LOQTORZI Concentrate for solution for infusion Ref.[115078] Active ingredients: Toripalimab
Source: European Medicines Agency (EU) Revision Year: 2025 Publisher: Topalliance Biosciences Europe Limited, Ground Floor, Two Dockland Central, Guild Street, I.f.s.c., Dublin 1, Co. Dublin, D01 K2C5, Ireland
5.1. Pharmacodynamic properties
Pharmacotherapeutic group: Antineoplastic agents
ATC code: L01FF13
Mechanism of action
Toripalimab is a humanised IgG4 monoclonal antibody that binds to the PD-1 receptor and blocks its interaction with PD-L1 and PD-L2, releasing PD-1 pathway-mediated inhibition of the immune response, including the anti-tumour immune response. Binding of the PD-1 ligands, PD-L1 and PD-L2, to the PD-1 receptor found on T cells, inhibits T cell proliferation, cytokine production, and cytotoxic activity.
Clinical efficacy and safety
Nasopharyngeal carcinoma
The efficacy of toripalimab in combination with cisplatin and gemcitabine was investigated in JUPITER-02, a randomised, multi-centre, double-blind, placebo-controlled study in 289 patients with metastatic or recurrent, locally advanced nasopharyngeal carcinoma (NPC) not amenable to curative therapy who had not received previous systemic chemotherapy for recurrent or metastatic disease. Patients with recurrent NPC after treatment with curative intent were required to have an interval of at least 6 months between the last dose of radiotherapy or chemotherapy and recurrence. Patients with autoimmune disease, other than stable hypothyroidism or Type I diabetes, and patients who required systemic immunosuppression were ineligible.
Randomisation was stratified according to ECOG PS (0 versus 1) and disease stage (recurrent versus metastatic) at study entry. Patients were randomised (1:1) to receive one of the following treatments:
- Toripalimab 240 mg intravenously on Day 1 in combination with cisplatin 80 mg/m² on Day 1 and gemcitabine 1 000 mg/m² on Days 1 and 8 every 3 weeks for up to 6 cycles, followed by toripalimab 240 mg once every 3 weeks, or
- Placebo intravenously on Day 1 in combination with cisplatin 80 mg/m² on Day 1 and gemcitabine 1 000 mg/m² on Days 1 and 8 every 3 weeks for up to 6 cycles, followed by placebo once every 3 weeks.
Treatment with toripalimab or placebo continued until disease progression per response evaluation criteria in solid tumours (RECIST) v1.1 (with the exception noted below), unacceptable toxicity, or a maximum of 2 years. Administration of toripalimab was permitted beyond radiographic progression if the patient was deriving benefit as assessed by the investigator. Tumour assessments were performed every 6 weeks for the first 12 months and every 9 weeks thereafter. The main efficacy outcome measure was Blinded Independent Review Committee (BIRC)-assessed progression-free survival (PFS) according to RECIST v1.1.
The study population characteristics were: median age of 48 years (range: 19 to 72), 4.8% age 65 or older, 83% male, 100% Asian, and ECOG PS of 0 (57%) or 1 (43%). Approximately 86% of the study population had metastatic disease at randomisation, with histological subtypes of NPC including 98% non-keratinizing, 1% keratinizing squamous cell carcinoma, and 1% unclassified NPC/other. The majority (63%) of patients had serum Epstein-Barr virus (EBV) titres ≥2000 U/mL.
The study showed statistically significant improvements in BIRC-assessed PFS and OS for patients randomised to toripalimab in combination with cisplatin/gemcitabine compared to cisplatin and gemcitabine with placebo.
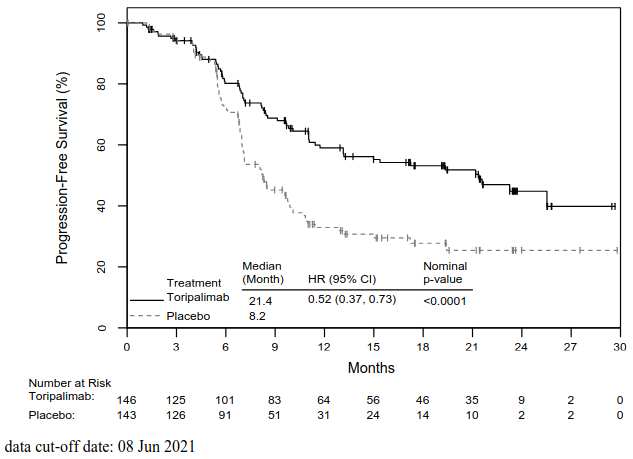
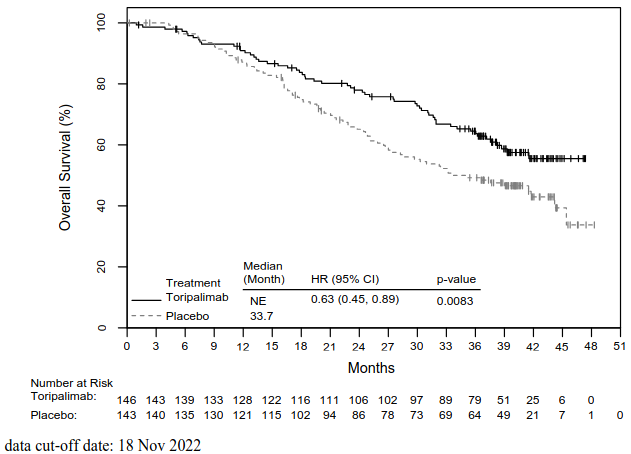
Efficacy results are summarised in Table 3, Figure 1 and Figure 2 below.
