OJEMDA Powder for oral suspension Ref.[116575] Active ingredients: Tovorafenib
Source: European Medicines Agency (EU) Revision Year: 2026 Publisher: Ipsen Pharma, 70 rue Balard, 75015 Paris, France
4.1. Therapeutic indications
Ojemda is indicated as monotherapy for the treatment of patients 6 months of age and older with paediatric low-grade glioma (LGG) harbouring a BRAF fusion or rearrangement, or BRAF V600 mutation, who have progressed after one or more prior systemic therapies (for biomarkers-based patient selection, see section 4.2).
4.2. Posology and method of administration
Treatment with tovorafenib should be initiated and supervised by a qualified physician experienced in the use of anti-cancer medicinal products.
Patient selection
Before taking tovorafenib, patients must have confirmation of BRAF fusion or rearrangement, or BRAF V600 mutation assessed by a CE-marked in vitro diagnostic (IVD) medical device with the corresponding intended purpose. If the CE-marked IVD is not available, confirmation of BRAF fusion or rearrangement, or BRAF V600 mutation should be assessed by an alternative validated test.
Posology
The recommended dose of tovorafenib based on body surface area (BSA) is 380 mg/m² once weekly. The maximum recommended dose is 600 mg once weekly (see Table 1).
Ojemda may be administered as an oral suspension (see Table 1) or as an immediate release tablet (see tovorafenib 100 mg film-coated tablets SmPC).
A recommended dose for patients with BSA less than 0.3 m² has not been established.
Table 1. Recommended dose based on body surface area:
| Body surface area | Dose volume* | Recommended dose (once weekly) |
| 0.30-0.35 m² | 5 ml | 125 mg |
| 0.36-0.42 m² | 6 ml | 150 mg |
| 0.43-0.48 m² | 7 ml | 175 mg |
| 0.49-0.54 m² | 8 ml | 200 mg |
| 0.55-0.63 m² | 9 ml | 225 mg |
| 0.64-0.77 m² | 11 ml | 275 mg |
| 0.78-0.83 m² | 12 ml | 300 mg |
| 0.84-0.89 m² | 14 ml | 350 mg |
| 0.90-1.05 m² | 15 ml | 375 mg |
| 1.06-1.25 m² | 18 ml | 450 mg |
| 1.26-1.39 m² | 21 ml | 525 mg |
| ≥1.40 m² | 24 ml | 600 mg |
* The maximum dose per bottle is 300 mg (12 ml).
Duration of treatment
Treatment with tovorafenib should be continued once weekly until disease progression, loss of clinical benefit, or unacceptable toxicity.
Missed or delayed doses
If a dose is missed by 3 days or less, the missed dose should be taken as soon as possible, and the next dose should be taken on its regularly scheduled day.
If a dose is missed by more than 3 days, the missed dose should be skipped, and the next dose should be taken on its regularly scheduled day.
A minimum of 4 days should occur between doses.
Vomiting
If vomiting occurs immediately after taking a dose, the dose should be repeated.
Dose modifications
The management of adverse reactions may require dose reduction, treatment interruption or treatment discontinuation.
The recommended dose reductions for adverse reactions for tovorafenib oral suspension are provided in Table 2.
Table 2. Recommended dose reductions for adverse reactions:
| Body surface area | First dose reduction | Second dose reduction | ||
| Volume | Dose | Volume | Dose | |
| 0.30-0.35 m² | 4 ml | 100 mg | 3 ml | 75 mg |
| 0.36-0.42 m² | 5 ml | 125 mg | 4 ml | 100 mg |
| 0.43-0.48 m² | 6 ml | 150 mg | 5 ml | 125 mg |
| 0.49-0.54 m² | 7 ml | 175 mg | 6 ml | 150 mg |
| 0.55-0.63 m² | 8 ml | 200 mg | 6 ml | 150 mg |
| 0.64-0.77 m² | 9 ml | 225 mg | 8 ml | 200 mg |
| 0.78-0.83 m² | 10 ml | 250 mg | 8 ml | 200 mg |
| 0.84-0.89 m² | 12 ml | 300 mg | 10 ml | 250 mg |
| 0.90-1.05 m² | 13 ml | 325 mg | 11 ml | 275 mg |
| 1.06-1.25 m² | 15 ml | 375 mg | 13 ml | 325 mg |
| 1.26-1.39 m² | 18 ml | 450 mg | 15 ml | 375 mg |
| ≥1.40 m² | 20 ml | 500 mg | 16 ml | 400 mg |
The recommended dose modifications for adverse reactions for tovorafenib are in Table 3.
