DOVATO Film-coated tablet Ref.[10718] Active ingredients: Dolutegravir Lamivudine Lamivudine and Dolutegravir
Source: European Medicines Agency (EU) Revision Year: 2023 Publisher: ViiV Healthcare BV, Van Asch van Wijckstraat 55H, 3811 LP Amersfoort, Netherlands
4.3. Contraindications
Hypersensitivity to the active substances or to any of the excipients listed in section 6.1.
Co-administration with medicinal products with narrow therapeutic windows, that are substrates of organic cation transporter (OCT) 2, including but not limited to fampridine (also known as dalfampridine; see section 4.5).
4.4. Special warnings and precautions for use
Hypersensitivity reactions
Hypersensitivity reactions have been reported with dolutegravir, and were characterized by rash, constitutional findings, and sometimes, organ dysfunction, including severe liver reactions. Dovato and other suspect medicinal products should be discontinued immediately if signs or symptoms of hypersensitivity reactions develop (including, but not limited to, severe rash or rash accompanied by raised liver enzymes, fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, facial oedema, eosinophilia, angioedema). Clinical status including liver aminotransferases and bilirubin should be monitored. Delay in stopping treatment with Dovato or other suspect active substances after the onset of hypersensitivity may result in a life-threatening allergic reaction.
Weight and metabolic parameters
An increase in weight and in levels of blood lipids and glucose may occur during antiretroviral therapy. Such changes may in part be linked to disease control and lifestyle. For lipids and weight, there is in some cases evidence for a treatment effect. For monitoring of blood lipids and glucose reference is made to established HIV treatment guidelines. Lipid disorders should be managed as clinically appropriate.
Liver disease
Patients with chronic hepatitis B or C and treated with combination antiretroviral therapy are at an increased risk of severe and potentially fatal hepatic adverse reactions. In case of concomitant antiviral therapy for hepatitis B or C, please refer also to the relevant product information for these medicinal products.
Dovato includes lamivudine, which is active against hepatitis B. Dolutegravir lacks such activity. Lamivudine monotherapy is generally not considered an adequate treatment for hepatitis B, since the risk for hepatitis B resistance development is high. If Dovato is used in patients co-infected with hepatitis B an additional antiviral is therefore generally needed. Reference should be made to treatment guidelines.
If Dovato is discontinued in patients co-infected with hepatitis B virus, periodic monitoring of both liver function tests and markers of HBV replication is recommended, as withdrawal of lamivudine may result in an acute exacerbation of hepatitis.
Patients with pre-existing liver dysfunction, including chronic active hepatitis have an increased frequency of liver function abnormalities during combination antiretroviral therapy, and should be monitored according to standard practice. If there is evidence of worsening liver disease in such patients, interruption or discontinuation of treatment must be considered.
Immune Reactivation Syndrome
In HIV-infected patients with severe immune deficiency at the time of institution of combination antiretroviral therapy (CART), an inflammatory reaction to asymptomatic or residual opportunistic pathogens may arise and cause serious clinical conditions, or aggravation of symptoms. Typically, such reactions have been observed within the first few weeks or months of initiation of CART. Relevant examples are Cytomegalovirus retinitis, generalised and/or focal mycobacterial infections, and Pneumocystis jirovecii pneumonia (often referred to as PCP). Any inflammatory symptoms should be evaluated and treatment instituted when necessary. Autoimmune disorders (such as Graves' disease and autoimmune hepatitis) have also been reported to occur in the setting of immune reactivation; however, the reported time to onset is more variable and these events can occur many months after initiation of treatment.
Liver chemistry elevations consistent with immune reconstitution syndrome were observed in some hepatitis B and/or C co-infected patients at the start of dolutegravir therapy. Monitoring of liver chemistries is recommended in patients with hepatitis B and/or C co-infection. (See 'Liver disease' earlier in this section and also see section 4.8).
