DOXY Powder for solution for injection Ref.[28060] Active ingredients: Doxycycline
Source: FDA, National Drug Code (US) Revision Year: 2021
Product description
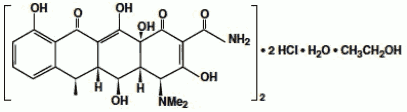
Doxycycline for Injection, USP is a sterile, lyophilized powder prepared from a solution of doxycycline hyclate, ascorbic acid and mannitol in Water for Injection. Doxycycline hyclate is a broad spectrum antibiotic derived from oxytetracycline. It is meant for INTRAVENOUS use only after reconstitution. Doxycycline hyclate is a yellowish crystalline powder which is chemically designated 4-(Dimethylamino)-1,4,4a,5,5a,6,11, 12a-octahydro-3,5,10,12,12a-pentahydroxy-6-methyl-1,11-de monohydrochloride, compound with ethyl alcohol (2:1), monohydrate.
It has the following structural formula:
(C22H24N2O8 • HCl)2 • C2H6O • H2O
M.W. 1025.89
Doxycycline hyclate is soluble in water and chars at 201°C without melting. The base doxycycline has a high degree of lipid solubility and a low affinity for calcium binding. It is highly stable in normal human serum.
Each 100 mg vial contains: Doxycycline hyclate equivalent to 100 mg doxycycline; ascorbic acid 480 mg; mannitol 300 mg. pH of the reconstituted solution (10 mg/mL) is between 1.8 and 3.3.
| How Supplied | ||||||||
|---|---|---|---|---|---|---|---|---|
|
Doxycycline for Injection, USP, sterile powder, supplied as follows:
The brand names mentioned in this document are the trademarks of their respective owners. Novaplus is a registered trademark of Vizient. Inc. Manufactured by: Fresenius Kabi, Lake Zurich, IL 60047 |
Drugs
| Drug | Countries | |
|---|---|---|
| DOXY | France, Hong Kong, Israel, Nigeria, New Zealand, United States |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.