FETROJA Powder for solution for injection Ref.[10076] Active ingredients: Cefiderocol
Source: FDA, National Drug Code (US) Revision Year: 2020
Product description
FETROJA is a cephalosporin antibacterial drug product consisting of cefiderocol sulfate tosylate for intravenous infusion. Cefiderocol functions as a siderophore [see Microbiology (12.4)].
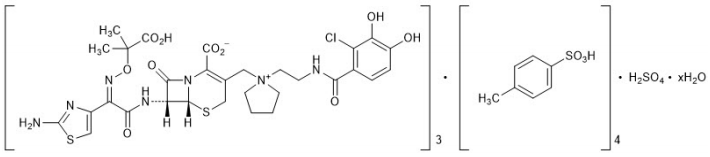
The chemical name of cefiderocol sulfate tosylate is Tris[(6R,7R)-7-[(2Z)-2-(2-amino-1,3-thiazol-4-yl)-2-{[(2-carboxypropan-2-yl)oxy]imino}acetamido]-3-({1-[2-(2-chloro-3,4-dihydroxybenzamido)ethyl]pyrrolidin-1-ium-1-yl}methyl)-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate] tetrakis(4-methylbenzenesulfonate) monosulfate hydrate, and the molecular weight is 3043.50 (anhydrous). The molecular formula is 3C30H34ClN7O10S2∙4C7H8O3S∙H2SO4∙xH2O.
FETROJA for injection is a white to off-white, sterile, lyophilized powder formulated with 1 gram of cefiderocol (equivalent to 1.6 grams of cefiderocol sulfate tosylate), sucrose (900 mg), sodium chloride (216 mg), and sodium hydroxide to adjust pH. The sodium content is approximately 176 mg/vial. The pH of the reconstituted solution of 1 gram cefiderocol (1 vial) dissolved in 10 mL water is 5.2 to 5.8.
| Dosage Forms and Strengths |
|---|
|
FETROJA 1 gram for injection is supplied as a white to off-white, sterile, lyophilized powder for reconstitution in single-dose, clear glass vials; each vial contains 1 gram of cefiderocol. |
| How Supplied |
|---|
|
FETROJA 1 gram (cefiderocol) for injection is supplied as a white to off-white sterile lyophilized powder for reconstitution in single-dose, clear glass vials (NDC 59630-266-01) sealed with a rubber stopper (not made with natural rubber latex) and an aluminum seal with flip-off cap. Each vial is supplied in cartons containing 10 single-dose vials. NDC 59630-266-10 - FETROJA (cefiderocol) 1 gram/vial, 10 vials/carton |
Drugs
| Drug | Countries | |
|---|---|---|
| FETROJA | Estonia, United States |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.