Mitochondrial dysfunction following exposure in utero
Nucleoside and nucleotide analogues may impact mitochondrial function to a variable degree, which is most pronounced with stavudine, didanosine and zidovudine. There have been reports of mitochondrial dysfunction in HIV-negative infants exposed in utero and/or post-natally to nucleoside analogues, these have predominantly concerned treatment with regimens containing zidovudine. The main adverse reactions reported are haematological disorders (anaemia, neutropenia), and metabolic disorders (hyperlactatemia, hyperlipasemia). These reactions have often been transitory. Some late-onset neurological disorders have been reported rarely (hypertonia, convulsion, abnormal behaviour). Whether such neurological disorders are transient or permanent is currently unknown. These findings should be considered for any child exposed in utero to nucleoside and nucleotide analogues, who presents with severe clinical findings of unknown aetiology, particularly neurologic findings. These findings do not affect current national recommendations to use antiretroviral therapy in pregnant women to prevent vertical transmission of HIV.
Osteonecrosis
Although the aetiology is considered to be multifactorial (including corticosteroid use, biphosphonates, alcohol consumption, severe immunosuppression, higher body mass index), cases of osteonecrosis have been reported in patients with advanced HIV-disease and/or long-term exposure to CART. Patients should be advised to seek medical advice if they experience joint aches and pain, joint stiffness or difficulty in movement.
Opportunistic infections
Patients should be advised that dolutegravir, lamivudine or any other antiretroviral therapy does not cure HIV infection and that they may still develop opportunistic infections and other complications of HIV infection. Therefore, patients should remain under close clinical observation by physicians experienced in the treatment of these associated HIV diseases.
Administration in subjects with moderate renal impairment
Patients with a creatinine clearance between 30 and 49 mL/min receiving Dovato may experience a 1.6-to 3.3-fold higher lamivudine exposure (AUC) than patients with a creatinine clearance ≥50 mL/min. There are no safety data from randomized, controlled trials comparing Dovato to the individual components in patients with a creatinine clearance between 30 and 49 mL/min who received dose-adjusted lamivudine. In the original lamivudine registrational trials in combination with zidovudine, higher lamivudine exposures were associated with higher rates of haematologic toxicities (neutropenia and anaemia), although discontinuations due to neutropenia or anaemia each occurred in <1% of subjects. Other lamivudine-related adverse events (such as gastro-intestinal and hepatic disorders) may occur.
Patients with a sustained creatinine clearance between 30 and 49 mL/min who receive Dovato should be monitored for lamivudine-related adverse events, notably haematologic toxicities. If new or worsening neutropenia or anaemia develop, a dose adjustment of lamivudine, per lamivudine prescribing information, is indicated, which cannot be achieved with Dovato. Dovato should be discontinued and the individual components should be used to construct the treatment regimen.
