EVRA Plaster Ref.[10483] Active ingredients: 17 alpha-Ethinylestradiol Norelgestromin
Source: European Medicines Agency (EU) Revision Year: 2020 Publisher: JANSSEN-CILAG INTERNATIONAL NV, Turnhoutseweg, 30, B-2340 Beerse, Belgium
4.3. Contraindications
Combined hormonal contraceptives (CHCs) should not be used in the following conditions. If one of these disorders occurs during the use of EVRA, EVRA must be discontinued immediately.
- Presence or risk of venous thromboembolism (VTE)
- Venous thromboembolism – current VTE (on anticoagulants) or history of (e.g. deep venous thrombosis [DVT] or pulmonary embolism [PE]);
- Known hereditary or acquired predisposition for venous thromboembolism, such as APC-resistance, (including Factor V Leiden), antithrombin-III-deficiency, protein C deficiency, protein S deficiency;
- Major surgery with prolonged immobilisation (see section 4.4);
- A high risk of venous thromboembolism due to the presence of multiple risk factors (see section 4.4);
- Presence or risk of arterial thromboembolism (ATE)
- Arterial thromboembolism – current arterial thromboembolism, history of arterial thromboembolism (e.g. myocardial infarction) or prodromal condition (e.g. angina pectoris);
- Cerebrovascular disease – current stroke, history of stroke or prodromal condition (e.g. transient ischaemic attack, TIA);
- Known hereditary or acquired predisposition for arterial thromboembolism, such as hyperhomocysteinaemia and antiphospholipid-antibodies (anticardiolipin-antibodies, lupus anticoagulant);
- History of migraine with focal neurological symptoms;
- A high risk of arterial thromboembolism due to multiple risk factors (see section 4.4) or to the presence of one serious risk factor such as:
- diabetes mellitus with vascular symptoms
- severe hypertension
- severe dyslipoproteinaemia
- Hypersensitivity to the active substances or to any of the excipients listed in section 6.1
- Known or suspected carcinoma of the breast
- Carcinoma of the endometrium or other known or suspected oestrogen-dependent neoplasia
- Abnormal liver function related to acute or chronic hepatocellular disease
- Hepatic adenomas or carcinomas
- Undiagnosed abnormal genital bleeding
- Concomitant use with the medicinal products containing ombitasvir/paritaprevir/ritonavir and dasabuvir (see sections 4.4 and 4.5).
4.4. Special warnings and precautions for use
Warnings
If any of the conditions/risk factors mentioned below is present, the suitability of EVRA should be discussed with the woman.
In the event of aggravation, or first appearance of any of the conditions or risk factors, the woman should be advised to contact her doctor to determine whether the use of EVRA should be discontinued.
There is no clinical evidence indicating that a transdermal patch is, in any aspect, safer than combined oral contraceptives.
EVRA is not indicated during pregnancy (see section 4.6).
Risk of venous thromboembolism (VTE)
The use of any combined hormonal contraceptive (CHC) increases the risk of venous thromboembolism (VTE) compared with no use. Products that contain levonorgestrel, norgestimate or norethisterone are associated with the lowest risk of VTE. Other products such as EVRA may have up to twice this level of risk. The decision to use any product other than one with the lowest VTE risk should be taken only after a discussion with the woman to ensure she understands the risk of VTE with EVRA, how her current risk factors influence this risk, and that her VTE risk is highest in the first ever year of use. There is also some evidence that the risk is increased when a CHC is re-started after a break in use of 4 weeks or more.
