ICLUSIG Film-coated tablet Ref.[9024] Active ingredients: Ponatinib
Source: European Medicines Agency (EU) Revision Year: 2019 Publisher: Incyte Biosciences Distribution B.V., Paasheuvelweg 25, 1105 BP Amsterdam, Netherlands
Pharmacodynamic properties
Pharmacotherapeutic group: antineoplastic agents, protein kinase inhibitors
ATC code: L01XE24
Ponatinib is a potent pan BCR-ABL inhibitor with structural elements, including a carbon-carbon triple-bond, that enable high affinity binding to native BCR-ABL and mutant forms of the ABL kinase. Ponatinib inhibits the tyrosine kinase activity of ABL and T315I mutant ABL with IC50 values of 0.4 and 2.0 nM, respectively. In cellular assays, ponatinib was able to overcome imatinib, dasatinib, and nilotinib resistance mediated by BCR-ABL kinase domain mutations. In preclinical mutagenesis studies, 40 nM was determined as the concentration of ponatinib sufficient to inhibit viability of cells expressing all tested BCR-ABL mutants by >50% (including T315I) and suppress the emergence of mutant clones. In a cell-based accelerated mutagenesis assay, no mutation in BCR-ABL was detected that could confer resistance to 40 nM ponatinib. Ponatinib elicited tumour shrinkage and prolonged survival in mice bearing tumours expressing native or T315I mutant BCR-ABL.
At doses of 30 mg or greater plasma steady state trough concentrations of ponatinib typically exceed 21 ng/mL (40 nM). At doses of 15 mg or greater, 32 of 34 patients (94%) demonstrated a ≥50% reduction of CRK-like (CRKL) phosphorylation, a biomarker of BCR-ABL inhibition, in peripheral blood mononuclear cells.
Ponatinib inhibits the activity of other clinically relevant kinases with IC50 values below 20 nM and has demonstrated cellular activity against RET, FLT3, and KIT and members of the FGFR, PDGFR, and VEGFR families of kinases.
Clinical efficacy and safety
The safety and efficacy of Iclusig in CML and Ph+ ALL patients who were resistant or intolerant to prior tyrosine kinase inhibitor (TKI) therapy were evaluated in a single-arm, open-label, international, multicenter trial. All patients were administered 45 mg of Iclusig once-daily with the possibility of dose de-escalations and dose interruptions followed by dose resumption and re-escalation. Patients were assigned to one of six cohorts based on disease phase (CP-CML; AP-CML; or BPCML/Ph+ ALL), resistance or intolerance (R/I) to dasatinib or nilotinib, and the presence of the T315I mutation. The trial is ongoing.
Resistance in CP-CML was defined as failure to achieve either a complete haematological response (by 3 months), a minor cytogenetic response (by 6 months), or a major cytogenetic response (by 12 months) while on dasatinib or nilotinib. CP-CML patients who experienced a loss of response or development of a kinase domain mutation in the absence of a complete cytogenetic response or progression to AP-CML or BP-CML at any time on dasatinib or nilotinib were also considered resistant. Resistance in AP-CML and BP-CML/Ph+ ALL was defined as failure to achieve either a major haematological response (AP-CML by 3 months, BP-CML/Ph+ ALL by 1 month), loss of major haematological response (at any time), or development of kinase domain mutation in the absence of a major haematological response while on dasatinib or nilotinib.
Intolerance was defined as the discontinuation of dasatinib or nilotinib due to toxicities despite optimal management in the absence of a complete cytogenetic response for CP CML patients or major haematological response for AP CML, BP CML, or Ph+ ALL patients.
The primary efficacy endpoint in CP-CML was major cytogenetic response (MCyR), which included complete and partial cytogenetic responses (CCyR and PCyR). The secondary efficacy endpoints in CP-CML were complete haematological response (CHR) and major molecular response (MMR).
The primary efficacy endpoint in AP-CML and BP-CML/Ph+ ALL was major haematological response (MaHR), defined as either a complete haematological response (CHR) or no evidence of leukaemia (NEL). The secondary efficacy endpoints in AP-CML and BP-CML/Ph+ ALL were MCyR and MMR.