Drug interactions
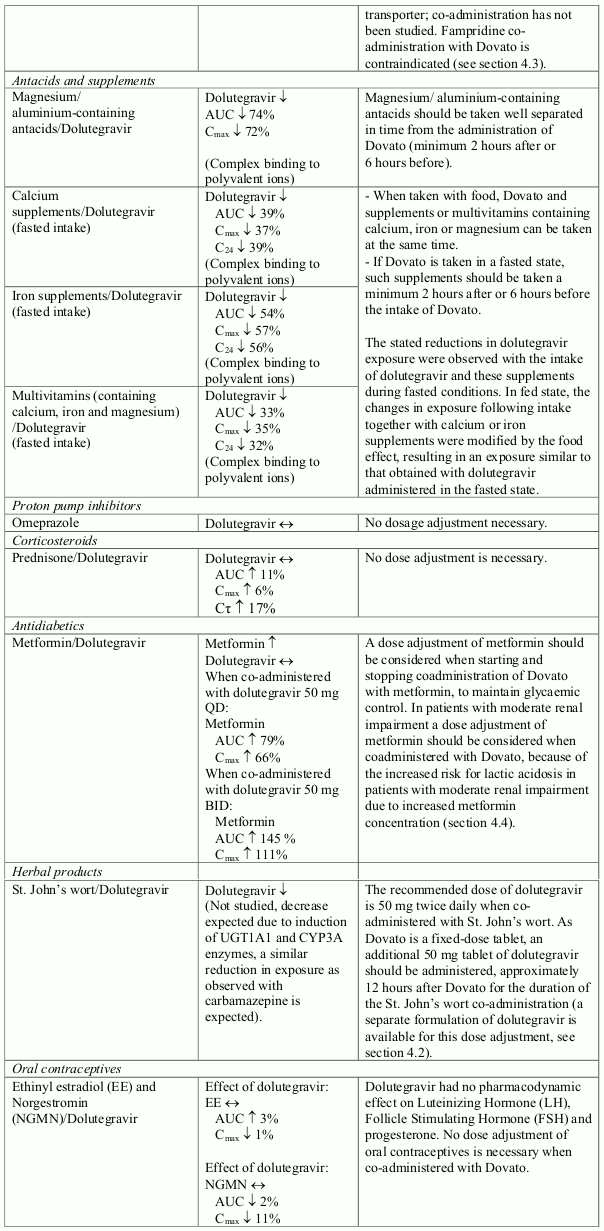
The recommended dose of dolutegravir is 50 mg twice daily when co-administered with rifampicin, carbamazepine, oxcarbazepine, phenytoin, phenobarbital, St. John's wort, etravirine (without boosted protease inhibitors), efavirenz, nevirapine, or tipranavir/ritonavir (see section 4.5).
Dovato should not be co-administered with polyvalent cation-containing antacids. Polyvalent cationcontaining antacids are recommended to be taken 2 hours after or 6 hours before Dovato (see section 4.5).
When taken with food, Dovato and supplements or multivitamins containing calcium, iron or magnesium can be taken at the same time. If Dovato is administered under fasting conditions, supplements or multivitamins containing calcium, iron or magnesium are recommended to be taken 2 hours after or 6 hours before Dovato (see section 4.5).
Dolutegravir increased metformin concentrations. A dose adjustment of metformin should be considered when starting and stopping coadministration of Dovato with metformin, to maintain glycaemic control (see section 4.5). Metformin is eliminated renally and, therefore, it is of importance to monitor renal function when co-treated with Dovato. This combination may increase the risk for lactic acidosis in patients with moderate renal impairment (stage 3a creatinine clearance 45–59 mL/min) and a cautious approach is recommended. Reduction of the metformin dose should be highly considered.
The combination of Dovato with cladribine is not recommended (see section 4.5).
Dovato should not be taken with any other medicinal product containing dolutegravir, lamivudine or emtricitabine, except where a dose adjustment of dolutegravir is indicated due to drug-drug interactions (see section 4.5).
4.5. Interaction with other medicinal products and other forms of interaction
No drug interaction studies have been conducted using Dovato. Dovato contains dolutegravir and lamivudine, therefore any interactions identified for these individually are relevant to Dovato. No clinically significant drug interactions are expected between dolutegravir and lamivudine.
Effect of other medicinal products on the pharmacokinetics of dolutegravir and lamivudine
Dolutegravir is eliminated mainly through metabolism by uridine diphosphate glucuronosyl transferase (UGT) 1A1. Dolutegravir is also a substrate of UGT1A3, UGT1A9, CYP3A4, P-glycoprotein (P-gp), and breast cancer resistance protein (BCRP). Co-administration of Dovato and other medicinal products that inhibit UGT1A1, UGT1A3, UGT1A9, CYP3A4, and/or P-gp may, therefore, increase dolutegravir plasma concentration. Medicinal products that induce those enzymes or transporters may decrease dolutegravir plasma concentration and reduce the therapeutic effect of dolutegravir.
The absorption of dolutegravir is reduced by certain metal cation-containing anti-acid substances and supplements (see Table 1).