Table 3. Efficacy results in JUPITER-02:
| Endpoints1 | Toripalimab + cisplatin/ gemcitabine N=146 | Placebo + cisplatin/ gemcitabine N=143 |
|---|---|---|
| BIRC-assessed progression-free survival (PFS) | ||
| Number of PFS events (%) | 63 (43.2) | 87 (60.8) |
| Median PFS, months (95% CI) | 21.4 (11.7, NE) | 8.2 (7.0, 9.8) |
| Hazard ratio (95% CI)2 | 0.52 (0.37, 0.73) | |
| Nominal p-value3 | <0.0001 | |
| Overall survival (OS) | ||
| Number of deaths (%) | 57 (39.0) | 76 (53.1) |
| Median OS, in months (95% CI) | NE (38.7, NE) | 33.7 (27.0, 44.2) |
| Hazard ratio (95% CI)2 | 0.63 (0.45, 0.89) | |
| p-value3 | 0.0083 | |
1 The final analysis of PFS was based on the data with cut-off date of 08 Jun 2021 and the final analysis of OS was based on the data with cut-off date of 18 Nov 2022.
2 The hazard ratio and its confidence interval were computed using a stratified Cox proportional-hazards model.
3 Two-sided p-value, based on stratified log-rank test.
BIRC = blinded independent review committee; CI = confidence interval; NE = Not estimable
Figure 1. Kaplan-Meier curves for BIRC-assessed PFS in JUPITER-02:
Figure 2. Kaplan-Meier curves for overall survival in JUPITER-02:
In exploratory subgroup analyses of PFS and OS, the magnitude of the treatment effects appeared similar across patient subgroups based on PD-L1 expression or EBV titres.
Elderly population
A minority of patients (4.8%; 14/289) were age ≥65 years. Data are too limited to draw conclusions on this population.
Oesophageal squamous cell carcinoma
The efficacy of toripalimab in combination with paclitaxel and cisplatin was investigated in JUPITER-06, a randomised, multi-centre, single region, double-blind, placebo-controlled study in 514 patients with metastatic or recurrent, locally advanced oesophageal squamous cell carcinoma (OSCC) who had not received previous systemic chemotherapy for recurrent or metastatic disease. Patients with recurrent OSCC after treatment with curative intent were required to have an interval of at least 6 months between the last dose of adjuvant, neoadjuvant chemotherapy, radiation, or chemoradiotherapy and recurrence or at least 12 months between the last dose of adjuvant chemotherapy/chemoradiotherapy with paclitaxel and cisplatin. Patients with autoimmune disease, other than stable hypothyroidism or Type I diabetes, and patients who required systemic immunosuppression were ineligible.
Randomisation was stratified according to ECOG PS (0 versus 1) and previous radiotherapy (yes versus no). Patients were randomized (1:1) to receive one of the following treatments:
- Toripalimab 240 mg intravenously in combination with paclitaxel 175 mg/m² intravenously and cisplatin 75 mg/m² intravenously on Day 1 every 3 weeks for 4 to 6 cycles, followed by toripalimab 240 mg once every 3 weeks, or
- Placebo intravenously in combination with paclitaxel 175 mg/ m² intravenously and cisplatin 75 mg/m² intravenously on Day 1 every 3 weeks for 4 to 6 cycles, followed by placebo once every 3 weeks.
Treatment with toripalimab or placebo continued until disease progression per RECIST v1.1, unacceptable toxicity (with the exception noted below), or a maximum of 2 years. Tumour assessments were performed every 6 weeks for the first 12 months and every 9 weeks thereafter. The co-primary endpoints were Blinded Independent Review Committee (BIRC)-assessed progression-free survival (PFS) according to RECIST v1.1 and OS.