For all patients, additional secondary efficacy endpoints included: confirmed MCyR, time to response, duration of response, progression free survival, and overall survival. Also, post-hoc analyses to assess the relationship of shorter-term cytogenetic (MCyR) and molecular (MMR) response outcomes with longer-term outcomes of PFS and OS, maintenance of response (MCyR and MMR) after dose reductions, and PFS and OS by Arterial Occlusive Event status were conducted.
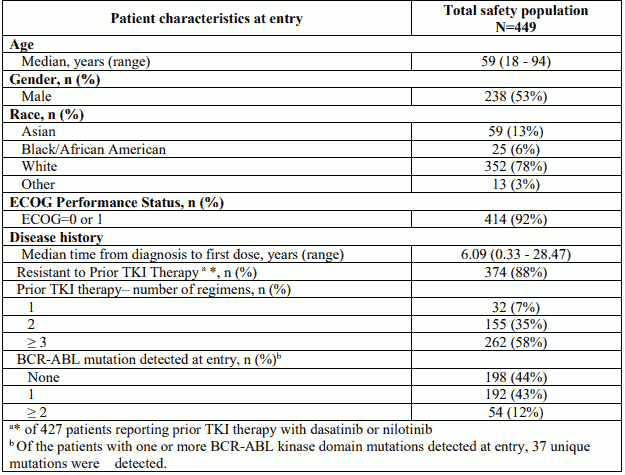
The trial enrolled 449 patients of which 444 were eligible for analysis: 267 CP-CML patients (R/I Cohort: n=203, T315I Cohort: n=64), 83 AP-CML patients (R/I Cohort: n=65, T315I Cohort: n=18), 62 BP-CML (R/I Cohort: n=38, T315I Cohort: n=24), and 32 Ph+ ALL patients (R/I Cohort: n=10, T315I Cohort: n=22). A prior MCyR or better (MCyR, MMR, or CMR) to dasatinib or nilotinib was only achieved in 26% patients with CP-CML and a prior MaHR or better (MaHR, MCyR, MMR, or CMR) was only achieved in 21%, and 24% of AP-CML, and BP-CML/Ph+ALL patients, respectively. Baseline demographic characteristics are described in Table 6 below.
Table 6. Demographics and disease characteristics:
Overall, 55% of patients had one or more BCR-ABL kinase domain mutation at entry with the most frequent being: T315I (29%), F317L (8%), E255K (4%) and E359V (4%). In 67% of CP-CML patients in the R/I cohort, no mutations were detected at study entry.
Efficacy results are summarized in Table 7, Table 8, and Table 9.
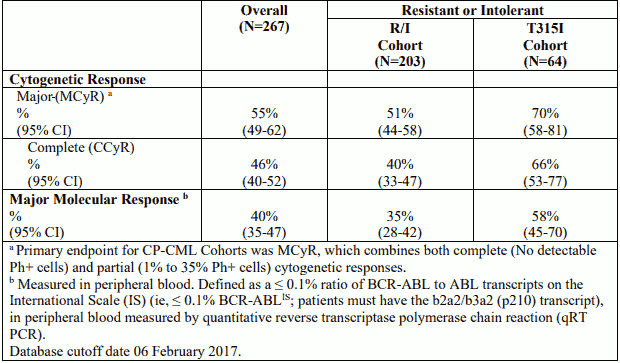
Table 7. Efficacy of Iclusig in resistant or intolerant chronic phase CML patients:
CP-CML patients who received fewer prior TKIs attained higher cytogenetic, haematological, and molecular responses. Of the CP-CML patients previously treated with one, two, three or four prior TKIs, 75% (12/16), 68% (66/97), 44% (63/142), and 58% (7/12)) achieved a MCyR while on Iclusig, respectively.
Of the CP-CML patients with no mutation detected at entry, 49% (66/136) achieved a MCyR.
For every BCR-ABL mutation detected in more than one CP-CML patient at entry, a MCyR was achieved following treatment with Iclusig.