Lamivudine is cleared renally. Active renal secretion of lamivudine in the urine is mediated through the OCT2 and multidrug and toxin extrusion transporters (MATE1 and MATE2-K). Trimethoprim (an inhibitor of these transporters) has been shown to increase lamivudine plasma concentrations, however the resulting increase was not clinically significant (see Table 1). Dolutegravir is an OCT2 and MATE1 inhibitor; however, lamivudine concentrations were similar with or without co-administration of dolutegravir based on a cross study analysis, indicating that dolutegravir has no relevant effect on lamivudine exposure in vivo. Lamivudine is also substrate of the hepatic uptake transporter OCT1. As hepatic elimination plays a minor role in the clearance of lamivudine, drug interactions due to inhibition of OCT1 are unlikely to be of clinical significance.
Although lamivudine is a substrate of BCRP and P-gp in vitro, given its high absolute bioavailability, (see section 5.2), inhibitors of these efflux transporters are unlikely to result in a clinically relevant impact on lamivudine concentrations. Effect of dolutegravir and lamivudine on the pharmacokinetics of other medicinal products
In vivo, dolutegravir did not have an effect on midazolam, a CYP3A4 probe. Based on in vivo and/or in vitro data, dolutegravir is not expected to affect the pharmacokinetics of medicinal products that are substrates of any major enzyme or transporter such as CYP3A4, CYP2C9 and P-gp (for more information see section 5.2).
In vitro, dolutegravir inhibited the renal transporters OCT2 and MATE1. In vivo, a 10-14% decrease of creatinine clearance (secretory fraction is dependent on OCT2 and MATE1 transport) was observed in patients. In vivo, dolutegravir may increase plasma concentrations of medicinal products in which excretion is dependent upon OCT2 and/or MATE1 (e.g. fampridine [also known as dalfampridine], metformin) (see Table 1 and section 4.3).
In vitro, dolutegravir inhibited the renal uptake organic anion transporters (OAT)1 and OAT3. Based on the lack of effect on the in vivo pharmacokinetics of the OAT substrate tenofovir, in vivo inhibition of OAT1 is unlikely. Inhibition of OAT3 has not been studied in vivo. Dolutegravir may increase plasma concentrations of medicinal products in which excretion is dependent upon OAT3.
In vitro, lamivudine was an inhibitor of OCT1 and OCT2; the clinical consequences are not known.
Established and theoretical interactions with selected antiretrovirals and non-antiretroviral medicinal products are listed in Table 1.
Interaction table
Interactions between dolutegravir, lamivudine and co-administered medical products are listed in Table 1 (increase is indicated as "↑", decrease as "↓", no change as "↔", area under the concentration versus time curve as "AUC", maximum observed concentration as "Cmax", concentration at end of dosing interval as "Cτ"). The table should not be considered exhaustive but is representative of the classes studied.
Paediatric population
Interaction studies have only been performed in adults.
4.6. Fertility, pregnancy and lactation
Women of childbearing potential
Women of childbearing potential (WOCBP) should be counselled about the potential risk of neural tube defects with dolutegravir (a component of Dovato, see below), including consideration of effective contraceptive measures.
If a woman plans pregnancy, the benefits and the risks of continuing treatment with Dovato should be discussed with the patient.
Pregnancy
The safety and efficacy of a dual regimen has not been studied in pregnancy.
Human experience from a birth outcome surveillance study in Botswana shows a small increase of neural tube defects; 7 cases in 3,591 deliveries (0.19%; 95% CI 0.09%, 0.40%) to mothers taking dolutegravircontaining regimens at the time of conception compared to 21 cases in 19,361 deliveries (0.11%: 95% CI 0.07%, 0.17%) to women exposed to non-dolutegravir regimens at the time of conception.
The incidence of neural tube defects in the general population ranges from 0.5-1 case per 1,000 live births (0.05-0.1%). Most neural tube defects occur within the first 4 weeks of embryonic development after conception (approximately 6 weeks after the last menstrual period). If a pregnancy is confirmed in the first trimester while on Dovato, the benefits and risks of continuing Dovato versus switching to another antiretroviral regimen should be discussed with the patient taking the gestational age and the critical time period of neural tube defect development into account.
