (until ready for transfer, thawing and infusion).
Figure 2. Patient-specific identifiers:
Precautions to be taken before handling or administering the medicinal product
Aucatzyl must be transported within the treatment centre in closed, break-proof, leak-proof containers.
This medicinal product contains genetically modified human blood cells. Healthcare professionals handling Aucatzyl must take appropriate precautions (wearing gloves, protective clothing and eye protection) to avoid potential transmission of infectious diseases.
Planning prior to Aucatzyl preparation
The patient batch-specific RfIC and Dose Schedule Planner will be provided in the cryoshipper Confirm the patient identifiers on RfIC and infusion bags match (see Figure 2).
1. Ensure the patient's BM assessment results are available (see section 4.2, Bone marrow assessment).
2. The Aucatzyl Dose Schedule Planner, provided with the RfIC, assists the determination of the appropriate dose regimen to be administered on day 1 (3 days [± 1 day] after the completion of lymphodepleting chemotherapy) and day 10 (± 2 days). Record the following information on the
Dose Schedule Planner:
a. The blast percentage from the patient's BM assessment.
b. The Aucatzyl bag serial number(s); number of bag type required for each dose; and the specified volume to administer via syringe (for the 10 × 106 dose) transcribed from the RfIC.
3. The completion of the Aucatzyl Dose Schedule Planner will guide the treating physician on the number of bags and the respective dose required, and the preparation of Aucatzyl for the day 1 and day 10 (± 2 days) dose, see Figure 1.
Transfer and thawing prior to infusion
- Using the completed Dose Schedule Planner for guidance, transfer ONLY the cassette(s)/infusion bag(s) required for the given dosing day from the on-site vapour phase liquid nitrogen storage to an appropriate transfer vessel (i.e., a vapour phase liquid nitrogen cryoshipper, maintaining temperature ≤ -150°C) for transport to the bag thaw location.
- Transfer the required cassette(s) one by one, confirming the Aucatzyl bag serial numbers and patient identifiers on each infusion bag label (see Figure 2).
- Time out of the vapour phase liquid nitrogen environment should be kept to an absolute minimum to avoid premature product thaw (it is recommended not to exceed 90 seconds).
- If more than one infusion bag has been required on a given dosing day, thaw each infusion bag one at a time; Do not remove subsequent bags from the vapour phase liquid nitrogen storage (≤ -150°C) until infusion of the previous bag is complete.
- Leave the Aucatzyl infusion bag in its overwrap, thaw at 37°C using a water bath or thawing device until there are no visible frozen clumps left in the infusion bag. Each bag should be gently massaged until the cells have just thawed. Thawing of each infusion bag takes between 2 to 8 minutes. Remove from the water bath or thaw device immediately after thawing is complete. Carefully remove the infusion bag from the overwrap taking care to avoid damage to the bag and ports.
- Gently mix the contents of the bag to disperse clumps of cellular material and administer immediately to the patient.
- Do not refreeze or refrigerate thawed product.
Measures to take in case of accidental exposure
In case of accidental exposure, local guidelines on handling of human-derived material must be followed. Work surfaces and materials which have potentially been in contact with Aucatzyl must be decontaminated with appropriate disinfectant.
Precautions to be taken for the disposal of the medicinal product
Unused medicinal product and all material that has been in contact with Aucatzyl (solid and liquid waste) must be handled and disposed of as potentially infectious waste in accordance with local guidelines on handling of human-derived material.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.