LOKELMA Powder for oral suspension Ref.[10414] Active ingredients: Sodium zirconium cyclosilicate
Source: FDA, National Drug Code (US) Revision Year: 2020
12.1. Mechanism of Action
LOKELMA (sodium zirconium cyclosilicate) is a non-absorbed zirconium silicate that preferentially captures potassium in exchange for hydrogen and sodium. In vitro, LOKELMA has a high affinity for potassium ions, even in the presence of other cations such as calcium and magnesium. LOKELMA increases fecal potassium excretion through binding of potassium in the lumen of the gastrointestinal tract. Binding of potassium reduces the concentration of free potassium in the gastrointestinal lumen, thereby lowering serum potassium levels.
12.2. Pharmacodynamics
In a study in healthy adult subjects, LOKELMA administered as 5 g or 10 g once daily for four days caused a dose-dependent increase in fecal potassium excretion. Corresponding dose-dependent decreases in urinary potassium excretion and serum potassium were also observed.
In patients with hyperkalemia treated with LOKELMA 10 g three times a day for up to 48 hours, reductions in serum potassium were observed one hour after initiation of therapy; serum potassium concentrations continued to decline over the 48-hour treatment period [see Clinical Studies (14.2)]. In patients not continuing LOKELMA, potassium levels increased. Patients with higher starting serum potassium levels or receiving a higher dose have greater reductions in serum potassium.
LOKELMA causes a small dose-dependent increase in serum bicarbonate concentrations (1.1 mmol/L at 5 g once daily, 2.3 mmol/L at 10 g once daily and 2.6 mmol/L at 15 g once daily as compared with a mean increase of 0.6 mmol/L in patients treated with placebo). The clinical significance of this finding is unclear.
12.3. Pharmacokinetics
LOKELMA is an inorganic, insoluble compound that is not subject to enzymatic metabolism. In a clinical study in patients with hyperkalemia in which zirconium concentrations were measured in the urine and blood, zirconium concentrations were similar in treated and untreated patients (i.e., either undetectable or around the lower limit of quantification of the assay). An in vivo mass balance study in rats showed that LOKELMA was recovered in the feces with no evidence of systemic absorption.