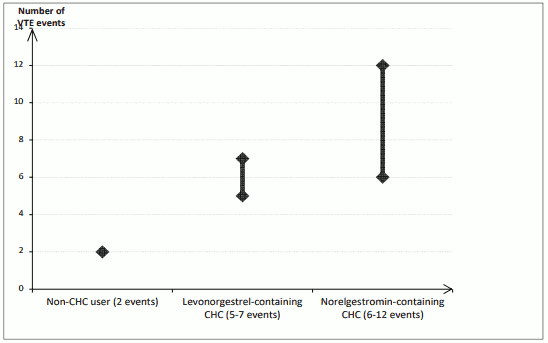
In women who do not use a CHC and are not pregnant about 2 out of 10,000 will develop a VTE over the period of one year. However, in any individual woman the risk may be far higher, depending on her underlying risk factors (see below).
It is estimated that out of 10,000 women who use a low dose CHC that contains levonorgestrel, about 61 will develop a VTE in one year. Studies have suggested that the incidence of VTE in women who used EVRA is up to 2-fold higher than in users of CHCs that contain levonorgestrel. This corresponds to between about 6 and 12 VTEs in a year out of 10,000 women who use EVRA.
1 Mid-point of range of 5-7 per 10,000 WY, based on a relative risk for CHCs containing levonorgestrel versus non-use of approximately 2.3 to 3.6
In both cases, the number of VTEs per year is fewer than the number expected in women during pregnancy or in the postpartum period.
VTE may be fatal in 1-2% of cases.
Number of VTE events per 10,000 women in one year:
Extremely rarely, thrombosis has been reported to occur in CHC users in other blood vessels, e.g. hepatic, mesenteric, renal or retinal veins and arteries.
Risk factors for VTE
The risk for venous thromboembolic complications in CHC users may increase substantially in a woman with additional risk factors, particularly if there are multiple risk factors (see table).
EVRA is contraindicated if a woman has multiple risk factors that put her at high risk of venous thrombosis (see section 4.3). If a woman has more than one risk factor, it is possible that the increase in risk is greater than the sum of the individual factors – in this case her total risk of VTE should be considered. If the balance of benefits and risks is considered to be negative a CHC should not be prescribed (see section 4.3).
Table. Risk factors for VTE:
| Risk factor | Comment |
|---|---|
| Obesity (body mass index over 30 kg/m²) | Risk increases substantially as BMI rises. Particularly important to consider if other risk factors also present. |
| Prolonged immobilisation, major surgery, any surgery to the legs or pelvis, neurosurgery, or major trauma Note: temporary immobilisation including air travel >4 hours can also be a risk factor for VTE, particularly in women with other risk factors | In these situations it is advisable to discontinue the use of the patch (in the case of elective surgery at least four weeks in advance) and not resume until two weeks after complete remobilisation. Another method of contraception should be used to avoid unintentional pregnancy. Antithrombotic treatment should be considered if EVRA has not been discontinued in advance. |
| Positive family history (venous thromboembolism ever in a sibling or parent at relatively early age) | If a hereditary predisposition is suspected, the woman should be referred to a specialist for advice before deciding about any CHC use. |
| Other medical conditions associated with VTE | Cancer, systemic lupus erythematosus, haemolytic uraemic syndrome, chronic inflammatory bowel disease (Crohn's disease or ulcerative colitis) and sickle cell disease. |
| Increasing age | Particularly above 35 years. |
There is no consensus about the possible role of varicose veins and superficial thrombophlebitis in the onset or progression of venous thrombosis.
The increased risk of thromboembolism in pregnancy, and particularly the 6 week period of the puerperium, must be considered (for information on "Pregnancy and lactation" see section 4.6).
Symptoms of VTE (deep vein thrombosis and pulmonary embolism)
In the event of symptoms women should be advised to seek urgent medical attention and to inform the healthcare professional that she is taking a CHC.
Symptoms of deep vein thrombosis (DVT) can include:
- unilateral swelling of the leg and/or foot or along a vein in the leg;
- pain or tenderness in the leg which may be felt only when standing or walking;
- increased warmth in the affected leg; red or discoloured skin on the leg.
Symptoms of pulmonary embolism (PE) can include:
- sudden onset of unexplained shortness of breath or rapid breathing;
- sudden coughing which may associated with haemoptysis;
- sharp chest pain;
- severe light headedness or dizziness;
- rapid or irregular heartbeat.