The study population characteristics were: median age of 63 years (range: 20 to 75), 38% age 65 or older, 85% male, 100% Asian, and ECOG PS of 0 (26%) or 1 (74%). Seventy-nine percent of patients had metastatic disease at study entry.
The results of the final analysis of BIRC-determined PFS showed a statistically significant improvement in PFS. At the final analysis of OS (data cut-off 23 Feb 2023), the study showed consistent improvement in OS (HR 0.72; 95% CI 0.58-0.88).
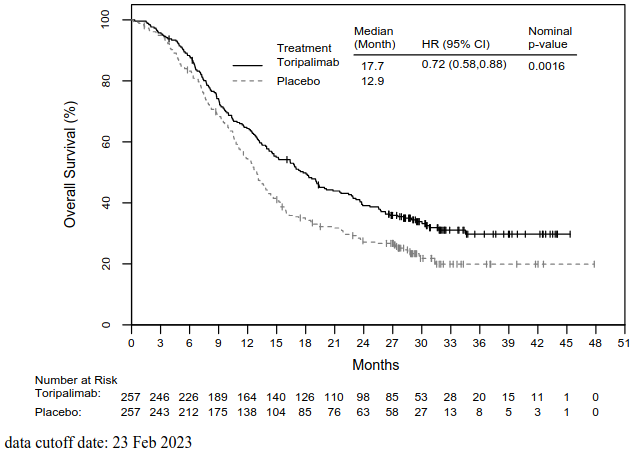
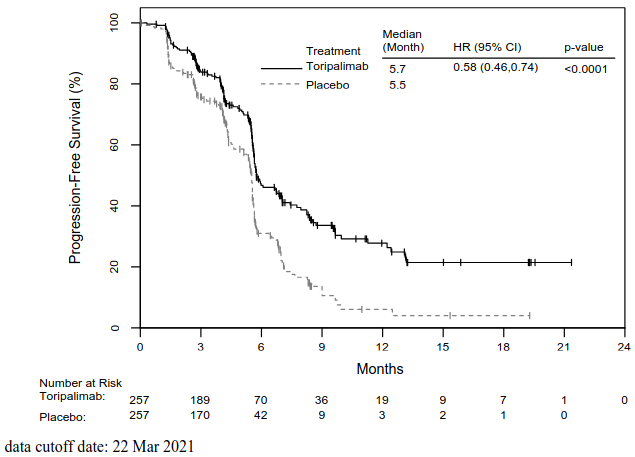
Efficacy results of OS and BIRC-determined PFS are summarised in Table 4, Figure 3 and Figure 4 below.
Table 4. Efficacy results in JUPITER-06:
| Toripalimab + paclitaxel/cisplatin N=257 | Placebo + paclitaxel/cisplatin N=257 | |
|---|---|---|
| Overall survival (OS)1 | ||
| Number of OS events (%) | 172 (66.9) | 195 (75.9) |
| Median OS, months (95% CI) | 17.7 (14.6, 20.8) | 12.9 (11.6, 14.1) |
| Hazard ratio (95% CI)2 | 0.72 (0.58, 0.88) | |
| p-value3 | 0.0016 | |
| BIRC-assessed progression-free survival (PFS)4 | ||
| Number of PFS event (%) | 132 (51.4) | 164 (63.8) |
| Median PFS, months (95% CI) | 5.7 (5.6, 7.0) | 5.5 (5.2, 5.6) |
| Hazard ratio 2 (95% CI) | 0.58 (0.46, 0.74) | |
| p-value3 | <0.0001 | |
1 The data cut-off for the final analysis of OS was 23 Feb 2023.
2 The hazard ratio and its confidence interval were computed using the stratified Cox proportional-hazards model.
3 Two-sided p-value, based on the stratified log-rank test.
4 The data cut-off for the final analysis of PFS was 22 Mar 2021.
BIRC = blinded independent review committee; CI = confidence interval
Figure 3. Kaplan-Meier curves for overall survival in JUPITER-06:
Figure 4. Kaplan-Meier curves for BIRC-assessed PFS in JUPITER-06:
Efficacy and PD-L1 status
In exploratory subgroup analyses of PFS and OS, the magnitude of the treatment effects appeared similar across patient subgroups based on PD-L1 expression.
Elderly population
There were 195 patients (38%) who were age 65 years or older. No overall differences in efficacy were observed between patients ≥65 years of age and younger patients receiving toripalimab in combination with paclitaxel/cisplatin.
Paediatric population
The European Medicines Agency has waived the obligation to submit the results of studies with LOQTORZI in all subsets of the paediatric population in the treatment of all conditions in the category of malignant neoplasms (except CNS, haematopoietic and lymphoid tissue and melanoma) (see section 4.2 for information on paediatric use).