Drug Interactions
Thirty-six (36) drugs were tested to determine potential interactions with LOKELMA.
Sixteen (16) drugs tested did not show an in vitro interaction with LOKELMA (allopurinol, apixaban, aspirin, captopril, cyclosporine, digoxin, ethinyl estradiol, lisinopril, magnesium, metformin, phenytoin, prednisone, propranolol, quinapril, spironolactone and ticagrelor).
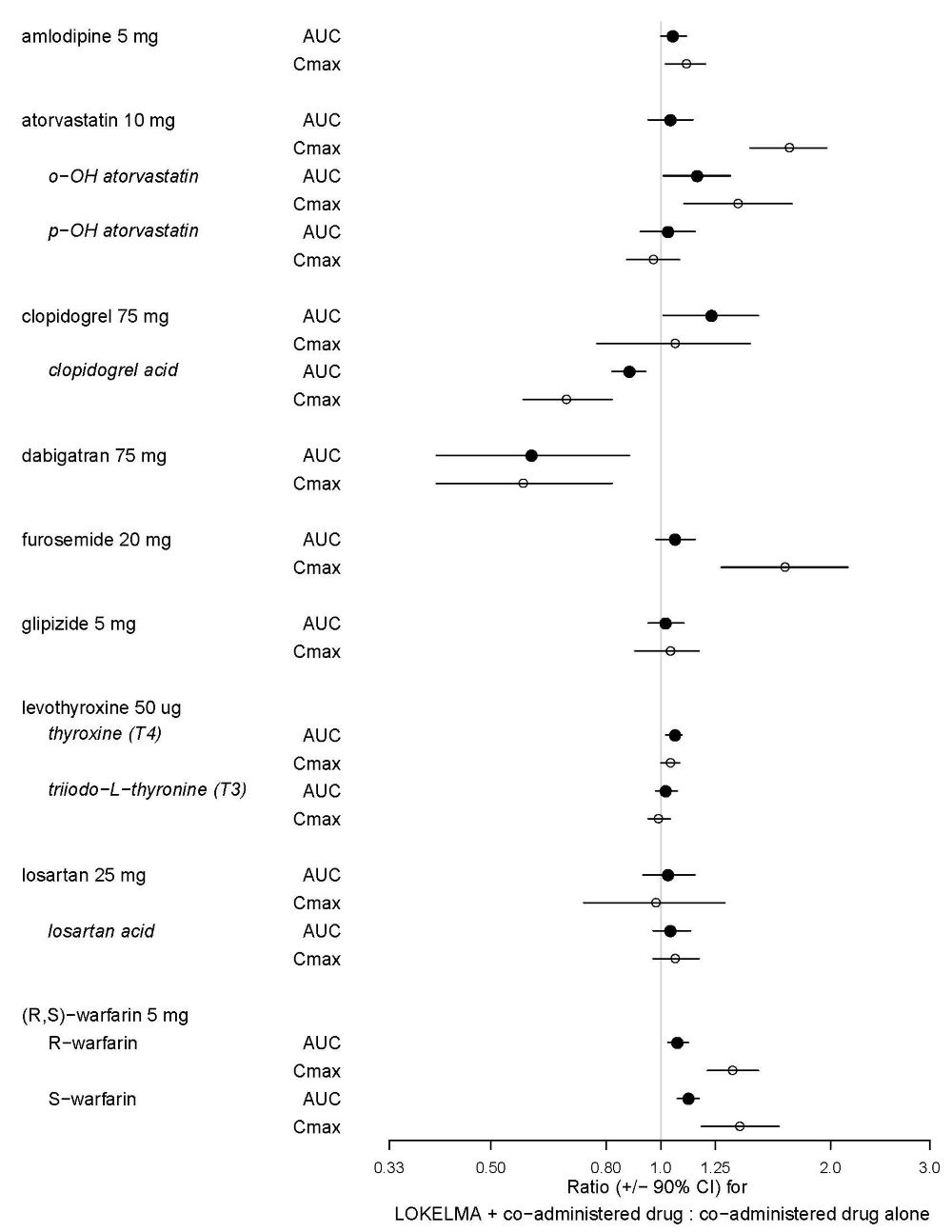
Nine (9) of the 20 drugs that showed an in vitro interaction were subsequently tested in vivo in healthy volunteers. Losartan, glipizide and levothyroxine did not show any changes in exposure when co-administered with LOKELMA. However, there was an increase in systemic exposure to weak acids such as furosemide and atorvastatin, and a decrease in systemic exposure to weak bases such as dabigatran when co-administered with LOKELMA, as shown in Figure 2. These changes are consistent with the hypothesis that LOKELMA, by elevating gastric pH, affects the systemic exposure of co-administered drugs whose solubility is pH-dependent [see Drug Interactions (7)].
Figure 2. Effects of LOKELMA on the Pharmacokinetic Exposures of Other Orally Administered Medications:
13.1. Carcinogenesis, Mutagenesis, Impairment of Fertility
The following tests for mutagenic potential of sodium zirconium cyclosilicate were negative: (1) the Ames (S. typhimurium and E. coli) test; (2) chromosomal aberration assay in Chinese Hamster Ovary (CHO) cells; and (3) in vivo rat micronucleus assay. Given that zirconium cyclosilicate is not genotoxic, not absorbed from the gastrointestinal tract, and did not cause local gastrointestinal alterations in a chronic toxicity study in dogs, carcinogenicity studies in animals to evaluate tumorigenic potential of sodium zirconium cyclosilicate were not deemed to be necessary.
Fertility in male and female rats has been assessed at doses up to a Human Equivalent Dose (HED) of 58 g per day (the maximum feasible dose) with no adverse effects.
14. Clinical Studies
14.1 Study 1
The effectiveness of LOKELMA in lowering serum potassium was demonstrated in a two-part, double-blind, randomized, placebo-controlled clinical trial (NCT01737697) in patients with hyperkalemia (5 to 6.5 mEq/L, mean potassium 5.3 mEq/L), Study 1.