Table 3. Recommended dose modifications for adverse reactions:
| Severity of ADRa | Dose modificationb |
| Haemorrhage and intratumoural haemorrhage | |
| • Intolerable Grade 2 • Grade 3 | Withhold administration. - If improved to Grade 0-1, resume at reduced dose. - If not improved, consider permanent discontinuation. |
| • First occurrence of any Grade 4 | Withhold administration. - If improved to Grade 0-1, resume at reduced dose. - If not improved, consider permanent discontinuation. |
| • Recurrent Grade 4 | Permanent discontinuation. |
| Skin toxicity, including photosensitivity | |
| • Intolerable Grade 2 • Grade 3 or 4 | Withhold administration. - If improved to Grade 0‑1, resume at reduced dose. - If not improved, consider permanent discontinuation. |
| Liver related events | |
| • Grade 3 AST or ALT • Grade 3 bilirubin | Withhold administration. If improved to Grade ≤ 2 or baseline resume as follows: - If laboratory abnormality resolves within 8 days, resume at the same dose. - If laboratory abnormality does not resolve within 8 days, resume at lower dosage. |
| • First occurrence of any Grade 4 | Withhold administration. - If improved to Grade 0-1, resume at lower dosage. - If not improved, consider permanent discontinuation. |
| • Recurrent Grade 4 | Permanent discontinuation. |
| Other adverse reactions | |
| • Intolerable Grade 2 • Grade 3 | Withhold administration. - If improved to Grade 0-1, resume at reduced dose. - If not improved, consider permanent discontinuation. |
| • Grade 4 | Withhold administration. - If improved to Grade 0-1, resume at reduced dose. - If not improved, consider discontinuation. |
a National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) version 5.0.
b See Table 2 for recommended dose reductions.
Special populations
Hepatic impairment
No dose adjustment is recommended for patients with mildly abnormal liver function tests (defined as bilirubin ≤ upper limit of normal [ULN] and aspartate aminotransferase [AST] > ULN or bilirubin > 1x to 1.5x ULN and any AST). Tovorafenib has not been studied in patients with moderately abnormal liver function tests (defined as bilirubin > 1.5x to 3x ULN and any AST) or severely abnormal liver function tests (defined as bilirubin > 3x ULN and any AST) (see section 5.2). Patients with moderately or severely abnormal liver function tests should be monitored carefully when treated with tovorafenib.
Renal impairment
No dose adjustment is recommended for patients with mild-to-moderate (eGFR ≥30 ml/min/1.73 m² calculated by Schwartz equation or MDRD equation) renal impairment. Tovorafenib has not been studied in patients with severe (eGFR <30 ml/min/1.73 m²) renal impairment (see section 5.2).
Paediatric population
Tovorafenib paediatric clinical experience is limited, particularly in the specific age range 6 months to 2 years. The safety and efficacy of tovorafenib in children below 6 months of age have not been established. No data are available.
Method of administration
Ojemda is for oral use.
If the patient is unable to swallow and has a nasogastric tube in situ, the powder for oral suspension can be administered via the tube (see section 6.6).
Ojemda can be taken with or without food (see section 5.2) and should be taken at a regularly scheduled time once weekly.
Ojemda should be administered to paediatric patients under adult supervision.
Ojemda powder for oral suspension must be reconstituted prior to being administered (see section 6.6).
Prior to first time use of the oral suspension, caregivers (and if appropriate, patients) should be instructed on the proper preparation, dose, and administration of Ojemda.
Detailed instructions on the preparation and administration of the powder for oral suspension are given in section 6.6 and at the end of the package leaflet.