5.2. Pharmacokinetic properties
Toripalimab pharmacokinetics were characterised using population PK analyses that included data from 574 patients across 5 clinical studies with various solid tumours who received fixed (80 to 480 mg Q2W or Q3W) or weight-based (range: 1 to 10 mg/kg Q2W) dosing, including 92 patients with NPC and 236 patients with OSCC who received toripalimab at doses of 240 mg every 3 weeks in JUPITER-02 and JUPITER-06, respectively.
Toripalimab pharmacokinetic parameters are presented as geometric mean (coefficient of variation [CV]%) unless otherwise noted.
Absorption
Toripalimab is administered via the intravenous route; therefore, it is completely bioavailable.
Distribution
Toripalimab is primarily distributed in the plasma with a geometric mean volume of distribution at steady state of approximately 3.8 L (CV=27.4%).
Biotransformation
Dedicated metabolism studies were not performed. As a monoclonal antibody, toripalimab is expected to be metabolized into small peptides, amino acids, and small carbohydrates by catabolic pathways or by receptor-mediated endocytosis. The degradation products are eliminated by renal excretion or returned to the nutrient pool without biological effects.
Elimination
Toripalimab pharmacokinetics followed a 2-compartment model with time-varying clearance (CL). The mean CL was 12.01 mL/h (CV=27%) after the first dose and 8.49 mL/h (CV=24.4%) at steady state. The geometric mean value (CV%) for the terminal half-life is 14 days (32.5%) at steady-state with toripalimab administered at 240 mg Q3W.
Linearity/non-linearity
Exposure to toripalimab, as expressed by peak concentrations (Cmax), increased dose proportionally over the dose range of 80 to 480 mg Q2W. The geometric mean trough concentrations (Cmin) at steady state were estimated in the population PK model to be 26.3 μg/mL in patients receiving 240 mg every 3 weeks. The mean accumulation of Cmin at steady state is 2.7-fold compared to the Cmin after the first dose.
Pharmacokinetic/pharmacodynamic relationship(s)
Toripalimab exposure-response relationships for efficacy are essentially flat over the range of exposures achieved for nasopharyngeal carcinoma in JUPITER-02 and for OSCC in JUPITER-06. The toripalimab exposure-response relationships for safety showed negative (inverse) relationships over the range of exposures achieved; however, this is likely an artifact reflecting toripalimab accumulation.
Anticipated full receptor occupancy of PD-1 in immune cells was achieved at exposures below mean trough concentrations after the first dose and steady state at dose of 240 mg Q3W.
Special populations
No clinically significant differences in the pharmacokinetics of toripalimab were observed based on age (range: 19 to 85 years), body weight (range: 39 to 164 kg), sex, concomitant chemotherapy, mild or moderate renal impairment, mild hepatic impairment, tumour burden and primary cancer.
Renal impairment
The effect of renal impairment based on the estimated creatinine clearance on the clearance and volume of distribution of toripalimab were evaluated using population pharmacokinetic analyses. No differences in clearance or volume of distribution were found between patients with mild (CLcr 60 to 89 mL/min; n=483) or moderate (CLcr 30 to 59 mL/min; n=114) renal impairment and patients with normal renal function. The effect of severe (CLcr 15 to 29 mL/min) renal impairment on the pharmacokinetics of toripalimab has not been studied.
Hepatic impairment
The effects of hepatic impairment using the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) grading system for hepatic dysfunction on the clearance and volume of distribution of toripalimab were evaluated using population pharmacokinetic analyses. No differences in clearance or volume of distribution were found between patients with mild (Grade 1, n=166) hepatic impairment (total bilirubin up to 1.5 times the upper limit of normal (ULN) or total bilirubin within normal limits and aspartate transaminase (AST) or alanine transaminase (ALT) >1 and ≤3 ULN) compared to patients with normal liver function. There was a limited number of patients with moderate (Grade 2, n=1; total bilirubin >1.5 to 3 times ULN and any AST) hepatic impairment and no patients with severe (Grade 3; total bilirubin >3 times ULN and any AST) hepatic impairment enrolled in clinical studies of toripalimab.
5.3. Preclinical safety data
No studies have been performed to test the potential of toripalimab for carcinogenicity or genotoxicity.
Animal reproduction studies have not been conducted with toripalimab to evaluate its effect on reproduction and foetal development. A central function of the PD-1/PD-L1 pathway is to preserve pregnancy by maintaining maternal immune tolerance to the foetus. In murine models of pregnancy, blockade of PD-L1 signalling has been shown to disrupt tolerance to the foetus and to result in an increase in foetal loss.
Fertility studies have not been conducted with toripalimab. In 4-week and 26-week repeat-dose toxicology studies in cynomolgus monkeys, there were no adverse or notable effects in the male and female reproductive organs. However, those animals were unlikely sexually mature.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.