In CP-CML patients who achieved MCyR, the median time to MCyR was 2.8 months (range: 1.6 to 11.3 months) and in patients who achieved MMR, the median time to MMR was 5.5 months (range: 1.8 to 55.5 months). At the time of updated reporting with minimum follow-up for all ongoing patients of 64 months, the median durations of MCyR and MMR had not yet been reached. Based on the Kaplan-Meier estimates, 82% (95% CI: [74%–88%]) of CP-CML (median duration of treatment: 32.2 months) patients who achieved a MCyR are projected to maintain that response at 48 months and 61% (95% CI: [51%-70%]) of CP-CML patients who achieved a MMR are projected to maintain that response at 36 months. The probability of all patients with CP CML maintaining MCyR and MMR did not change further when the analysis was extended out to 5 years.
With a minimum follow-up of 64 months, 3.4% (9/267) of CP-CML patients experienced transformation of their disease to AP-CML or BP-CML.
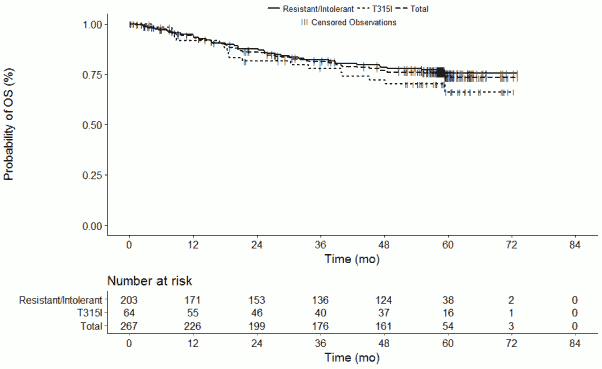
For CP-CML patients overall (N=267), as well as for CP-CML R/I Cohort A patients (N=203) and T315I Cohort B patients (N=64), the median OS has not yet been reached. For the overall CP-CML disease group, the probability of survival at 2, 3, 4, and 5 years is estimated as 86.0%, 81.2%, 76.9%, and 73.3%, respectively, as shown in Figure 1.
Figure 1. Kaplan-Meier estimates for overall survival in the CP-CML population (Treated Population):
CP-CML patients who achieved MCyR or MMR response within the first year of treatment had statistically significantly improved progression-free (PFS) and overall survival (OS) compared to those patients who did not meet the treatment milestones. A MCyR at the 3-month landmark correlated strongly and statistically significantly with PFS and OS (p<0.0001 and p=0.0006, respectively). Statistical significance was achieved in the correlation of PFS and OS with a MCyR at the 12-month landmark (p=<0.0001 and p=0.0012, respectively).
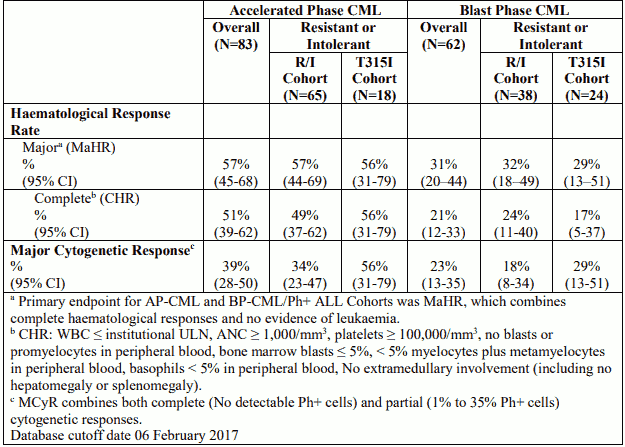
Table 8. Efficacy of Iclusig in resistant or intolerant advanced phase CML patients:
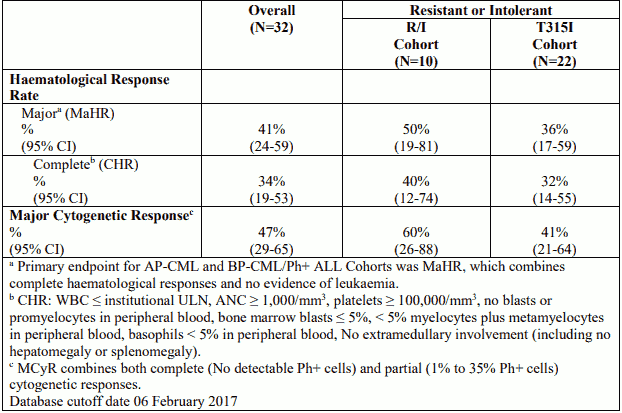
Table 9. Efficacy of Iclusig in resistant or intolerant Ph+ ALL patients:
The median time to MaHR in patients with AP-CML, BP-CML, and Ph+ ALL was 0.7 months (range: 0.4 to 5.8 months), 1.0 months (range: 0.4 to 3.7 months), and 0.7 months (range: 0.4 to 5.5 months), 19 respectively. At the time of updated reporting with minimum follow-up for all ongoing patients of 64 months, the median duration of MaHR for AP-CML (median duration of treatment: 19.4 months) BP-CML (median duration of treatment: 2.9 months), and Ph+ ALL (median duration of treatment: 2.7 months) patients was estimated as 12.9 months (range: 1.2 to 68.4 months), 6.0 months (range: 1.8 to 59.6 months), and 3.2 months (range: 1.8 to 12.8 months), respectively.