The powder for oral suspension and the film-coated tablets may be used interchangeably (see tovorafenib 100 mg film-coated tablets SmPC). For patients who are not able to swallow or with BSA less than 0.9 m² the oral suspension should be provided.
4.9. Overdose
There is no information on overdose with tovorafenib. If overdose occurs, tovorafenib should be withheld and the patient should be treated supportively with appropriate monitoring as necessary. Since tovorafenib is highly bound to plasma proteins, haemodialysis is likely to be ineffective in the treatment of overdose with tovorafenib.
6.3. Shelf life
Powder for oral suspension: 3 years.
Reconstituted oral suspension: 15 minutes.
6.4. Special precautions for storage
This medicinal product does not require any special storage conditions.
6.5. Nature and contents of container
30 ml clear Type III glass bottle with induction-seal and a white polypropylene cap.
Each pack contains one bottle, a 20 ml oral dosing syringe, and a bottle adaptor.
6.6. Special precautions for disposal and other handling
- The instructions for use should be read carefully each time before preparing a dose of Ojemda.
- The doctor or pharmacist should show the patient or the caregiver how to prepare, measure and give a dose of Ojemda correctly.
- The bottle is made of glass. This medicine should not be used if the bottle is broken or damaged, or if the safety seal under the cap is broken or missing.
- Only 14 ml of room temperature water should be used for preparing Ojemda.
- Only use up to 12 ml of Ojemda from each prepared bottle. If the prescribed dose is more than 12 ml (300 mg), split the dose as equally as possible between each prepared bottle (e.g., 6 ml and 7 ml for a 325 mg dose). Prepare the first bottle and administer dose prior to preparing the second bottle.
- Each dose must be given within 15 minutes after the medicine has been prepared.
Instruction for reconstitution of Ojemda powder for oral suspension
Note: if more than one bottle is needed for the prescribed dose, bottles should be constituted one by one. Split the dose as equally as possible between each prepared bottle.
This procedure should be performed on a clean and flat work surface with clean hands.
Step 1: Fill a cup half-way with room temperature water. Do not use cold water.
Step 2: Pull up on the plunger of the oral dosing syringe to draw water exactly until the 14 ml mark.
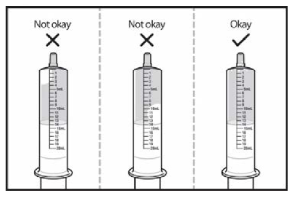
Step 3: Turn the oral dosing syringe tip upward and check for air bubbles. If large air bubbles appear in the oral dosing syringe, push the water back into the cup and then draw up the water again to the 14 ml mark. Repeat this step until there are no large air bubbles present. Small air bubbles are fine (see Figure 1)
Figure 1:
Step 4: Open the bottle with powder by pushing down firmly on the cap and turning it to the left (counterclockwise). Do not use the product if the bottle is broken, damaged or if the safety seal under the cap is broken or missing. Do not throw away the cap.
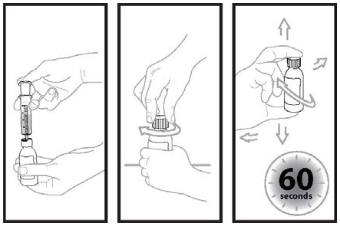
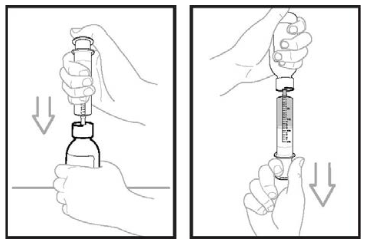
Step 5: Using the oral dosing syringe, inject exactly 14 ml of water into the bottle (see Figure 2). Right away, replace the cap back onto the bottle by pushing down while twisting the cap to the right (clockwise). Shake the bottle well for 60 seconds in all directions.
Turn the bottle upside down to check for any powder stuck to the inside of the bottle (see Figure 3). If you still see powder in the bottle, continue to shake the bottle for another 15 seconds until you no longer see the powder inside the bottle. Do not shake the bottle for more than 2 minutes total time. If you still see powder in the bottle, ask for a new bottle.
