JASCAYD Film-coated tablet Ref.[116083] Active ingredients: Nerandomilast
Source: FDA, National Drug Code (US) Revision Year: 2025
12.1. Mechanism of Action
Nerandomilast is an inhibitor of phosphodiesterase 4 (PDE4) with at least nine-fold preferential inhibition of the PDE4B isoenzyme over PDE4A, PDE4C and PDE4D based on in vitro data. PDE4 hydrolyzes and inactivates cyclic adenosine monophosphate (cAMP). Nerandomilast exerts both anti-fibrotic and immunomodulatory effects as PDE4B inhibition elevates intracellular cAMP levels and reduces the expression of pro-fibrotic growth factors and inflammatory cytokines, which are overexpressed in IPF.
12.2. Pharmacodynamics
Exposure-Response
Increases in nerandomilast steady state trough concentrations were associated with better efficacy, as indicated by a smaller reduction in forced vital capacity (FVC) from baseline over 52 weeks.
Cardiac Electrophysiology
At 2.1 times the maximal concentration provided by the maximum recommended dose of nerandomilast, clinically significant QTc interval prolongation was not observed.
12.3. Pharmacokinetics
Nerandomilast exposure increased in a dose proportional manner following administration of single doses of 0.0032 to 2.6 times a single dose of 18 mg and multiple doses of 0.056 to 1 times the maximum recommended dose of 18 mg twice daily.
Following administration of multiple doses of 18 mg twice daily, Cmax increases 1.3-fold and AUCtau increases 1.38-fold. Nerandomilast steady state is reached in approximately 4 days.
No clinically relevant differences in nerandomilast pharmacokinetics were observed between healthy subjects and patients with IPF.
Absorption
The absolute oral bioavailability of nerandomilast is 73%. Nerandomilast median (min, max) time to maximum plasma concentration (Tmax) is 1 to 1.25 hours (range 0.5 to 4 hours).
Effect of Food
No clinically relevant differences in nerandomilast pharmacokinetics were observed following administration with a high-fat meal (1000 calories, 50% fat).
Distribution
Nerandomilast steady state apparent (oral) central volume of distribution is 93 L (CV 37%). In vitro, nerandomilast plasma protein binding is 77% and is not concentration dependent. Nerandomilast blood-to-plasma ratio is approximately 0.6 to 0.8.
Elimination
The geometric mean (CV%) elimination half-life of nerandomilast is 17 hours (46%) with an apparent (oral) clearance of 15.2 L/h.
Metabolism
Nerandomilast is primarily metabolized by CYP3A and to a lesser extent by multiple UGT enzymes.
After oral administration of nerandomilast, chiral inversion from the pharmacologically active R-enantiomer to the pharmacologically inactive S-enantiomer occurs via metabolism. The S-enantiomer was identified as a minor metabolite of nerandomilast. The R-enantiomer is the predominant circulating enantiomer.
Excretion
After a single oral dose of radiolabeled nerandomilast, approximately 58% of the dose was recovered in feces (14% as unchanged nerandomilast) and 36% of the dose was recovered in urine (13% as unchanged nerandomilast).
Specific Populations
No clinically significant differences in the pharmacokinetics of nerandomilast were observed based on age (range: 18 to 90 years), body weight (range: 34 to 136 kg), sex, race (66% White and 32.5% Asian), mild (eGFR ≥60 to <90 mL/min/1.73 m² [calculated according to Chronic Kidney Disease Epidemiology Collaboration]), moderate (eGFR ≥30 to <60 mL/min/1.73 m²), or severe renal impairment (eGFR ≥15 to <30 mL/min/1.73 m²), or mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment.
Subjects with end stage renal disease or severe hepatic impairment (Child-Pugh Class C) have not been studied [see Use in Specific Populations (8.6, 8.7)].
