IMAAVY Concentrate for solution for infusion Ref.[116055] Active ingredients: Nipocalimab
Source: European Medicines Agency (EU) Revision Year: 2025 Publisher: Janssen-Cilag International NV, Turnhoutseweg 30, B-2340 Beerse, Belgium
5.1. Pharmacodynamic properties
Pharmacotherapeutic group: Immunosuppressants, monoclonal antibodies
ATC code: L04AL03
Mechanism of action
Nipocalimab is a human IgG1 monoclonal antibody specifically targeting the IgG Fc binding site of FcRn with high specificity and high affinity at both neutral (extracellular) and acidic pH (intracellular) resulting in the reduction of circulating IgG, including IgG autoantibodies, without affecting other immunoglobulins (IgA, IgE or IgM). Nipocalimab did not demonstrate any clinically relevant impact on circulating levels of albumin, which binds at a different site on FcRn.
IgG autoantibodies are the underlying cause of the pathogenesis of MG. IgG autoantibodies impair neuromuscular transmission by binding to AChR, MuSK or LRP4.
Nipocalimab reduces the placental transfer of IgG from mother to foetus (see section 4.6).
Pharmacodynamic effects
In a double-blind placebo-controlled study in gMG patients, intravenous administration of nipocalimab at the recommended dose regimen (see section 4.2) resulted in a significant rapid reduction in total IgG serum concentrations of 75% compared to baseline within 2 weeks of treatment initiation, followed by a sustained reduction of approximately 70% compared to baseline from Week 4 through to Week 24. Similar dose-dependent reductions in all IgG subclasses (IgG1, IgG2, IgG3, and IgG4) were observed.
Immunisations (vaccinations)
The impact of nipocalimab on a T-cell dependent (Tdap) and a T-cell independent (PPSV23) vaccine response was assessed in a randomised, open-label study in healthy participants (n=15 for control, n=16 for nipocalimab). In the nipocalimab group, participants received nipocalimab at Week 0 (30 mg/kg IV), Week 2 (15 mg/kg IV) and Week 4 (15 mg/kg IV), and Tdap and PPSV23 were administered subcutaneously 3 days after the first nipocalimab dose.
Participants were able to mount a specific IgG response to these vaccines, but vaccine-specific IgG antibody levels were reduced during nipocalimab treatment with recovery to levels similar to the control group after treatment cessation.The anti-TT (tetanus toxoid) specific IgG levels are shown in Table 3. In participants who received nipocalimab, the anti-TT specific IgG levels achieved the peak response at Week 2, decreased at Week 4 and then increased through Week 16, 12 weeks after the last dose of nipocalimab at Week 4. Anti-PCP (pneumococcal capsular polysaccharide) specific IgG levels followed a similar pattern over time. See sections 4.4 and 4.5.
Table 3. Anti-TT IgG levels (mean±SE) over time:
| Time Point | Nipocalimab (n=16) IU/mL | Control (n=15) IU/mL |
|---|---|---|
| Baseline | 1.97±0.612 | 2.38±0.538 |
| Week 2 | 3.38±0.325 | 4.92±0.619 |
| Week 4 | 1.63±0.269 | 4.56±0.591 |
| Week 8 (4 weeks post last dose) | 2.39±0.491 | 3.87±0.538 |
| Week 16 (12 weeks post last dose) | 2.53±0.223 | 3.20±0.474 |
Immunogenicity
Anti-drug antibodies (ADA) were very commonly detected at low titre. However, no evidence of ADA impact on pharmacokinetics, pharmacodynamics, efficacy or safety was observed.
Clinical efficacy and safety
Study MOM-M281-011 (adults)
The safety and efficacy of nipocalimab for the treatment of adults with gMG was studied in a 24-week, randomised, double-blind, placebo-controlled study. Patients participating in this study were subsequently allowed to enter an open-label extension phase during which all patients received nipocalimab.
The study enrolled patients who met the following main criteria at screening:
- Myasthenia Gravis Foundation of America (MGFA) clinical classification class II to IV
- MG-Activities of Daily Living (MG-ADL) total score of ≥6
- On stable dose of standard of care (SoC) therapy prior to baseline, including acetylcholinesterase (AChE) inhibitors, steroids or non-steroidal immunosuppressive therapies (NSISTs), either in combination or alone.