Some of these symptoms (e.g. "shortness of breath", "coughing") are non-specific and might be misinterpreted as more common or less severe events (e.g. respiratory tract infections).
Other signs of vascular occlusion can include: sudden pain, swelling and slight blue discoloration of an extremity.
If the occlusion occurs in the eye symptoms can range from painless blurring of vision which can progress to loss of vision. Sometimes loss of vision can occur almost immediately.
Risk of arterial thromboembolism (ATE)
Epidemiological studies have associated the use of CHCs with an increased risk for arterial thromboembolism (myocardial infarction) or for cerebrovascular accident (e.g. transient ischaemic attack, stroke). Arterial thromboembolic events may be fatal.
Risk factors for ATE
The risk of arterial thromboembolic complications or of a cerebrovascular accident in CHC users increases in women with risk factors (see table). EVRA is contraindicated if a woman has one serious or multiple risk factors for ATE that puts her at high risk of arterial thrombosis (see section 4.3). If a woman has more than one risk factor, it is possible that the increase in risk is greater than the sum of the individual factors - in this case her total risk should be considered. If the balance of benefits and risks is considered to be negative a CHC should not be prescribed (see section 4.3).
Table. Risk factors for ATE:
| Risk factor | Comment |
|---|---|
| Increasing age | Particularly above 35 years |
| Smoking | Women should be advised not to smoke if they wish to use a CHC. Women over 35 who continue to smoke should be strongly advised to use a different method of contraception. |
| Hypertension | |
| Obesity (body mass index over 30 kg/m²) | Risk increases substantially as BMI rises. Particularly important in women with additional risk factors. |
| Positive family history (arterial thromboembolism ever in a sibling or parent at relatively early age e.g. below 50) | If a hereditary predisposition is suspected, the woman should be referred to a specialist for advice before deciding about any CHC use. |
| Migraine | An increase in frequency or severity of migraine during CHC use (which may be prodromal of a cerebrovascular event) may be a reason for immediate discontinuation. |
| Other medical conditions associated with adverse vascular events | Diabetes mellitus, hyperhomocysteinaemia, valvular heart disease and atrial fibrillation, dyslipoproteinaemia, systemic lupus erythematosus. |
Symptoms of ATE
In the event of symptoms women should be advised to seek urgent medical attention and to inform the healthcare professional that she is taking a CHC.
Symptoms of a cerebrovascular accident can include:
- sudden numbness or weakness of the face, arm or leg, especially on one side of the body;
- sudden trouble walking, dizziness, loss of balance or coordination;
- sudden confusion, trouble speaking or understanding;
- sudden trouble seeing in one or both eyes;
- sudden, severe or prolonged headache with no known cause;
- loss of consciousness or fainting with or without seizure.
Temporary symptoms suggest the event is a transient ischaemic attack (TIA).
Symptoms of myocardial infarction (MI) can include:
- pain, discomfort, pressure, heaviness, sensation of squeezing or fullness in the chest, arm, or below the breastbone;
- discomfort radiating to the back, jaw, throat, arm, stomach;
- feeling of being full, having indigestion or choking;
- sweating, nausea, vomiting or dizziness;
- extreme weakness, anxiety, or shortness of breath;
- rapid or irregular heartbeats.
Women using combined contraceptives should be emphatically advised to contact their physician in case of possible symptoms of thrombosis. In case of suspected or confirmed thrombosis, hormonal contraceptive use should be discontinued. Adequate contraception should be initiated because of the teratogenicity of anti-coagulant therapy (coumarins).
Tumours
An increased risk of cervical cancer in long-term users of COCs has been reported in some epidemiological studies, but there continues to be controversy about the extent to which this finding is attributable to the confounding effects of sexual behaviour and other factors such as human papilloma virus (HPV).
