RADIOGENIX SYSTEM Technetium tc 99m generator injection Ref.[10398] Active ingredients: Technetium ⁹⁹ᵐTc pertechnetate
Source: FDA, National Drug Code (US) Revision Year: 2020
Product description
Chemical Characteristics
The RadioGenix System provides Sodium Pertechnetate Tc 99m Injection, USP for intravenous use, intravesicular use, ophthalmic use, or for preparing radiopharmaceutical kits. The RadioGenix System uses a non-uranium potassium molybdate Mo-99 source solution to produce Sodium Pertechnetate Tc 99m Injection, USP. The RadioGenix System uses potassium molybdate Mo-99 sources at an activity of 6 Ci/29 mL (222 GBq) at the date and time of calibration.
Elution of RadioGenix System produces Sodium Pertechnetate Tc-99m (Na99mTcO4) in approximately 5 mL of sterile 0.9% sodium chloride injection solution. The activity of Sodium Pertechnetate Tc-99m produced varies (30 mCi/mL to 1,153 mCi/mL of technetium Tc-99m) and depends on the activity of potassium molybdate Mo-99 present in the source container originally, the decay time since the calibration time, and the elapsed time since the previous Sodium Pertechnetate Tc-99m elution.
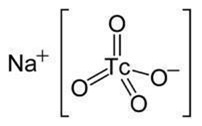
Sodium Pertechnetate Tc-99m is an inorganic compound with the formula Na99mTcO4. In solution, Sodium Pertechnetate exists as dissociated Na+ cations and pertechnetate TcO4-anions with the following molecular structure:
The eluted Sodium Pertechnetate Tc 99m Injection, USP is a sterile, non-pyrogenic, clear, and colorless solution. The pH of the solution is between 4.5 and 7.5.
Physical Characteristics
Technetium Tc-99m
Technetium Tc-99m decays by isomeric transition with a physical half-life of 6.01 hours. The principal photon that is useful for detection and imaging studies is shown in Table 8.
Table 8. Principal Radiation Emission Data technetium Tc-99m:
| Radiation | Mean Percent Per Disintegration | Energy (keV) |
|---|---|---|
| Gamma-2 | 88.5 | 140.5 |
The air-kerma-rate (exposure-rate) constant for technetium Tc-99m is 5.23 m²·pGy·(MBq)-1·s-1 [0.795 cm²·R·(mCi)-1·h-1]. A range of values for the relative radiation attenuation by the various thicknesses of Pb is shown in Table 9. For example, the use of 3 mm thickness of Pb will attenuate the radiation exposure by a factor of about 1,000.
Table 9. Radiation Attenuation by Lead Shielding:
| Shield Thickness (Pb) mm | Coefficient of Attenuation |
|---|---|
| 0.25 | 0.5 |
| 1 | 10-1 |
| 2 | 10-2 |
| 3 | 10-3 |
| 4 | 10-4 |
Molybdenum Mo-99
Molybdenum Mo-99 decays to technetium Tc-99m with a molybdenum Mo-99 half-life of 66 hours. This means that 77.7% of the activity remains after 24 hours; 60.4% remains after 48 hours, see Table 10.
Table 10. Molybdenum Mo-99 Decay Chart half-life 66.0 hours:
| Days | Percent Remaining | Days | Percent Remaining |
|---|---|---|---|
| 0* | 100 | 10 | 8 |
| 1 | 77.7 | 11 | 6.3 |
| 2 | 60.4 | 12 | 4.9 |
| 3 | 46.9 | 13 | 3.8 |
| 4 | 36.5 | 14 | 2.9 |
| 5 | 28.4 | 15 | 2.3 |
| 6 | 22.0 | 20 | 0.6 |
| 7 | 17.1 | 25 | 0.2 |
| 8 | 13.3 | 30 | 0.1 |
| 9 | 10.3 |
* calibration time
The physical decay characteristics of molybdenum Mo-99 are such that 88.6% of the decaying molybdenum Mo-99 atoms form Technetium Tc-99m. RadioGenix System elutions may be made at any time, but the amount of technetium Tc-99m available will depend on the time interval measured from the last elution cycle. Eluting the RadioGenix System every 24 hours will provide the maximal yield of Sodium Pertechnetate Tc-99m.
To correct for physical decay of technetium Tc-99m, the fractions that remain at selected intervals of time are shown in Table 11.
Table 11. Physical Decay Chart. Technetium Tc-99m, half-life 6.01 Hours:
| Hours | Percent Remaining | Hours | Percent Remaining |
|---|---|---|---|
| 0* | 100 | 7 | 44.7 |
| 1 | 89.1 | 8 | 39.8 |
| 2 | 79.4 | 9 | 35.5 |
| 3 | 70.8 | 10 | 31.6 |
| 4 | 63.1 | 11 | 28.2 |
| 5 | 56.2 | 12 | 25.1 |
| 6 | 50.1 |
* calibration time
RadioGenix System 1.2
Chemical Characteristics
The RadioGenix System provides Sodium Pertechnetate Tc 99m Injection, USP for intravenous use, intravesicular use, ophthalmic use, or for preparing radiopharmaceutical kits. The RadioGenix System uses a non-uranium potassium molybdate Mo-99 source solution to produce Sodium Pertechnetate Tc 99m Injection, USP. The RadioGenix System uses potassium molybdate Mo-99 sources at an activity of 6 Ci/29 mL (222 GBq) at the date and time of calibration.
Elution of RadioGenix System produces Sodium Pertechnetate Tc-99m (Na99mTcO4) in approximately 5 mL of sterile 0.9% sodium chloride injection solution. The activity of Sodium Pertechnetate Tc-99m produced varies (25 mCi/mL to 1,015 mCi/mL of technetium Tc-99m) and depends on the activity of potassium molybdate Mo-99 present in the source container originally, the decay time since the calibration time, and the elapsed time since the previous Sodium Pertechnetate Tc-99m elution.
Sodium Pertechnetate Tc-99m is an inorganic compound with the formula Na99mTcO4. In solution, Sodium Pertechnetate exists as dissociated Na+ cations and pertechnetate TcO4-anions with the following molecular structure:
?renderMultiMedia?
The eluted Sodium Pertechnetate Tc 99m Injection, USP is a sterile, non-pyrogenic, clear, and colorless solution. The pH of the solution is between 4.5 and 7.5.
Physical Characteristics
Technetium Tc-99m
Technetium Tc-99m decays by isomeric transition with a physical half-life of 6.01 hours. The principal photon that is useful for detection and imaging studies is shown in Table 8.
Table 8. Principal Radiation Emission Data technetium Tc-99m:
| Radiation | Mean Percent Per Disintegration | Energy (keV) |
|---|---|---|
| Gamma-2 | 88.5 | 140.5 |
The air-kerma-rate (exposure-rate) constant for technetium Tc-99m is 5.23 m²·pGy·(MBq)-1·s-1 [0.795 cm²·R·(mCi)-1·h-1]. A range of values for the relative radiation attenuation by the various thicknesses of Pb is shown in Table 9. For example, the use of 3 mm thickness of Pb will attenuate the radiation exposure by a factor of about 1,000.
Table 9. Radiation Attenuation by Lead Shielding:
| Shield Thickness (Pb) mm | Coefficient of Attenuation |
|---|---|
| 0.25 | 0.5 |
| 1 | 10 -1 |
| 2 | 10 -2 |
| 3 | 10 -3 |
| 4 | 10 -4 |
Molybdenum Mo-99
Molybdenum Mo-99 decays to technetium Tc-99m with a molybdenum Mo-99 half-life of 66 hours. This means that 77.7% of the activity remains after 24 hours; 60.4% remains after 48 hours, see Table 10.
Table 10. Molybdenum Mo-99 Decay Chart half- life 66.0 hours:
| Days | Percent Remaining | