BRONCHITOL Inhalation powder, hard capsule Ref.[8203] Active ingredients: Mannitol
Source: European Medicines Agency (EU) Revision Year: 2019 Publisher: Pharmaxis Europe Limited, 108 Q House, Furze Road, Sandyford, Dublin 18, D18AY29, Ireland
Pharmacodynamic properties
Pharmacotherapeutic group: Cough and cold preparations, Mucolytic
ATC code: R05CB16
Mechanism of action
Bronchitol is an inhaled hyperosmotic medicinal product. While the exact mechanism of action is unknown, inhaled mannitol may change the viscoelastic properties of mucus, increase the hydration of the periciliary fluid layer and contribute to increased mucus clearance of the retained secretions through mucociliary activity. Productive cough can contribute to sputum clearance.
Pharmacodynamic effects
In the ITT population of an open label dose response study, DPM-CF-202, the mean (SD) percent change in FEV1 for the 400 mg dose was 8.75 (SD: 12.4) and -1.569 (SD: 9.0) for 40 mg dose (p < 0.0001).
Clinical efficacy and safety
Three Phase 3, 26-week double blind, randomised, parallel arm, controlled, intervention studies (DPM-CF-301,DPM-CF-302 and DPM-CF-303) have been performed in which 324 (DPM-CF-301) and 318 (DPM-CF-302) patients aged 6 years and above were randomised in a 3:2 ratio to inhaled mannitol 400 mg twice daily or to control (inhaled mannitol 50 mg twice daily). In the third study (DPM-CF-303) 423 adult patients were randomised in a 1:1 ratio to inhaled mannitol 400 mg twice daily or to control. Twenty seven (7.1%) out of 378 patients who undertook the mannitol tolerance test (MTT) in study 301, 18 (5.3) out of 341 patients in study 302 and 25 out of 486 patients (5.1%) in study 303 had a positive MTT defined as either 1) a fall in FEV1 >20% from baseline at midpoint (step 4) or 2) fall from baseline > 20 % at end of test that did not recover to < 20% within 15 minutes or 3) who had a fall in FEV1 > 50% from baseline at end of test (step 6) or 4) who had a fall in Sp02 to < 89% during the procedure. An additional 2.8% (n=34) of patients from the three studies had incomplete MTTs and were not randomised.
Mean (SD) baseline FEV1 percent predicted in study DPM-CF-301 (safety population, N= 295) was 62.4 (SD:16.45) and 61.4 (SD:16.13) in the mannitol and control groups, respectively. These figures for study DPM-CF-302 (N=305) are as follows: 65.24 (SD:13.90) and 64.35 (SD:15.29). In study DPM-CF-303 (N=423) the baseline FEV1 percent predicted was 63.17 (SD: 15.15) and 62.98 (SD: 13.65).In study DPM-CF-301 64.4 % of the patient population were adults while in study DPM-CF302 this figure was 49.5%. Study DPM-CF-303 was all adult patients. Fifty five % of patients were receiving rhDNase in study DPM-CF-301 while in study DPM-CF-302 this number was 75% and for DPM-CF-303 this was 67.6%. The percentage of patients receiving inhaled antibiotics was 55% in study DPM-CF-301, 56% in study DPM-CF-302 and 52% in Study DPM-CF-303. Concomitant administration with hypertonic saline was not permitted in these trials.
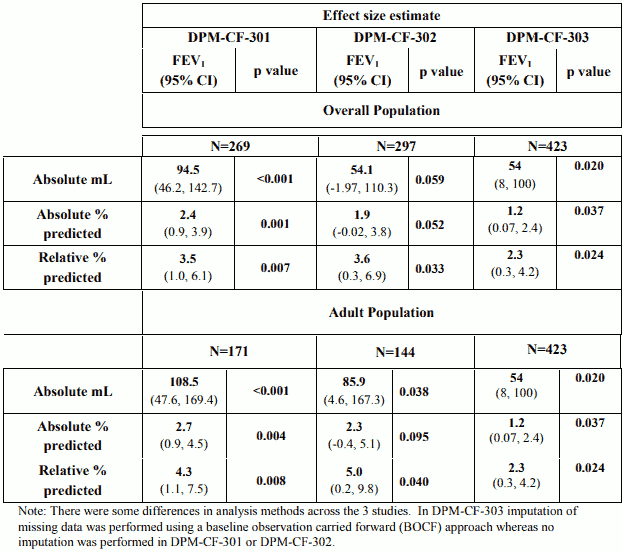
The primary pre-specified endpoint i.e. the change from baseline in FEV1 (ml) in the modified ITT (mITT) population (n=269, 297 and 423 in studies DPM-CF-301,DPM-CF-302 and DPM-CF-303, respectively) compared to control over the 26 weeks period is provided in Table 3 alongside FEV1 presented as absolute and relative change % predicted.
Table 3 – Change in FEV1 from baseline over 26 weeks in the mITT and adult populations
The treatment effect of Bronchitol on FEV1 was less evident in the subgroup of patients who were receiving concomitant rhDNase.In rhDNase users in study 301 the relative change in FEV1 % predicted from baseline across 26 weeks of treatment was 2.83 (95% CI -0.62, 6.27). For non-users the relative change was 4.30 (95% CI 0.53, 8.07). In study 302 the relative change (95% CI) for rhDNase users and non-users was 3.21 (-0.61, 7.03) and 4.73 (-1.93, 11.40), respectively. In study 303 the relative change (95% CI) for rhDNase users and non-users was 1.30 (-0.91, 3.51) and 4.45 (0.52, 8.38), respectively.
Study 303 did not show a superior treatment effect of Bronchitol on FEV1 for female patients, in whom the underlying cystic fibrosis disease course may be worse than males for reasons that are not fully understood. In female patients, the adjusted mean change in FEV1 was 27ml for Bronchitol and 44ml for the control arm, suggesting potentially inferior benefit on lung function with Bronchitol compared to the control, although the difference was not statistically significant (p=0.480).
The number of subjects with at least one protocol defined pulmonary exacerbation (PDPE, defined by the presence of at least 4 symptoms and signs plus the use of intravenous antibiotics) was 18.1% in the mannitol arm and 28% in the control arm in study 301 (ITT population). In study 302 15.2% subjects in the mannitol arm and 19% in the control had a PDPE. In study 303 13.4% subjects in the mannitol arm and 13.6% in the control had a PDPE.
The estimated effect of treatment (mean change and 95% CI from baseline over 26 weeks, mITT population) on FVC was 108.78 ml (95% CI: 49.21, 168.35) in study 301 and 71.4 ml (95% CI: 10.57, 132.13) in study 302 and 40 ml (95% CI: -12, 92) in study 303.
Paediatric population
The safety and efficacy of Bronchitol in children and adolescents aged less than 18 years has not been established (see section 4.2).
In studies DPM-CF-301 and 302 relative % predicted FEV1 compared to control in children (6-11 years) was improved by 0.44% (95% CI -5.90, 6.77, N=43) and 6.1% (95% CI -1.28, 13.54, N=59) over 26 weeks (p=0.892 and 0.104) respectively.
In adolescents (12-17 years) relative change in % predicted FEV1 compared to control improved by 3.31% (95% CI -2.29, 8.90, N=55) and 0.42% (95% CI -5.45, 6.29, N=94) over 26 weeks (p=0.245 and 0.888) respectively.
Pharmacokinetic properties
Absorption
In a study of 18 healthy male adult volunteers, the absolute bioavailability of mannitol powder for inhalation by comparison to mannitol administered intravenously was 0.59% ± 0.15. The rate and extent of absorption of mannitol after inhaled administration was very similar to that observed after oral administration. The Tmax after inhaled administration was 1.5 ± 0.5 hours.
In a study of 9 cystic fibrosis patients (6 adults, 3 adolescents), using 400 mg inhaled mannitol as a single dose (Day 1) then twice a day for 7 days (Days 2-7), pharmacokinetic parameters were similar for adults and adolescents, except for a longer average apparent terminal half life for adolescents (Day 1 = 7.29 hours, Day 7 = 6.52 hours) compared with adults (Day 1 = 6.10 hours, Day 7 = 5.42 hours). Overall, the comparison of AUCs between Day 1 and Day 7 showed a time independence of pharmacokinetics, indicating linearity at the dose level administered in this study.
Biotransformation
A small percentage of systemically absorbed mannitol undergoes hepatic metabolism to glycogen and carbon dioxide. Studies in rats, mice and humans have demonstrated that mannitol has no toxic metabolites. The metabolic pathway of inhaled mannitol was not examined in pharmacokinetic studies.
Distribution
Lung deposition studies have demonstrated a 24.7% deposition of inhaled mannitol confirming its distribution to the target organ. Nonclinical toxicology studies indicate that mannitol inhaled into the lungs is absorbed into the bloodstream, with the maximum serum concentration being achieved occurring at 1 hour. There is no evidence that mannitol is accumulated in the body, therefore distribution of inhaled mannitol was not examined in PK studies.
Elimination
The cumulative amount of mannitol filtered into the urine over the 24 hour collection period was similar for inhaled (55%) and oral (54%) mannitol. When administered intravenously, mannitol is eliminated largely unchanged by glomerular filtration and 87% of the dose is excreted in the urine within 24 hours. The mean terminal half-life in adults was approximately 4 to 5 hours from serum and approximately 3.66 hours from urine.
Paediatric population
The safety and efficacy of Bronchitol in children and adolescents aged 6 to 18 years has not yet been established.
The limited data available in adolescents aged 12 to 17 years indicate the pharmacokinetic parameters of inhaled mannitol are similar to the adult population.
There are no data available for children under 12 years of age.
Preclinical safety data
In male rats after 13 weeks of inhaled mannitol dosing, elevated circulating lymphocyte numbers and mandibular lymph node plasmacytosis was observed at doses greater than 9.3 fold the maximal dose.
The elevated lymphocyte count was within historical control values, did not progress and was essentially resolved by the end of the in life phase of the study and following withdrawal of treatment. This effect was not noted in any other species and did not result in clinical signs.
In dogs an increased occurrence of coughing was observed both during and immediately post dose for low and high dose inhaled mannitol administration. No treatment-related adverse effect occurred greater than 13 fold the maximal therapeutic dose.
No mutagenic or genotoxic effect has been revealed when mannitol was assayed in a standard battery of genotoxicity tests.
Mannitol was shown not to be an irritant in an isolated bovine eye assay or when introduced into rabbit eyes.
No evidence of carcinogenicity was observed when dietary mannitol (≤5%) was administered to mice and rats for 2 years. Carcinogenicity studies have not been carried out with inhaled mannitol.
Reproduction and developmental toxicity studies have not been carried out with inhaled mannitol. However, studies conducted with mannitol administered via other routes indicated no effect on foetal survival in mice, rats and hamsters and on embryo and foetal development in rats and rabbits.
Animal reproduction studies have not been carried out with inhaled mannitol. However, studies conducted with orally administered mannitol indicated no teratogenic effects in mice or rats, at doses of up to 1.6 g/kg, or in hamsters at 1.2 g/kg.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.