ENFLONSIA Solution for injection Ref.[116536] Active ingredients: Clesrovimab
Source: European Medicines Agency (EU) Revision Year: 2026 Publisher: Merck Sharp & Dohme B.V., Waarderweg 39, 2031 BN Haarlem, The Netherlands
4.1. Therapeutic indications
Enflonsia is indicated for the prevention of respiratory syncytial virus (RSV) lower respiratory tract disease in neonates and infants during their first RSV season.
Enflonsia should be used in accordance with official recommendations.
4.2. Posology and method of administration
Posology
Neonates and infants: first RSV season
The recommended dose is 105 mg administered as a single 0.7 mL intramuscular (IM) injection.
For neonates and infants born during the RSV season, Enflonsia should be administered starting from birth. For infants born outside the RSV season, it should be administered once prior to the start of their first RSV season (see section 5.1).
Dosing in infants with a body weight between 0.5 kg and 1.1 kg is based on extrapolation; no clinical data are available. Exposure in infants <1.1 kg is anticipated to yield higher exposures than in those weighing more. The benefits and risks of clesrovimab in infants <1.1 kg should be carefully considered.
There are limited clinical data available in extremely preterm infants (gestational age (GA) <29 weeks) who are of chronological age less than 8 weeks. No clinical data are available in infants with a postmenstrual age (GA plus chronological age) of less than 32 weeks (see section 5.1).
Infants undergoing cardiac surgery with cardiopulmonary bypass
For infants undergoing cardiac surgery with cardiopulmonary bypass during the RSV season, an additional 105 mg dose is recommended as soon as the infant is stable after surgery to ensure adequate clesrovimab serum levels.
Children from 1 to 18 years of age
The safety and efficacy of clesrovimab in children aged 1 to 18 years have not yet been established. No data are available.
Method of administration
Enflonsia is for intramuscular use only.
The medicinal product should be administered intramuscularly by a healthcare professional, in the anterolateral aspect of the thigh. It should not be injected in the gluteal area or areas where there may be a major nerve trunk and/or blood vessel.
For instructions on handling of the medicinal product before administration, see section 6.6.
4.9. Overdose
There is no specific treatment for an overdose with clesrovimab. In the event of an overdose, the individual should be monitored for the occurrence of adverse reactions and provided with symptomatic treatment as appropriate.
6.3. Shelf life
30 months.
Enflonsia may be kept at room temperature (20°C-25°C) for a maximum 48 hours. After removal from the refrigerator, it must be used within 48 hours or discarded.
6.4. Special precautions for storage
Store in a refrigerator (2°C–8°C). Do not freeze.
Keep the pre-filled syringe in the outer carton in order to protect from light.
Do not shake.
6.5. Nature and contents of container
0.7 mL solution in pre-filled syringe (Type I glass) with a plunger stopper and a tip cap with or without needles.
Enflonsia is available in the following pack sizes:
- 1 pre-filled syringe
- 1 pre-filled syringe + 1 needle
- 1 pre-filled syringe + 2 needles
- 10 pre-filled syringes
- 10 pre-filled syringes + 10 needles
- 10 pre-filled syringes + 20 needles
- Multipacks containing 50 (5 packs of 10) pre-filled syringes
Not all pack sizes may be marketed.
6.6. Special precautions for disposal and other handling
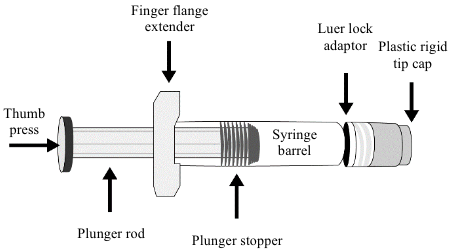
Before injection, remove the carton from the refrigerator and allow the pre-filled syringe to come to room temperature for approximately 15 minutes. Parenteral medicinal products should be inspected visually for particulate matter and discolouration prior to administration. It should not be used if particulate matter or discolouration is found. Do not use Enflonsia if the pre-filled syringe has been dropped or damaged, the security seal on the carton has been broken, or the expiry date has passed. Refer to Figure 1 for pre-filled syringe components.
Figure 1. Pre-filled syringe components:
Step 1: Hold the syringe barrel in one hand and unscrew the tip cap by twisting it counter-clockwise with the other hand. Do not remove the Luer lock adaptor and the finger flange extender.
Step 2: Attach a sterile Luer lock needle by twisting in a clockwise direction until the needle fits securely on the syringe. If not provided, due to the viscosity of the product, use a 25 gauge or larger needle.
Step 3: Inject the entire contents of the pre-filled syringe intramuscularly, in the anterolateral aspect of the thigh. The medicinal product should not be injected in the gluteal area or areas where there may be a major nerve trunk and/or blood vessel.
Enflonsia is for single use only. Any unused medicinal product or waste material should be disposed of in accordance with local requirements.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.