A meta-analysis of 54 epidemiological studies reported that there is a slightly increased risk (RR = 1.24) of having breast cancer diagnosed in women who are currently using COCs. The excess risk gradually disappears during the course of the 10 years after cessation of COC use. Because breast cancer is rare in women under 40 years of age, the excess number of breast cancer diagnoses in current and recent COC users is small in relation to the overall risk of breast cancer. The breast cancers diagnosed in ever-users tend to be less advanced clinically than the cancers diagnosed in never-users. The observed pattern of increased risk may be due to an earlier diagnosis of breast cancer in COC users, the biological effects of COCs or a combination of both.
In rare cases, benign liver tumours, and even more rarely, malignant liver tumours have been reported in users of COCs. In isolated cases, these tumours have led to life-threatening intra-abdominal haemorrhages. Therefore a hepatic tumour should be considered in the differential diagnosis when severe upper abdominal pain, liver enlargement or signs of intra-abdominal haemorrhage occur in women using EVRA.
ALT elevations
During clinical trials with patients treated for hepatitis C virus infections (HCV) with the medicinal products containing ombitasvir/paritaprevir/ritonavir and dasabuvir with or without ribavirin, transaminase (ALT) elevations higher than 5 times the upper limit of normal (ULN) occurred significantly more frequent in women using ethinyl estradiol-containing medications such as combined hormonal contraceptives (CHCs) (see sections 4.3 and 4.5).
Psychiatric Disorders
Depressed mood and depression are well-known undesirable effects of hormonal contraceptive use (see section 4.8). Depression can be serious and is a well-known risk factor for suicidal behaviour and suicide. Women should be advised to contact their physician in case of mood changes and depressive symptoms, including shortly after initiating the treatment.
Other conditions
- Contraceptive efficacy may be reduced in women weighing equal or greater than 90 kg (see sections 4.2 and 5.1).
- Women with hypertriglyceridaemia, or a family history thereof, may be at an increased risk of pancreatitis when using combined hormonal contraceptives.
- Although small increases of blood pressure have been reported in many women using hormonal contraceptives, clinically relevant increases are rare. A definitive relationship between hormonal contraceptive use and clinical hypertension has not been established. If, during the use of combined hormonal contraceptives in pre-existing hypertension, constantly elevated blood pressure values or a significant increase in blood pressure do not respond adequately to antihypertensive treatment, the combined hormonal contraceptive must be withdrawn. Combined hormonal contraceptive use may be resumed if normotensive values can be achieved with antihypertensive therapy.
- The following conditions have been reported to occur or deteriorate with both pregnancy and COC use, but the evidence of an association with COC use is inconclusive: Jaundice and/or pruritus related to cholestasis; gallbladder disease including cholecystitis and cholelithiasis; porphyria; systemic lupus erythematosus; haemolytic ureamic syndrome; Sydenham's chorea; herpes gestationis; otosclerosis-related hearing loss.
- Acute or chronic disturbances of liver function may necessitate the discontinuation of combined hormonal contraceptives until markers of liver function return to normal. Recurrence of cholestatic-related pruritus, which occurred during a previous pregnancy or previous use of sex steroids necessitates the discontinuation of combined hormonal contraceptives.
- Although combined hormonal contraceptives may have an effect on peripheral insulin resistance and glucose tolerance, there is no evidence for a need to alter the therapeutic regimen in diabetes during use of combined hormonal contraceptives. However, diabetic women should be carefully observed, particularly in the early stage of EVRA use.
- Worsening of endogenous depression, of epilepsy, of Crohn's disease and of ulcerative colitis has been reported during COC use.
- Chloasma may occasionally occur with the use of hormonal contraception, especially in users with a history of chloasma gravidarum. Users with a tendency to chloasma should avoid exposure to the sun or ultraviolet radiation while using EVRA. Chloasma is often not fully reversible.