A total of 196 patients (with or without autoantibodies) were randomised and received either nipocalimab plus SoC (n=98) or placebo plus SoC (n=98). Of these, 153 patients were antibody positive (n=77 for nipocalimab, n=76 for placebo). Patients were treated with nipocalimab at the recommended dose regimen (see section 4.2).
Of the 153 antibody positive patients, 88% were antibody positive for AChR, 10% were antibody positive for MuSK, and 2% were antibody positive for LRP4. Baseline characteristics were similar between treatment groups, including median age at screening (52 [20-81] years, 24% patients ≥65 years of age), median time since diagnosis (6 [0-38] years), gender (60% female), and race (63% white, 32% Asian). Mean MG-ADL total score was 9.2, and mean Quantitative Myasthenia Gravis total score was 15.4.
At baseline, over 97% in each treatment group were on stable background SoC therapy. During treatment, 85% were on AChE inhibitors, 66% were on steroids, and 54% were on non-steroidal immunosuppressive therapies (NSISTs) at stable doses.
The efficacy of nipocalimab was measured using the Myasthenia Gravis Activities of Daily Living scale (MG-ADL) which assesses the impact of gMG on daily functions. A total score ranges from 0 to 24 with the higher scores indicating more impairment. In this study, an MG-ADL response was defined as a ≥2-point reduction in the total MG-ADL score compared to baseline. The efficacy of nipocalimab was also measured using the Quantitative Myasthenia Gravis (QMG) total score which measures muscle weakness. A total possible score ranges from 0 to 39, where higher scores indicate more severe impairment. A QMG response was defined as a ≥3-point reduction in the total QMG score compared to baseline.
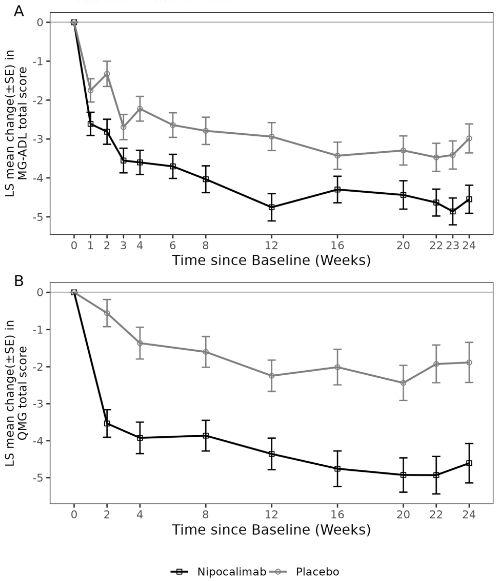
The key efficacy results for the primary and major secondary study endpoints are shown in Table 4. A statistically significant difference favouring nipocalimab was observed on MG-ADL and QMG changes from baseline.
Table 4. Summary of the primary and key secondary clinical responses:
| Nipocalimab (n=77) LS mean (SE) | Placebo (n=76) LS mean (SE) | Nipocalimab change relative to placebo LS mean difference (95% CI) | P-value | |
|---|---|---|---|---|
| MG-ADL1 | -4.68 (0.324) | -3.29 (0.333) | -1.39 (-2.31, -0.47) | 0.003 |
| QMG2 | -4.77 (0.488) | -1.90 (0.491) | -2.87 (-4.23, -1.50) | <0.001 |
| MG-ADL responder based on average change over Weeks 22, 23, and 243 | 68.8% | 52.6% | 16.2 (0.9, 31.5) | 0.021 |
| MG-ADL responder from Week 4 through Week 244 | 55.8% | 26.3% | 29.5 (14.7, 44.4) | - |
| ≥50% improvement on MG-ADL based on average change over Weeks 22, 23 and 245 | 46.8% | 25.0% | 21.8 (7.0, 36.6) | - |
1 Mean change from baseline over Weeks 22, 23, and 24.
2 Mean change from baseline over Weeks 22 and 24.
3 Average change over Weeks 22, 23, and 24 is at least a 2-point improvement from baseline.
4 At least 2-point improvement in MG-ADL total score at Week 4 and Week 24, and at least 2 point improvement at Week 6 through Week 23 with no more than 2 non-consecutive excursions (improvement less than 2 points) allowed.
5 Average change over Weeks 22, 23, and 24 is at least a 50% improvement from baseline.
Response over time (double-blind phase)
Improvements with nipocalimab compared to placebo were observed through Week 24.