Data analysed from the Antiretroviral Pregnancy Registry do not indicate an increased risk of major birth defects in over 600 women exposed to dolutegravir during pregnancy but are currently insufficient to address the risk of neural tube defects.
In animal reproductive toxicology studies with dolutegravir, no adverse development outcomes, including neural tube defects, were identified (see section 5.3).
More than 1000 outcomes from exposure to dolutegravir during second and third trimester pregnancy indicate no evidence of increased risk of foetal/neonatal toxicity. Dovato may be used during the second and third trimester of pregnancy when the expected benefit justifies the potential risk to the foetus.
There is insufficient information on the effects of dolutegravir on neonates.
A large amount of data on the use of lamivudine in pregnant women (more than 5200 outcomes from first trimester) indicates no malformative toxicity.
Animal studies showed lamivudine may inhibit cellular DNA replication (see section 5.3). The clinical relevance of these findings is unknown.
Mitochondrial dysfunction
Nucleoside and nucleotide analogues have been demonstrated in vitro and in vivo to cause a variable degree of mitochondrial damage. There have been reports of mitochondrial dysfunction in HIV-negative infants exposed in utero and/or post-natally to nucleoside analogues (see section 4.4).
Breast-feeding
Dolutegravir is excreted in human milk in small amounts (a median dolutegravir breast milk to maternal plasma ratio of 0.033 has been shown). There is insufficient information on the effects of dolutegravir in neonates/infants.
Based on more than 200 mother/child pairs treated for HIV, serum concentrations of lamivudine in breastfed infants of mothers treated for HIV are very low (<4% of maternal serum concentrations) and progressively decrease to undetectable levels when breastfed infants reach 24 weeks of age. There are no data available on the safety of lamivudine when administered to babies less than three months old.
It is recommended that women living with HIV do not breast-feed their infants in order to avoid transmission of HIV.
Fertility
There are no data on the effects of dolutegravir or lamivudine on human male or female fertility. Animal studies indicate no effects of dolutegravir or lamivudine on male or female fertility (see section 5.3).
4.7. Effects on ability to drive and use machines
Dovato has no or negligible influence on the ability to drive and use machines. Patients should be informed that dizziness and somnolence has been reported during treatment with dolutegravir. The clinical status of the patient and the adverse reaction profile of Dovato should be borne in mind when considering the patient's ability to drive or operate machinery.
4.8. Undesirable effects
Summary of the safety profile
The most frequently reported adverse reactions are headache (3%), diarrhoea (2%), nausea (2%) and insomnia (2%).
The most severe adverse reaction reported with dolutegravir was a hypersensitivity reaction that included rash and severe liver effects (see section 4.4).
Tabulated list of adverse reactions
The adverse reactions from clinical study and post-marketing experience are listed in Table 2 by body system, organ class and absolute frequency. Frequencies are defined as very common (≥1/10), common (≥1/100 to <1/10), uncommon (≥1/1,000 to <1/100), rare (≥1/10,000 to <1/1,000), very rare (<1/10,000), not known (cannot be estimated from the available data).
