DYCLOPRO Topical solution Ref.[10046] Active ingredients: Dyclonine
Source: FDA, National Drug Code (US) Revision Year: 2020
Product description
DycloPro (Dyclonine Hydrochloride Topical Solution, USP, 0.5% and 1%) topical anesthetics contain a local anesthetic agent and are administered topically. See INDICATIONS for specific uses.
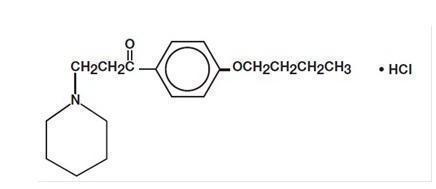
DycloPro (Dyclonine Hydrochloride Topical Solution, USP, 0.5% and 1%) contain dyclonine HCl, which is chemically designated as 4'-butoxy-3-piperidinopropiophenone HCl. Dyclonine HCl is a white crystalline powder that is sparingly soluble in water and has the following structural formula:
Composition of Dyclonine HCl Topical Solution, USP, 0.5% and 1% topical anesthetics:
Each mL of Dyclonine HCl Topical Solution, USP, 0.5% contains dyclonine HCl, 5 mg.
Each mL of Dyclonine HCl Topical Solution, USP, 1% contains dyclonine HCl, 10 mg.
Both solutions also contain chlorbutanol hydrous and sodium chloride, and the pH is adjusted 3.0-5.0 by means of hydrochloric acid.
| How Supplied |
|---|
|
Clear and colorless sterile solution, in one fluid ounce bottles. Multi dose Dyclonine HCl Topical Solution 0.5% (NDC 0362-3971-05) Dyclonine HCl Topical Solution 1% (NDC 0362-3918-10) Manufactured for SEPTODONT, Inc., 205 Granite Run Dr., Suite 150, Lancaster, PA, USA 17601 by Novocol Pharmaceuitcal of Canada, Inc., Ontario, Canada N1R 6X3 |
Drugs
| Drug | Countries | |
|---|---|---|
| DYCLOPRO | United States |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.