Medical examination / consultation
Prior to the initiation or reinstitution of EVRA a complete medical history (including family history) should be taken and pregnancy should be ruled out. Blood pressure should be measured and a physical examination should be performed guided by the contra-indications (see section 4.3) and warnings (see section 4.4). It is important to draw a woman's attention to the information on venous and arterial thrombosis, including the risk of EVRA compared with other CHCs, the symptoms of VTE and ATE, the known risk factors and what to do in the event of a suspected thrombosis.
The woman should also be instructed to carefully read the user leaflet and to adhere to the advice given. The frequency and nature of examinations should be based on established practice guidelines and be adapted to the individual woman.
Women should be advised that hormonal contraceptives do not protect against HIV infections (AIDS) and other sexually transmissible diseases.
Bleeding irregularities
With all combined hormonal contraceptives, irregular blood loss (spotting or breakthrough bleeding) can occur, especially during the initial months of usage. For this reason, a medical opinion on irregular blood loss will only be useful after an adjustment period of approximately three cycles. If breakthrough bleeding persists, or breakthrough bleeding occurs after previously regular cycles, while EVRA has been used according to the recommended regimen, a cause other than EVRA should be considered. Non-hormonal causes should be considered and, if necessary, adequate diagnostic measures taken to rule out organic disease or pregnancy. This may include curettage. In some women withdrawal bleeding may not occur during this transdermal patch free period. If EVRA has been taken according to the directions described in section 4.2, it is unlikely that the woman is pregnant. However, if EVRA has not been taken according to these directions prior to the first missed withdrawal bleed or if two withdrawal bleeds are missed, pregnancy must be ruled out before EVRA use is continued.
Some users may experience amenorrhoea or oligomenorrhoea after discontinuing hormonal contraception, especially when such a condition was pre-existent.
4.5. Interaction with other medicinal products and other forms of interaction
Note: The prescribing information of concomitant medicinal products should be consulted to identify potential interactions.
Pharmacodynamic interactions
Concomitant use with the medicinal products containing ombitasvir/paritaprevir/ritonavir and dasabuvir, with or without ribavirin may increase the risk of ALT elevations (see sections 4.3 and 4.4). Therefore, EVRA-users must switch to an alternative method of contraception (e.g., progestagen-only contraception or non-hormonal methods) prior to starting therapy with this combination drug regimen. EVRA can be restarted 2 weeks following completion of treatment with this combination drug regimen.
Effects of other medicinal products on EVRA
Interactions can occur with drugs that induce microsomal enzymes which can result in increased clearance of sex hormones and which may lead to breakthrough bleeding and/or contraceptive failure. The following interactions have been reported in the literature.
Substances increasing the clearance of CHCs (diminished efficacy of CHCs by enzyme-induction), e.g.
Barbiturates, bosentan, carbamazepine, phenytoin, primidone, rifampicin, modafinil, and HIV medications ritonavir, nevirapine andefavirenz; and possibly also felbamate, griseofulvin, oxcarbazepine, topiramate and products containing the herbal remedy St. John's Wort (hypericum perforatum).
Management
Enzyme induction may be observed after a few days of treatment. Maximal enzyme induction is generally seen in about 10 days but may then be sustained for at least 4 weeks after the cessation of medicinal product therapy.
Short-term
A woman on short-term treatment with medicinal products that induce hepatic drug metabolising enzymes or individual active substances that induce these enzymes should temporarily use a barrier method in addition to EVRA, i.e. during the time of concomitant medicinal product administration and for 28 days after their discontinuation.
If concomitant medicinal product administration extends beyond the end of the three-week patch period, the next transdermal patch should be applied without the usual transdermal patch-free interval.
Long-term
In women on long-term treatment with enzyme-inducing active substances, another reliable, non-hormonal, method of contraception is recommended.
Substances with variable effects on the clearance of CHCs
When co-administered with CHCs, many combinations of HIV protease inhibitors and non-nucleoside reverse transcriptase inhibitors, including combinations with HCV inhibitors can increase or decrease plasma concentrations of estrogen or progestins. The net effect of these changes may be clinically relevant in some cases.