In the first phase of the trial (the acute phase), 753 patients were randomized to receive one of four doses of LOKELMA (1.25, 2.5, 5 or 10 g) or placebo, administered three times daily for the initial 48 hours with meals.
The mean age of patients was 66 years, 59% of patients were men, and 86% were Caucasian. Approximately 60% of patients had chronic kidney disease, 10% had heart failure, 62% had diabetes mellitus and 67% were on renin angiotensin aldosterone system (RAAS) inhibitor therapy at baseline.
The primary endpoint in the acute phase was the difference in the exponential rate of change in serum potassium levels during the initial 48 hours of study drug treatment, comparing placebo-treated patients and LOKELMA-treated patients. The study met its primary endpoint demonstrating a greater reduction in serum potassium levels for the 2.5, 5 and 10 g (three times a day) dose groups compared to the placebo group (p<0.001). As displayed in Table 1 for the secondary endpoint of potassium change from baseline, LOKELMA showed dose-dependent reductions in serum potassium at 2.5, 5 and 10 g. In patients administered 10 g TID, the mean serum potassium reduction was -0.7 mEq/L at 48 hours. Patients with higher starting potassium levels had a greater response to LOKELMA. LOKELMA was effective in lowering potassium levels in patients with chronic kidney disease, heart failure, diabetes mellitus and those taking RAAS inhibitor therapy.
Table 1. Study 1 - Potassium Change from Baseline to 48 hours:
| Mean Serum Potassium Change mEq/L (95% Confidence Intervals) Sample Size | Placebo | 1.25 g TID | 2.5 g TID | 5 g TID | 10 g TID |
|---|---|---|---|---|---|
| All Patients | -0.2 (-0.3, -0.2) n=158 | -0.3 (-0.4, -0.2) n=150 | -0.5 (-0.5, -0.4) n=137 | -0.5 (-0.6, -0.5) n=152 | -0.7 (-0.8, -0.7) n=140 |
| Baseline Serum Potassium >5.5 mEq/L | -0.4 (-0.6, -0.3) n=40 | -0.3 (-0.5, -0.2) n=40 | -0.6 (-0.7, -0.4) n=37 | -0.9 (-1.0, -0.7) n=29 | -1.1 (-1.3, -0.9) n=22 |
Patients who achieved a potassium level between 3.5 and 5 mEq/L after receiving LOKELMA during the acute phase were re-randomized to receive once daily placebo or 1.25, 2.5, 5 or 10 g of once daily LOKELMA for 12 days together with breakfast.
The primary endpoint in the maintenance phase was the difference in the exponential rate of change in serum potassium levels over the 12-day treatment interval, comparing patients receiving LOKELMA and patients receiving placebo. The study met the primary efficacy endpoint at the 5 and 10 g doses when compared with their respective placebo groups (p<0.01 and p<0.001).
14.2 Study 2
The efficacy of LOKELMA was also demonstrated in a two-part trial with an open-label acute phase and a month-long randomized, double-blind, placebo-controlled withdrawal phase (Study 2; NCT02088073).