Table 2. Tabulated summary of adverse reactions to Dovato based on clinical study and postmarketing experience with Dovato and its individual components:
| Frequency | Adverse reaction |
|---|---|
| Blood and lymphatic systems disorders | |
| Uncommon: | neutropenia, anaemia, thrombocytopenia |
| Very rare: | pure red cell aplasia |
| Immune system disorders | |
| Uncommon: | hypersensitivity (see section 4.4), immune reconstitution syndrome (see section 4.4) |
| Metabolism and nutrition disorders | |
| Very rare: | lactic acidosis |
| Psychiatric disorders | |
| Common: | depression, anxiety, insomnia, abnormal dreams |
| Uncommon: | suicidal ideation*, suicide attempt*, panic attack *particularly in patients with a pre-existing history of depression or psychiatric illness. |
| Rare: | completed suicide* *particularly in patients with a pre-existing history of depression or psychiatric illness. |
| Nervous system disorders | |
| Very common: | headache |
| Common: | dizziness, somnolence |
| Very rare: | peripheral neuropathy, paraesthesia |
| Gastrointestinal disorders | |
| Very common: | nausea, diarrhoea |
| Common: | vomiting, flatulence, abdominal pain/ discomfort |
| Rare: | pancreatitis |
| Hepatobiliary disorders | |
| Common: | alanine aminotransferase (ALT) and/or aspartate aminotransferase (AST) elevations |
| Uncommon: | hepatitis |
| Rare: | acute hepatic failure1, increased bilirubin2 |
| Skin and subcutaneous tissue disorders | |
| Common: | rash, pruritus, alopecia |
| Rare: | angioedema |
| Musculoskeletal and connective tissue disorders | |
| Common: | arthralgia, muscle disorders (including myalgia) |
| Rare: | rhabdomyolysis |
| General disorders and administration site conditions | |
| Common: | fatigue |
| Investigations | |
| Common: | creatine phosphokinase (CPK) elevations, weight increased |
| Rare: | amylase elevations |
1 This adverse reaction was identified through post-marketing surveillance for dolutegravir in combination with other ARVs. The frequency category of rare was estimated based on post-marketing reports.
2 In combination with increased transaminases.
Description of selected adverse reactions
Changes in laboratory biochemistries
Dolutegravir has been associated with an increase in serum creatinine occuring in the first week of treatment when administered with other antiretroviral medicinal products. Increases in serum creatinine occurred within the first four weeks of treatment with dolutegravir plus lamivudine and remained stable through 48 weeks. In the pooled GEMINI studies a mean change from baseline of 10.3 µmol/L (range: -36.3 µmol/L to 55.7 µmol/L) was observed after 48 weeks of treatment. These changes are linked to the inhibiting effect of dolutegravir on renal tubular transporters of creatinine. The changes are not considered to be clinically relevant and do not reflect a change in glomerular filtration rate.
Co-infection with Hepatitis B or C
In the Phase III studies for the dolutegravir single agent, patients with hepatitis B and/or C co-infection were permitted to enrol provided that baseline liver chemistry tests did not exceed 5 times the upper limit of normal (ULN). Overall, the safety profile in patients co-infected with hepatitis B and/or C was similar to that observed in patients without hepatitis B or C co-infection, although the rates of AST and ALT abnormalities were higher in the subgroup with hepatitis B and/or C co-infection for all treatment groups. Liver chemistry elevations consistent with immune reconstitution syndrome were observed in some subjects with hepatitis B and/or C co-infection at the start of dolutegravir therapy, particularly in those whose anti-hepatitis B therapy was withdrawn (see section 4.4).
Metabolic parameters
Weight and levels of blood lipids and glucose may increase during antiretroviral therapy (see section 4.4).
Osteonecrosis
Cases of osteonecrosis have been reported, particularly in patients with generally acknowledged risk factors, advanced HIV disease or long-term exposure to CART. The frequency of this is unknown (see section 4.4).
Immune response syndrome
In HIV-infected patients with severe immune deficiency at the time of initiation of combination antiretroviral therapy (CART), an inflammatory reaction to asymptomatic or residual opportunistic infections may arise. Autoimmune disorders (such as Graves' disease and autoimmune hepatitis) have also been reported; however, the reported time to onset is more variable and these events can occur many months after initiation of treatment (see section 4.4).
Paediatric population
There are no clinical study data on the effects of Dovato in the paediatric population. Individual components have been investigated in adolescents (12 to 17 years).
Based on limited available data with the dolutegravir single entity or lamivudine single entity used in combination with other antiretroviral agents to treat adolescents (12 to 17 years), there were no additional types of adverse reactions beyond those observed in the adult population.
Reporting of suspected adverse reactions
Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the national reporting system listed in Appendix V.
6.2. Incompatibilities
Not applicable.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.