Drug Interaction Studies
Strong CYP3A Inhibitors
Nerandomilast Cmax increased by 1.3-fold and AUC increased by 2.2-fold following concomitant administration with itraconazole (a strong CYP3A and P-gp inhibitor) 200 mg oral solution once daily for 4 days [see Drug Interactions (7.1)].
Pirfenidone
Nerandomilast trough concentrations at steady state (Ctrough,ss) decreased by approximately 50% following concomitant administration with pirfenidone in patients with IPF [see Drug Interactions (7.1)].
Other Drugs
No clinically significant differences in nerandomilast Ctrough,ss were observed when used concomitantly with nintedanib. No clinically significant differences in the pharmacokinetics of the following drugs were observed when used concomitantly with nerandomilast: oral midazolam (CYP3A4 substrate), pirfenidone, and nintedanib.
In Vitro Studies
CYP450 Enzymes: Nerandomilast is a CYP3A4 substrate. Nerandomilast does not inhibit CYP3A4, CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, and CYP2D6.
Transporter Systems: Nerandomilast is a P-glycoprotein (P-gp) substrate, but is not a substrate of BCRP, OATP1B1, OATP1B3, OAT1, OAT3, and OCT2. Nerandomilast is an inhibitor of P-gp, MATE1, MATE2-K, but is not expected to cause clinically significant interactions. Nerandomilast is not an inhibitor of BCRP, OAT1, OAT3, OCT2, OATP1B1, or OATP1B3.
13.1. Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
The carcinogenic potential of nerandomilast was assessed in Tg.rasH2 mice and Wistar Han rats. No evidence of tumorigenicity was observed in male and female Tg.rasH2 mice that received nerandomilast for 26 weeks at oral doses up to 60 and 100 mg/kg/day, respectively. No evidence of tumorigenicity was observed in male or female rats that received nerandomilast for up to 102 weeks at oral doses up to 2 mg/kg/day (approximately 2 times the MRHD on an AUC basis).
Mutagenesis
Nerandomilast was not mutagenic or clastogenic in the following assays: in vitro bacterial reverse mutation (Ames) assay, in vitro chromosomal aberration assay in human peripheral blood lymphocytes, and in vivo rat micronucleus assay.
Impairment of Fertility
Nerandomilast had no effect on fertility in male or female rats at oral doses up to 6 mg/kg/day (approximately 3 or 4 times the MRHD on an AUC basis, respectively). Decreased mating, pregnancy, and fertility indices at the highest tested dose of 9 mg/kg/day (approximately 4 or 9 times the MRHD on an AUC basis) were attributed to excessive general toxicity.
Sexually mature female monkeys administered nerandomilast by the oral route for 39 weeks showed sporadic menstrual cycle prolongation at dose levels of 10 mg/kg/day and 30 mg/kg/day (approximately 3- and 10 times the MRHD on an AUC basis, respectively). Menstrual cycles were not affected in monkeys at oral doses of 3 mg/kg/day (equivalent to the MRHD on an AUC basis).
14. Clinical Studies
The efficacy of JASCAYD was evaluated in two randomized, double-blind, placebo-controlled trials (FIBRONEER-IPF [NCT05321069] and Trial 2 [NCT04419506]).
FIBRONEER-IPF enrolled a total of 1,177 adult patients with IPF with or without background antifibrotic treatments (nintedanib or pirfenidone). They were randomized in a 1:1:1 ratio to receive JASCAYD 9 mg twice daily, JASCAYD 18 mg twice daily, or placebo twice daily until the last patient received treatment for 52 weeks (blinded trial duration up to 91 weeks; end of trial duration up to 109 weeks). Randomization was stratified by the presence or absence of background antifibrotic treatments (nintedanib or pirfenidone) at baseline. The FIBRONEER-IPF trial consisted of 83% male and 17% female patients with a mean age of 70 years (age range: 42 to 90 years). The study population included 68% White, 32% Asian, and <1% Black or African American. For ethnicity, 8% of patients identified as Hispanic or Latino. At baseline, the mean FVC was 78% of predicted normal; 78% of the patients were on stable antifibrotic treatment (nintedanib 46%, pirfenidone 32%) and 22% were not on either treatment (15% treatment naïve, 7% previously discontinued treatment).