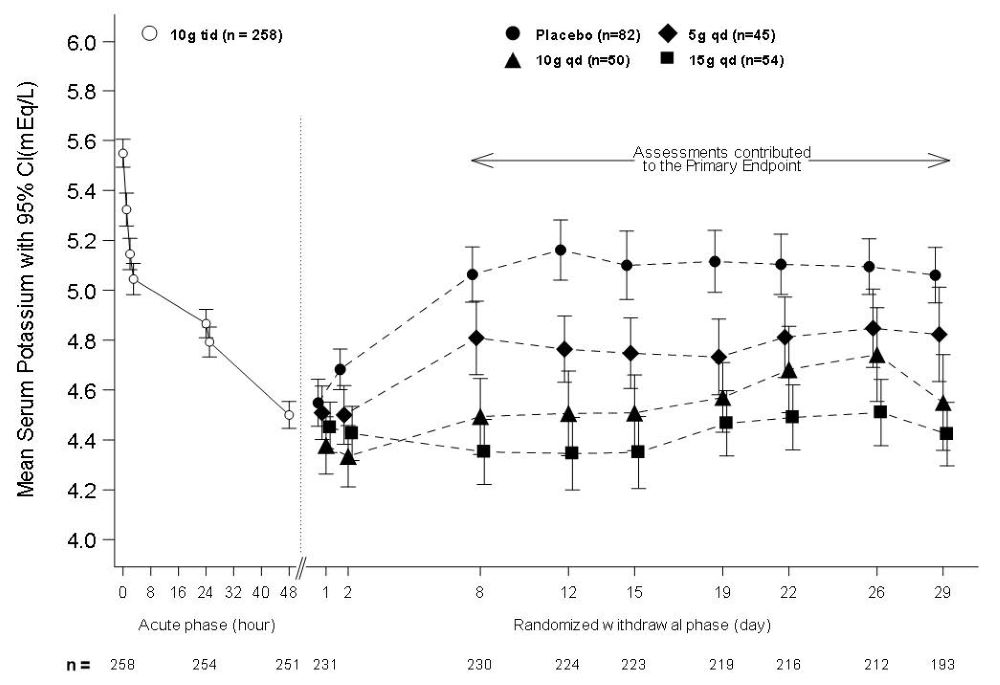
In the open-label acute phase of Study 2, 258 patients with hyperkalemia (baseline mean 5.6 mEq/L, range 5.1 to 7.4 mEq/L) received 10 g of LOKELMA administered three times daily with meals for 48 hours. As shown in Figure 3, left, average serum potassium levels decreased from 5.6 to 4.5 mEq/L during treatment with LOKELMA in the acute phase.
Following the acute phase of the study, there was a double-blind randomized withdrawal phase where patients who achieved potassium levels between 3.5 and 5 mEq/L were randomized to one of three doses of LOKELMA administered once-daily for 28 days, or placebo just before breakfast. Of the patients enrolled in the acute phase, 92% achieved a potassium level within this range and were enrolled into the second phase of the trial.
The primary endpoint in the randomized withdrawal phase was the mean serum potassium value over the period from Day 8 to Day 29, comparing LOKELMA-treated and placebo-treated patients. All three doses (5, 10 and 15 g) of once daily LOKELMA maintained mean potassium at lower levels than placebo (mean serum potassium was 4.8, 4.5, and 4.4 mEq/L for the 5, 10 and 15 g dose groups, respectively, vs. 5.1 mEq/L in the placebo group, p≤0.001 for all doses, Figure 3, right). A greater proportion of patients had mean serum potassium levels in the normal range (3.5 to 5 mEq/L) while on LOKELMA than while on placebo (80%, 90% and 94% at the 5, 10 and 15 g doses, respectively, vs. 46% on placebo).
Figure 3. Study 2 - Mean Serum Potassium Levels in the Acute and Randomized Withdrawal Phases:
Intent-to-Treat population includes subjects with at least one valid serum potassium measurement on or after Day 8
14.3 Eleven-Month Extension Study
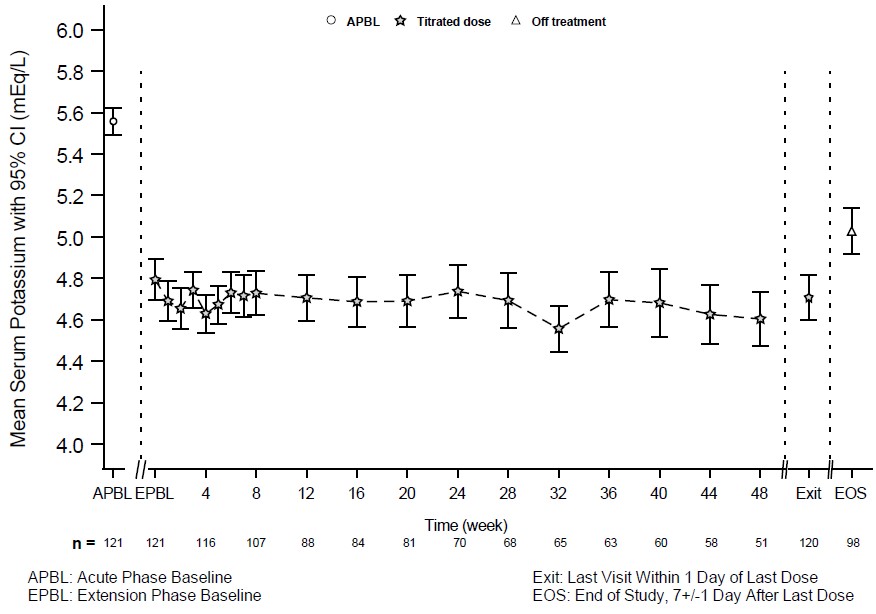
Patients who completed the 28-day randomized withdrawal phase had the option to continue treatment with LOKELMA, taken just before breakfast, in an open-label extension phase for up to 11 months (n=123; NCT02107092). Figure 4 shows that the treatment effect on serum potassium was maintained during continued therapy.