The time course of response for the primary efficacy endpoint (MG-ADL) and key secondary efficacy endpoint (QMG) is shown in Figure 1.
Figure 1. Least Squares Mean Change from Baseline in MG-ADL Total Score (A) and QMG Total Score (B) Over 24 Weeks:
Over time, a higher proportion of patients achieved sustained MG-ADL (improvement >2 points from Week 2 through Week 24) and QMG (improvement >3 points from Week 2 through Week 24) responses in the nipocalimab group (45.5% and 33.8%, respectively) compared to the placebo group (21.1% and 7.9%, respectively).
Response over time (open-label extension phase)
Of the 153 antibody positive patients in the double-blind placebo-controlled phase, 137 entered into the open-label extension phase to receive nipocalimab. At the time of the analysis, in patients who initially received nipocalimab during the double-blind phase and continued to receive nipocalimab during the first 48 weeks (n=52) and 84 weeks (n=20) of the open-label extension phase, the mean improvements in MG-ADL and QMG total scores were maintained.
Paediatric population
Study 80202135MYG2001 (adolescent cohort)
The pharmacodynamics, pharmacokinetics, and efficacy of nipocalimab for the treatment of gMG in adolescent patients are evaluated at Week 24 in an ongoing open-label study.
Main study inclusion criteria are as follows:
- MGFA clinical classification class II to IV
- Positive for autoantibodies to AChR or MuSK
- On stable dose of SoC therapy prior to screening, including AChE inhibitors, steroids or NSISTs, either in combination or alone.
Eight patients had a median age of 13.5 years at screening (range 12 to 16 years) and a median time since diagnosis of 3.6 years (range 0.8 to 11.5). Seven patients were female; 5 were Asian, 1 was Black and 2 were of unknown race. Mean (SD) MG-ADL total score at baseline was 4.4 (2.26), and mean (SD) QMG total score was 13.3 (4.13). All patients were AChR antibody positive. At baseline, 4 patients were on AChE inhibitors, 6 were on steroids, and 7 were on NSISTs at stable doses.
Seven of the 8 adolescent patients were evaluated through Week 24 and received nipocalimab at the recommended dose regimen (see section 4.2). The primary endpoint was the effect of nipocalimab on total serum IgG. At Week 24, the median pre-dose percent reduction in total IgG from baseline (n=7) was 73.3%, consistent with the IgG reduction seen in the adult gMG study. The mean (SD) change at Week 24 in MG-ADL was -2.57 (0.535) and the mean change at Week 24 in QMG was -4.93 (3.81).
The European Medicines Agency has deferred the obligation to submit the results of studies with Imaavy in one or more subsets of the paediatric population in the treatment of myasthenia gravis (see section 4.2 for information on paediatric use).
5.2. Pharmacokinetic properties
Distribution
The mean (SD) volume of distribution is 2.84 (0.63) L or 0.0359 (0.0087) L/kg.
Biotransformation
Nipocalimab is expected to be degraded by proteolytic enzymes into small peptides and amino acids via catabolic pathways in the same manner as endogenous IgG.
Elimination
Nipocalimab exhibits concentration-dependent pharmacokinetics. Following a single intravenous administration of 15 mg/kg of nipocalimab, the mean clearance is 0.0627 L/h and half-life is 29.3 hours. After stopping administration, serum IgG concentrations recover towards baseline levels within approximately 8 weeks.
Linearity/non-linearity
Nipocalimab exhibits non-linear, dose-dependent pharmacokinetics. Following a single intravenous infusion of nipocalimab at doses ranging from 0.3 to 60 mg/kg in healthy participants, Cmax increased in a dose-proportional manner while AUC increased in a greater than dose proportional manner.
Due to the relatively short half-life of nipocalimab, repeated dosing administered in accordance with the recommended maintenance dose (see section 4.2) results in no drug accumulation over time.
Special populations
Paediatric population
Pharmacokinetics of nipocalimab were evaluated in adolescent patients 12 to 17 years of age with gMG (n=8). Following treatment with nipocalimab at the recommended dose regimen (see section 4.2), the observed serum nipocalimab concentrations were comparable between adult and adolescent patients with gMG (Table 5).