For all patients in the phase 2 trial, the dose intensity-safety relationship indicated that there are significant increases in grade ≥3 adverse events (cardiac failure, arterial thrombosis, hypertension, thrombocytopenia, pancreatitis, neutropenia, rash, ALT increase, AST increase, lipase increase, myelosuppression, arthralgia) over the dose range of 15 to 45 mg once-daily.
The analysis of the dose intensity-safety relationship in the phase 2 trial concluded that after adjusting for covariates, the overall dose intensity is significantly associated with an increased risk of vascular occlusion, with an odds ratio of approximately 1.6 for each 15 mg increase. In addition, results from logistic regression analyses of data from patients in the phase 1 trial, suggest a relationship between systemic exposure (AUC) and occurrence of arterial thrombotic events. A reduction in dose is therefore expected to reduce the risk of vascular occlusive events, however, the analysis suggested that there may be a 'carry over' effect of higher doses such that it might take up to several months before a dose reduction manifests in risk reduction. Other covariates that show a statistically significant association with the occurrence of vascular occlusive events in this analysis are medical history of ischemia and age.
Dose reduction in CP-CML patients
In the phase 2 trial, dose reductions were recommended following adverse events; in addition in October 2013 new recommendations for prospective dose reduction in all CP-CML patients in the absence of adverse events were introduced in this trial with the aim of reducing the risk of vascular occlusive events.
With a minimum follow-up of 48 months, and approximately 2 years after the recommendation for prospective dose reduction, there were 110 CP-CML patients ongoing. A majority of these ongoing patients (82/110 patients; 75%) were reported to be receiving 15 mg at the last dose, while 24/110 patients (22%) were receiving 30 mg, and 4/110 (4%) were receiving 45 mg. At the time of study closure initiation (minimum follow-up of 64 months, and more than 3 years after the recommendation for prospective dose reduction), 99 CP-CML patients were ongoing and 77 (78%) of these patients received 15 mg as their last dose on study.
Safety
In the phase 2 trial, 86 CP-CML patients achieved MCyR at a dose of 45 mg, 45 CP-CML patients achieved MCyR after a dose reduction to 30 mg, mostly for adverse events. Vascular occlusive events occurred in 44 of these 131 patients. Most of these events occurred at the dose at which the patient achieved MCyR; fewer events occurred after dose reduction.