Figure 2:
Figure 3:
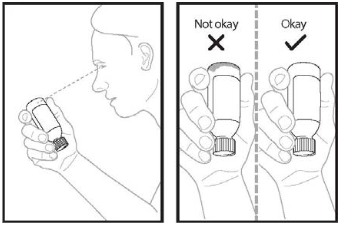
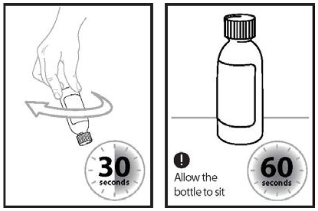
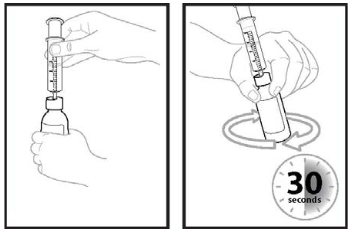
Step 6: Turn the bottle upside down again and swirl for 30 seconds (see Figure 4). Remove the cap and check that no solids are stuck in the bottle neck. If you see solids in the bottle neck when removing the cap, recap the bottle, turn the bottle upside down, and swirl for an additional 15 seconds.
Allow the bottle to sit for 60 seconds to allow most of the foam to settle. Note: Foaming in the bottle will reduce the amount of Ojemda for oral suspension.
Figure 4:
Step 7: Firmly insert the bottle adaptor into the bottle by pushing it tightly into the top of the bottle. The top edge of the bottle adaptor should be even with the bottle top.
Do not remove the bottle adaptor after it is inserted into the bottle.
Step 8: Check the prescribed dose in millilitres (ml). Draw air into the oral dosing syringe by pulling the plunger out until the prescribed dose is reached.
Step 9: Insert the tip of the oral dosing syringe into the bottle adaptor. The tip of the oral dosing syringe should fit snugly into the hole of the bottle adaptor. With the oral dosing syringe in place and holding the bottle where the oral dosing syringe tip inserts into bottle adaptor, swirl the oral suspension for 30 seconds (see Figure 5).
Figure 5:
Step 10: Inject the air from the oral dosing syringe into the bottle (see Figure 6). Hold the oral dosing syringe in place and turn the bottle upside down. To measure the prescribed dose, keep the tip of the oral dosing syringe facing up and pull down on the plunger until the top of the plunger lines up with the prescribed dose in millilitres.
Figure 6:
Step 11: While the syringe is still inserted into the adapter in the bottle, remove any air bubbles in the oral dosing syringe by gently pushing the Ojemda back into the bottle and then pulling down on the plunger again to draw up your prescribed dose.
Repeat this step until you see that few or no air bubbles remain or if you draw up the wrong dose in the oral dosing syringe. Only use up to 12 ml of Ojemda from each prepared bottle.
Step 12: Leave the tip of the oral dosing syringe in the bottle adaptor and carefully turn the bottle upright. Put the bottle onto a flat work surface again. Slowly remove the oral dosing syringe tip from the bottle adaptor by gently pulling straight up. Ojemda is ready for administration.
Administration using an oral syringe
Once the suspension is prepared, place the tip of the oral dosing syringe towards the inside of the mouth with the tip touching the inside of either cheek then slowly push the medicine into the mouth by pressing down on the plunger.
Do not forcefully push the plunger. This may cause choking. Allow the child to swallow while giving Ojemda.
Administration using a feeding tube
Only use a feeding tube with a minimum size of 12 French. Flush the feeding tube according to the manufacturer's instructions before administering the suspension. Use an ENFit syringe to draw up the suspension from the bottle then dispense the suspension into the feeding tube with an ENFit adaptor. Finally, flush the feeding tube after administering as per the manufacturer's instructions.
If 2 bottles are required to prepare the required dose, repeat Step 1 to 12 and give the remainder of the dose right away. Be sure to give the entire dose of Ojemda.
Any unused medicinal product or waste material should be disposed of in accordance with local requirements.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.