Table 5. Serum nipocalimab concentrations in adult and adolescent patients with gMG:
| Timepoint | Exposure Parameter | Adolescents (n=8) Median (IQ Range) | Adults (n=97) Median (IQ Range) |
|---|---|---|---|
| Initial dose | Ceoi,ld (μg/mL) | 701 (673, 922) | 864 (774, 1000) |
| Ctrough,ld (μg/mL) | 0.01 (BQL, 0.02) | 0.02 (BQL, 0.03) | |
| Maintenance doses | Ceoi,ss (μg/mL) | 394 (335, 491) | 424 (392, 479) |
| Ctrough,ss (μg/mL) | BQL | BQL |
BQL = Below Quantification Limit (i.e., <0.01 μg/mL); Ceoi,ld = end of infusion concentration after 30 mg/kg initial dose; Ctrough,ld = pre-dose concentration at Week 2 after 30 mg/kg initial dose; Ceoi,ss = end of infusion concentrations at steady state after 15 mg/kg every two weeks maintenance doses; Ctrough,ss = pre-dose concentration at steady state after 15 mg/kg every two weeks maintenance doses; IQ = interquartile.
Elderly
Clinical studies with nipocalimab did not include sufficient numbers of patients aged 65 and older to determine whether they respond differently from younger adult patients. No apparent differences in clearance and volume of distribution were observed in patients ≥65 years of age compared to patients <65 years of age, suggesting no dose adjustment is needed for elderly patients (see section 4.2).
Age, gender, and ethnicity
A population pharmacokinetics analysis assessing the effects of age, sex, and race did not suggest any clinically relevant impact of these covariates on nipocalimab exposures.
Body weight
Body weight has an impact on systemic exposure to nipocalimab. The recommended weight-based dosing (in mg/kg) accounts for the differences in patient body weight.
Renal impairment
No formal studies have been performed in patients with renal impairment. Renal impairment is not expected to affect the pharmacokinetics of nipocalimab. Based on a population pharmacokinetic analysis, which included patients with mild to moderate renal impairment, renal function had no clinically relevant effect on nipocalimab apparent clearance. No dose adjustment is required in patients with renal impairment (see section 4.2).
Hepatic impairment
No formal studies have been performed in patients with hepatic impairment. Nipocalimab is not metabolised by cytochrome P450 enzymes, and therefore, hepatic impairment is not expected to affect the pharmacokinetics of nipocalimab. Based on a population pharmacokinetic analysis, which included participants with mild and a limited number of participants with moderate hepatic impairment, there was no clinically relevant effect on nipocalimab apparent clearance. No dose adjustment is required in patients with hepatic impairment (see section 4.2).
5.3. Preclinical safety data
Non-clinical data reveal no special hazard for humans based on conventional studies of repeated dose toxicity.
There were no nipocalimab-related adverse effects on male and female fertility in non-pregnant cynomolgus monkeys in a 6-month repeat-dose study when evaluated by changes in the reproductive organs (organ weights and histopathology). All females and 80% of males, at the highest dose tested, became sexually mature during the 6-month treatment period in which exposure levels up to 44 times the expected exposure level in patients on the recommended maintenance dose (see section 4.2) were evaluated.
In an enhanced pre- and postnatal development study, where pregnant cynomolgus monkeys were intravenously exposed to nipocalimab during the late first, second and third trimesters of pregnancy, 16% (4/25) of placentas showed large, central placental infarctions and thrombosis of maternal spiral arteries. Three of the 4 cases of placental infarction were associated with second or third trimester foetal losses. The placental infarctions may be related to maternal immunogenicity in pregnant cynomolgus monkeys. The clinical relevance of these findings is unknown. Nipocalimab exposure levels (AUC) in pregnant cynomolgus monkeys reached at least 5 to 24 times the expected exposure level in non-pregnant women based on the recommended maintenance dose regimen (see section 4.2). No pre- or postnatal development concerns were raised. Foetuses and infants from treated dams showed negligible exposure to maternal nipocalimab but had low levels of IgG at birth. The infant IgG levels recovered within 6 months. There was no adverse impact on immune function of the infants of treated dams as assessed by a T-cell Dependent Antibody Response assay.
The mutagenic potential of nipocalimab has not been evaluated; however, monoclonal antibodies are not expected to alter DNA or chromosomes.
Carcinogenicity studies have not been conducted with nipocalimab.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.