Table 10. Vascular occlusive first adverse events in CP-CML patients who achieved MCyR at 45 mg or 30 mg (data extraction 7 April 2014):
| Most recent dose at onset of first vascular occlusive Event | |||
|---|---|---|---|
| 45 mg | 30 mg | 15 mg | |
| Achieved MCyR at 45 mg (N=86) | 19 | 6 | 0 |
| Achieved MCyR at 30 mg (N=45) | 1 | 13 | 5 |
The median time to onset of the first cardiovascular, cerebrovascular, and peripheral vascular arterial occlusive events was 351, 611, and 605 days, respectively. When adjusted for exposure, the incidence of first arterial occlusive events was greatest in the first two years of follow-up and declined with decreasing daily dose intensity (following recommendation for prospective dose reduction). Factors other than dose may also contribute to this risk of arterial occlusion.
Efficacy
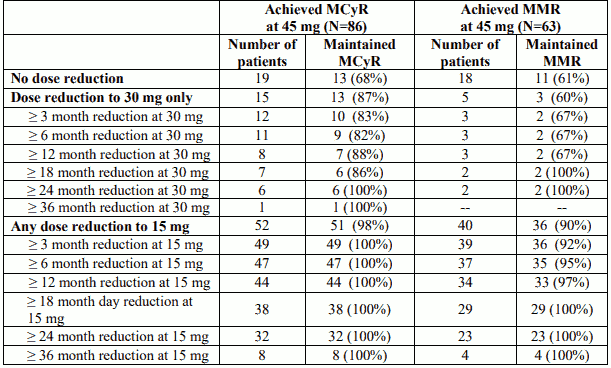
Data from the phase 2 trial are available on the maintenance of response (MCyR and MMR) in all CPCML patients who underwent dose reduction for any reason. Table 11 shows these data for patients who achieved MCyR and MMR at 45 mg; similar data are available for patients who achieved MCyR and MMR at 30 mg.
The majority of patients who underwent dose reduction maintained response (MCyR and MMR) for the duration of currently available follow-up. A proportion of patients did not undergo any dose reduction, based on an individual benefit-risk assessment.
Table 11. Maintenance of response in CP-CML patients who achieved MCyR or MMR at 45 mg dose (data extraction 6 February 2017):
The anti-leukaemic activity of Iclusig was also evaluated in a phase 1 dose escalation study that included 65 CML and Ph+ ALL patients; the study is completed. Of 43 CP-CML patients, 31 CP-CML patients achieved a MCyR with a median duration of follow-up of 55.5 months (range: 1.7 to 91.4 months). At the time of reporting, 25 CP-CML patients were in MCyR (median duration of MCyR had not been reached).
Cardiac electrophysiology
The QT interval prolongation potential of Iclusig was assessed in 39 leukaemia patients who received 30 mg, 45 mg, or 60 mg Iclusig once daily. Serial ECGs in triplicate were collected at baseline and at steady state to evaluate the effect of ponatinib on QT intervals. No clinically significant changes in the mean QTc interval (i.e., >20 ms) from baseline were detected in the study. In addition, the pharmacokinetic-pharmacodynamic models show no exposure-effect relationship, with an estimated QTcF mean change of -6.4 ms (upper confidence interval -0.9 ms) at Cmax for the 60 mg group.
Paediatric population
The European Medicines Agency has waived the obligation to submit the results of studies with Iclusig in children from birth to less than 1 year in CML and Ph+ ALL. The European Medicines Agency has deferred the obligation to submit the results of studies with Iclusig in paediatric patients from 1 year to less than 18 years in CML and Ph+ ALL (see section 4.2 for information on paediatric use).
Pharmacokinetic properties
Absorption
Peak concentrations of ponatinib are observed approximately 4 hours after oral administration. Within the range of clinically relevant doses evaluated in patients (15 mg to 60 mg), ponatinib exhibited dose proportional increases in both Cmax and AUC. The geometric mean (CV%) Cmax and AUC(0-τ) exposures achieved for ponatinib 45 mg daily at steady state were 77 ng/mL (50%) and 1296 ng•hr/mL (48%), respectively. Following either a high-fat and low-fat meal, plasma ponatinib exposures (Cmax and AUC) were not different versus fasting conditions. Iclusig may be administered with or without food. Co-administration of Iclusig with a potent inhibitor of gastric acid secretion resulted in a minor reduction in ponatinib Cmax without a reduction in AUC0-∞.