Figure 4. 11-Month Open-Label Extension Phase of Study 2 - Mean Serum Potassium (mEq/L):
14.4 Study 3
LOKELMA was evaluated in an open-label 12-month study in 751 hyperkalemic patients (NCT02163499). The mean baseline potassium level in this study was 5.6 mEq/L. Following the acute phase treatment of LOKELMA 10 g three times a day, patients who achieved normokalemia (3.5-5.0 mEq/L) within 72 hours (n=746; 99%) entered the maintenance phase. For maintenance treatment, the initial dosage of LOKELMA was 5 g once daily and was adjusted to a minimum of 5 g every other day up to maximum of 15 g once daily, based on serum potassium level. The treatment effect on serum potassium was maintained during continued therapy.
14.5 Study 4
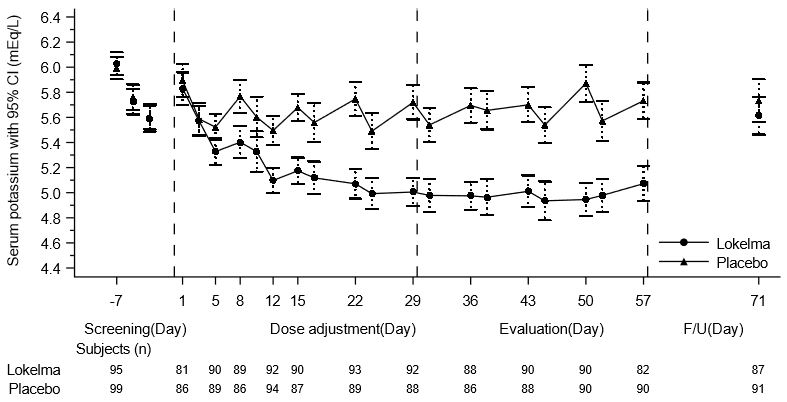
The effectiveness of LOKELMA in lowering serum potassium was studied in a double-blind, placebo-controlled trial of 196 chronic hemodialysis patients (mean age 58 years, range 20 to 86 years) with persistent pre-dialysis hyperkalemia (mean baseline potassium 5.8 mEq/L) who were randomized to receive LOKELMA 5 g or placebo once daily on non-dialysis days (NCT03303521). During the dose adjustment period (initial 4 weeks), the dose was adjusted weekly in 5 g increments up to 15 g once daily based on pre-dialysis serum potassium measurement after the long inter-dialytic interval to achieve a pre-dialysis serum potassium level between 4.0-5.0 mEq/L. The dose reached at the end of the dose-adjustment period was maintained throughout the subsequent 4-week evaluation period.
The primary endpoint in the trial was the proportion of responders, defined as patients who maintained a pre-dialysis serum potassium between 4.0 and 5.0 mEq/L on at least 3 out of 4 dialysis treatments after the long inter-dialytic interval and who did not receive rescue therapy during the evaluation period. A greater proportion of patients were responders in the LOKELMA arm as compared to placebo (41% vs 1%, respectively; p<0.001). The treatment effect on mean pre-dialysis serum potassium levels was maintained during continued treatment. Mean pre-dialysis serum potassium levels during the study are presented in Figure 5.
Figure 5. Mean Pre-Dialysis Serum Potassium Levels Over Time in Patients on Chronic Hemodialysis:
F/U - follow-up period
The displayed error bars correspond to 95% confidence intervals.
n = Number of patients with non-missing potassium measurements at a particular visit.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.