Trial 2 was a 12-week trial that enrolled a total of 147 adult patients with IPF with or without background antifibrotic treatments (nintedanib or pirfenidone) and were randomized 2:1 to receive JASCAYD 18 mg twice daily or placebo twice daily for 12 weeks. Randomization was stratified by the presence or absence of background antifibrotic treatments (nintedanib or pirfenidone) at baseline. The study population in Trial 2 consisted of 77% male, and mean age of 70 years (age range: 40 to 85 years). The study population included 78% White and 22% Asian. For ethnicity, 9% of patients identified as Hispanic or Latino. At baseline, the mean FVC was 78% of predicted normal; 50% of the patients were on stable antifibrotic treatment (nintedanib 29%, pirfenidone 21%).
In both the FIBRONEER-IPF trial and Trial 2, patients were required to have a diagnosis of IPF based on ATS/ERS/JRS/ALAT criteria. Diagnosis was confirmed by the investigator based on chest high-resolution computed tomography (HRCT) scan and, if available, lung biopsy, and usual interstitial pneumonia (UIP) or probable UIP HRCT pattern consistent with the clinical diagnosis of IPF. Patients were also required to be greater than or equal to 40 years of age with an FVC greater than or equal to 45% of predicted and a carbon monoxide diffusing capacity (DLCO, corrected for hemoglobin) greater than or equal to 25% of predicted. Prior to Visit 1 and during screening, patients had to be on stable treatment with nintedanib or pirfenidone (no dose changes for at least 12 weeks) and planned to stay on this background antifibrotic treatment after randomization. Alternatively, patients were required to be naïve to or have previously discontinued nintedanib or pirfenidone for at least 8 weeks and did not plan to start or re-start background antifibrotic treatment. Patients with active vasculitis, severe depression or suicidal behavior or ideation, or use of immunomodulatory medications (other than prednisone ≤15 mg/day or equivalent) were excluded.
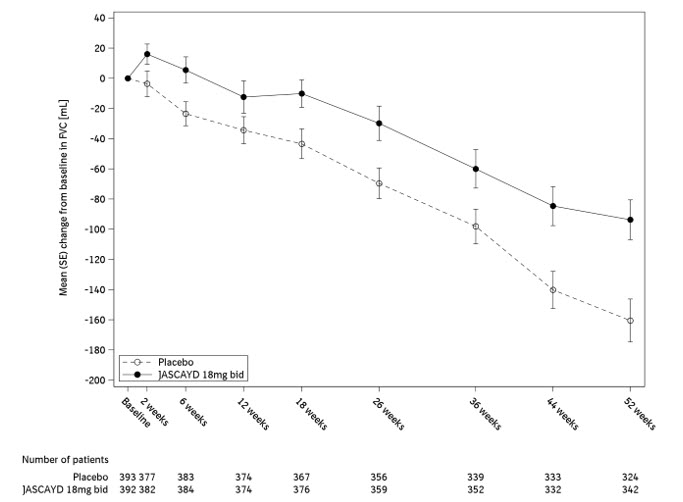
Change from Baseline in Forced Vital Capacity
The primary endpoint in FIBRONEER-IPF trial was the absolute change from baseline in forced vital capacity (FVC) in milliliters (mL) at 52 weeks for JASCAYD compared with placebo.
In the FIBRONEER-IPF study population, there was less decline in absolute change from baseline in FVC in patients who received JASCAYD compared with patients who received placebo (accounting for mortality), and this reduction in decline was statistically significant. The adjusted mean decline in patients receiving JASCAYD 18 mg or JASCAYD 9 mg was -106 mL and -122 mL, respectively, whereas in the placebo group, an adjusted mean decline of -170 mL was observed. The respective treatment differences compared with the placebo group were 64 mL (95% CI: 25, 102) and 48 mL (95% CI: 10, 86).