Distribution
Ponatinib is highly bound (>99%) to plasma proteins in vitro. The blood/plasma ratio of ponatinib is 0.96. Ponatinib is not displaced by concomitant administration of ibuprofen, nifedipine, propranolol, salicylic acid, or warfarin. At daily doses of 45 mg, the geometric mean (CV%) apparent steady state volume of distribution is 1101 L (94%) suggesting that ponatinib is extensively distributed in the extravascular space. In vitro studies suggested that ponatinib is either not a substrate or is a weak substrate for both P-gp and breast cancer resistance protein BCRP. Ponatinib is not a substrate for the human organic anion transporting polypeptides OATP1B1, OATP1B3 and the organic cation transporter OCT-1.
Biotransformation
Ponatinib is metabolized to an inactive carboxylic acid by esterases and/or amidases, and metabolized by CYP3A4 to an N-desmethyl metabolite that is 4 times less active than ponatinib. The carboxylic acid and the N-desmethyl metabolite comprise 58% and 2% of the circulating levels of ponatinib, respectively.
At therapeutic serum concentrations, ponatinib did not inhibit OATP1B1 or OATP1B3, OCT1 or OCT2, organic anion transporters OAT1 or OAT3, or bile salt export pump (BSEP) in vitro. Therefore, clinical medicinal product interactions are unlikely to occur as a result of ponatinib-mediated inhibition of substrates for these transporters. In vitro studies indicate that clinical medicinal product interactions are unlikely to occur as a result of ponatinib-mediated inhibition of the metabolism of substrates for CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP3A or CYP2D6.
An in vitro study in human hepatocytes indicated that clinical medicinal product interactions are also unlikely to occur as a result of ponatinib-mediated induction of the metabolism of substrates for CYP1A2, CYP2B6, or CYP3A.
Elimination
Following single and multiple 45 mg doses of Iclusig, the terminal elimination half-life of ponatinib was 22 hours, and steady state conditions are typically achieved within 1 week of continuous dosing. With once-daily dosing, plasma exposures of ponatinib are increased by approximately 1.5-fold between first dose and steady state conditions. Although plasma ponatinib exposures increased to steady-state levels with continuous dosing, a population pharmacokinetic analysis predicts a limited increase in apparent oral clearance within the first two weeks of continuous dosing, which is not considered clinically relevant. Ponatinib is mainly eliminated via faeces. Following a single oral dose of [14C]-labeled ponatinib, approximately 87% of the radioactive dose is recovered in the faeces and approximately 5% in the urine. Unchanged ponatinib accounted for 24% and <1% of the administered dose in faeces and urine, respectively, with the remainder of the dose comprising metabolites.
Renal impairment
Iclusig has not been studied in patients with renal impairment. Although renal excretion is not a major route of ponatinib elimination, the potential for moderate or severe renal impairment to affect hepatic elimination has not been determined (see section 4.2).
Hepatic impairment
A single dose of 30 mg ponatinib was administered to patients with mild, moderate, or severe hepatic impairment and to healthy volunteers with normal hepatic function. Ponatinib Cmax was comparable in patients with mild hepatic impairment and healthy volunteers with normal hepatic function. In patients with moderate or severe hepatic impairment, ponatinib Cmax and AUC0-∞ were lower and ponatinib plasma elimination half-life was longer in patients with mild, moderate, and severe hepatic impairment but not clinically significantly different than in healthy volunteers with normal hepatic function.
In vitro data showed no difference in plasma protein binding in plasma samples of healthy subjects and hepatically impaired (mild, moderate and severe) subjects. Compared to healthy volunteers with normal liver function, no major differences in ponatinib PK were observed in patients with varying degrees of hepatic impairment. A reduction of the starting dose of Iclusig in patients with hepatic impairment is not necessary (see sections 4.2 and 4.4).
Caution is recommended when administering Iclusig to patients with hepatic impairment (see sections 4.2 and 4.4).
Iclusig has not been studied at doses above 30 mg in patients with hepatic impairment (Childs-Pugh Classes A, B & C).
Intrinsic factors affecting ponatinib pharmacokinetics
No specific studies have been performed to evaluate the effects of gender, age, race, and body weight on ponatinib pharmacokinetics. An integrated population pharmacokinetic analysis completed for ponatinib suggests that age may be predictive of variability for ponatinib apparent oral clearance (CL/F). Gender, race and body weight were not predictive in explaining ponatinib pharmacokinetic intersubject variability.
Preclinical safety data
Iclusig has been evaluated in safety pharmacology, repeat-dose toxicity, genotoxicity, reproductive toxicity, phototoxicity and carcinogenicity studies.
Ponatinib did not exhibit genotoxic properties when evaluated in the standard in vitro and in vivo systems.
Adverse reactions not observed in clinical studies, but seen in animals at exposure levels similar to clinical exposure levels and with possible relevance to clinical use are described below.
Depletion of lymphoid organs was observed in repeat-dose toxicity studies in rats and cynomolgus monkeys. The effects were shown to be reversible after withdrawal of the treatment.
Hyper-/hypoplastic changes of the chondrocytes in the physis were noted in repeat-dose toxicity studies in rats.
In rats, inflammatory changes accompanied by increases in neutrophils, monocytes, eosinophils, and fibrinogen levels were found in the preputial and clitoral glands following chronic dosing.
Skin changes in the form of crusts, hyperkeratosis, or erythema were observed in toxicity studies in cynomolgus monkeys. Dry flaky skin was observed in toxicity studies in rats.
In a study in rats, diffuse corneal edema with neutrophilic cell infiltration, and hyperplastic changes in the lenticular epithelium suggestive of a mild phototoxic reaction were observed in animals treated with 5 and 10 mg/kg ponatinib.
In cynomolgus monkeys, systolic heart murmurs with no macroscopic or microscopic correlates were noted in individual animals treated with 5 and 45 mg/kg in the single dose toxicity study and at 1, 2.5 and 5 mg/kg in the 4-week repeat-dose toxicity study. The clinical relevance of this finding is unknown.
In cynomolgus monkeys, thyroid gland follicular atrophy mostly accompanied by a reduction in T3 levels and a tendency toward increased TSH levels were observed in the 4-week repeat-dose toxicity study in cynomolgus monkeys.
Ponatinib-related microscopic findings in the ovaries (increased follicular atresia) and testes (minimal germ cell degeneration) in animals treated with 5 mg/kg ponatinib were noted in repeat-dose toxicity studies in cynomolgus monkeys.
Ponatinib at doses of 3, 10, and 30 mg/kg produced increases in urine output and electrolyte excretions and caused a decrease in gastric emptying in safety pharmacology studies in rats.
In rats, embryo-foetal toxicity in the form of post-implantation loss, reduced foetal body weight, and multiple soft tissue and skeletal alterations were observed at maternal toxic dosages. Multiple foetal soft tissue and skeletal alterations were also observed at maternal nontoxic dosages.
In a fertility study in male and female rats, female fertility parameters were reduced at dose levels corresponding to human clinical exposures. Evidence for pre- and post-implantation loss of embryos was reported in female rats and ponatinib may therefore impair female fertility. There were no effects on male rat fertility parameters. The clinical relevance of these findings on human fertility is unknown.
In juvenile rats, mortality related to inflammatory effects was observed in animals treated with 3 mg/kg/day, and reductions in body weight gain were observed at doses of 0.75, 1.5 and 3 mg/kg/day during the pre-weaning and early post-weaning treatment phases. Ponatinib did not adversely affect important developmental parameters in the juvenile toxicity study.
In a two-year carcinogenicity study in male and female rats, oral administration of ponatinib at 0.05, 0.1 and 0.2 mg/kg/day in males and at 0.2 and 0.4 mg/kg/day in females did not result in any tumorigenic effects. The 0.8 mg/kg/day dose in females resulted in a plasma exposure level generally lower or equivalent to the human exposure at the range of dose from 15 mg to 45 mg daily. A statistically significant increased incidence of squamous cell carcinoma of the clitoral gland was observed at that dose. The clinical relevance of this finding for humans is not known.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.