Therefore, the prescribing information of concomitant HIV medications should be consulted to identify potential interactions and any related recommendations. In case of any doubt, an additional barrier contraceptive method should be used by women on protease inhibitor or non-nucleoside reverse transcriptase inhibitor therapy.
Inhibition of ethinyl estradiol metabolism
Etoricoxib has been shown to increase plasma levels of ethinyl estradiol (50 to 60%) when taken concomitantly with an oral triphasic hormonal contraceptive. It is thought that etoricoxib increases ethinyl estradiol levels because it inhibits sulfotransferase activity thereby inhibiting ethinyl estradiol metabolism.
Effect of EVRA on other medicinal products
Hormonal contraceptives may affect the metabolism of certain other active substances. Accordingly, plasma and tissue concentrations may increase (e.g. ciclosporin). Dosage adjustment of the concomitant medicinal product may be necessary.
Lamotrigine: Combined hormonal contraceptives have been shown to significantly decrease plasma concentrations of lamotrigine when coadministered likely due to induction of lamotrigine glucuronidation. This may reduce seizure control; therefore, dosage adjustments of lamotrigine may be necessary.
Laboratory tests
The use of contraceptive steroids may influence the results of certain laboratory tests, including biochemical parameters of liver, thyroid, adrenal and renal function, plasma levels of (carrier) proteins, e.g. corticosteroid-binding globulin and lipid/lipoprotein fractions, parameters of carbohydrate metabolism and parameters of coagulation and fibrinolysis. Changes generally remain within the normal laboratory range.
4.6. Fertility, pregnancy and lactation
Pregnancy
EVRA is not indicated during pregnancy.
Epidemiological studies indicate no increased risk of birth defects in children born to women who used combined oral contraceptives prior to pregnancy. The majority of recent studies also do not indicate a teratogenic effect when combined oral contraceptives are used inadvertently during early pregnancy.
Limited data on the outcomes of exposed pregnancies in women using EVRA do not allow for conclusions about its safety during pregnancy.
Animal studies have shown undesirable effects during pregnancy and lactation (see section 5.3). Based on these animal data, undesirable effects due to hormonal action of the active compounds cannot be excluded. However, general experience with combined oral contraceptives during pregnancy did not provide evidence for an actual undesirable effect in humans.
If pregnancy occurs during use of EVRA, EVRA should be stopped immediately.
The increased risk of VTE during the postpartum period should be considered when re-starting EVRA (see sections 4.2 and 4.4).
Breast-feeding
Breast-feeding may be influenced by combined hormonal contraceptives as they may reduce the quantity and change the composition of breast milk. Therefore, the use of EVRA is not to be recommended until the breast-feeding mother has completely weaned her child.
Fertility
Women may experience a delay in conception following discontinuation of EVRA.
4.7. Effects on ability to drive and use machines
EVRA has no or negligible influence on the ability to drive and use machines.
4.8. Undesirable effects
Summary of the safety profile
The most commonly reported adverse reactions in clinical trials were headache, nausea, and breast tenderness, occurring in approximately 21.0%, 16.6%, and 15.9% of patients, respectively. Adverse reactions that may occur at the beginning of treatment but usually diminish after the first three cycles include spotting, breast tenderness and nausea.
Description of selected adverse reactions
An increased risk of arterial and venous thrombotic and thrombo-embolic events, including myocardial infarction, stroke, transient ischemic attacks, venous thrombosis and pulmonary embolism has been observed in women using CHCs, which are discussed in more detail in section 4.4.