In the FIBRONEER-IPF trial, the results of the primary endpoint across subgroups by background antifibrotic treatment (nintedanib, pirfenidone, or none) for JASCAYD 18 mg versus placebo were consistent with the overall population. Efficacy was not observed in patients who received JASCAYD 9 mg twice daily with pirfenidone as background antifibrotic treatment [see Drug Interactions (7.1)].
Figure 1 shows the change in FVC from baseline over time in patients who received JASCAYD 18 mg twice daily compared to placebo in the FIBRONEER-IPF trial.
Figure 1. Change from Baseline in FVC (mL) in Adults with IPF over 52 weeks with JASCAYD 18 mg Compared to Placebo (FIBRONEER-IPF):
Display based on descriptive means and standard errors for change from baseline values from patients with results that were available at each week.
bid = twice daily
In Trial 2, patients taking JASCAYD 18 mg twice daily compared to placebo, with or without background antifibrotic treatments, had a reduction in FVC decline at Week 12 of 91 mL (95% CI: 44, 138).
Time To First Acute IPF Exacerbation, First Hospitalization for Respiratory Cause, Or Death
The key secondary endpoint in the FIBRONEER-IPF trial was time to first occurrence of any of the components of the composite endpoint over the blinded duration of the trial (up to 91 weeks): acute IPF exacerbation, hospitalization for respiratory cause, or death. Acute IPF exacerbation was defined as acute worsening or development of dyspnea typically less than one month duration, computed tomography with new bilateral ground-glass opacity and/or consolidation superimposed on a background pattern consistent with IPF, and deterioration not fully explained by cardiac failure or fluid overload. Neither acute IPF exacerbations nor respiratory hospitalizations were adjudicated.
Overall, there was no statistically significant treatment difference in hazard ratio (HR) for the JASCAYD 18 mg or 9 mg groups compared to placebo for the key secondary composite endpoint (JASCAYD 18 mg and 9 mg, respectively: HR 1.17 [95% CI: 0.86, 1.59] and HR 1.03 [95% CI: 0.75, 1.41]).
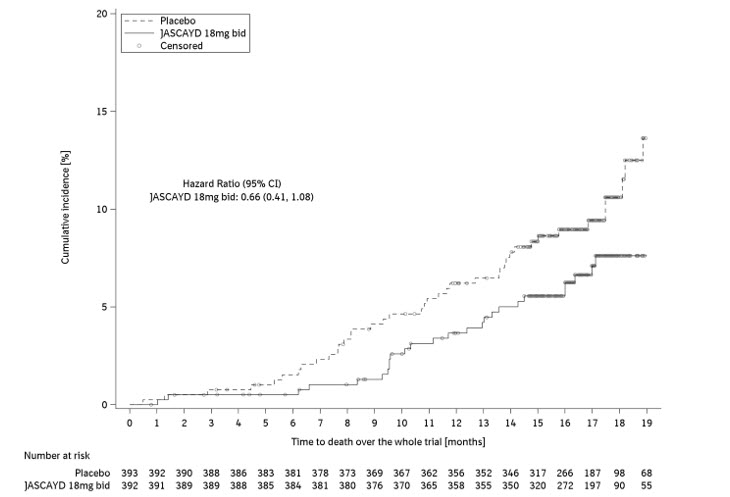
Survival
In the FIBRONEER-IPF study population, the hazard ratio for all-cause mortality, assessed until the end of trial (up to 109 weeks), did not show a significant treatment difference for JASCAYD 18 mg or 9 mg compared to placebo (HR: 0.66 [95% CI: 0.41, 1.08] and HR 0.95 [95% CI: 0.61, 1.49], respectively).
Figure 2 shows the cumulative incidence of death in adults with IPF in the FIBRONEER-IPF trial for JASCAYD 18 mg and placebo.
Figure 2. Cumulative Incidence of Death in Adults with IPF (FIBRONEER-IPF):
bid = twice daily
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.