Tabulated list of adverse reactions
Safety was evaluated in 3,322 sexually active women who participated in three Phase III clinical trials, which were designed to evaluate contraceptive efficacy. These subjects received six or 13 cycles of contraception (EVRA or oral contraceptive comparator), took at least one dose of study medicinal product and provided safety data. Table 1 below reflects the adverse reactions reported in clinical trials and from post-marketing experience. Frequency MedDRA convention: very common: (≥1/10); common: (≥1/100 to <1/10); uncommon: (≥1/1,000 to <1/100); rare: (≥1/10,000 to <1/1,000); very rare: (<1/10,000); not known (cannot be estimated from the available data).
Table 1. Frequency of adverse reactions:
Infections and infestations
Common: (Vulvo) vaginal fungal infection, Vaginal candidiasis
Rare: Rash pustular*, Application site pustules
Neoplasms benign, malignant and unspecified (incl cysts and polyps)
Rare: Hepatic neoplasm*†, Breast cancer*†, Cervix carcinoma*†, Hepatic adenoma*†, Uterine leiomyoma, Fibroadenoma of breast
Immune system disorders
Uncommon: Hypersensitivity
Rare: Anaphylactic reaction*
Metabolism and nutrition disorders
Uncommon: Hypercholesterolaemia, Fluid retention, Increased appetite
Rare: Hyperglycaemia*, Insulin resistance*
Psychiatric disorders
Common: Mood, affect and anxiety disorders
Uncommon: Insomnia, Libido decreased
Rare: Anger*, Frustration*, Libido increased
Nervous system disorders
Very common: Headache
Common: Migraine, Dizziness
Rare: Cerebrovascular accident**†, Cerebral haemorrhage*†, Abnormal taste*
Eye disorders
Rare: Contact lens intolerance*
Cardiac disorders
Rare: Arterial thromboembolism (Acute) myocardial infarction*†
Vascular disorders
Uncommon: Hypertension
Rare: Hypertensive crisis*, Arterial thrombosis**†, Venous thrombosis**†, Thrombosis*†, Venous thromboembolism
Respiratory, thoracic and mediastinal disorders
Rare: Pulmonary (artery) thrombosis*†, Pulmonary embolism†
Gastrointestinal disorders
Very common: Nausea
Common: Abdominal pain, Vomiting, Diarrhoea, Abdominal distension
Rare: Colitis*
Hepatobiliary disorders
Rare: Cholecystitis, Cholelithiasis†, Hepatic lesion*, Jaundice cholestatic*†, Cholestasis*†
Skin and subcutaneous tissue disorders
Common: Acne, Rash, Pruritus, Skin reaction, Skin irritation
Uncommon: Alopecia, Dermatitis allergic, Eczema, Photosensitivity reaction, Dermatitis contact, Urticaria, Erythema
Rare: Angioedema*, Erythema* (multiforme, nodosum), Chloasma†, Exfoliative rash*, Pruritus generalised, Rash (erythematous, pruritic), Seborrhoeic dermatitis*
Musculoskeletal and connective tissue disorders
Common: Muscle spasms
Reproductive system and breast disorders
Very common: Breast tenderness
Common: Dysmenorrhoea, Vaginal bleeding and menstrual disorders**†, Uterine spasm, Breast disorders, Vaginal discharge
Uncommon: Galactorrhoea, Premenstrual syndrome, Vulvovaginal dryness
Rare: Cervical dysplasia*, Suppressed lactation*, Genital discharge
General disorders and administration site conditions
Common: Malaise, Fatigue, Application site reactions (erythema, irritation, pruritus, rash)
Uncommon: Generalised oedema, Oedema peripheral, Application site reactions**
Rare: Face oedema*, Pitting oedema*, Swelling, Application site reactions* (e.g., abscess, erosion), Localised oedema*
Investigations
Common: Weight increased
Uncommon: Blood pressure increased, Lipid disorders**
Rare: Blood glucose decreased*†, Blood glucose abnormal*†
* Post-marketing reports.
** Includes adverse reactions reported in clinical trials and post-marketing reports.
† See section 4.4.
Reporting of suspected adverse reactions
Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the national reporting system listed in Appendix V.
6.2. Incompatibilities
Not applicable.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.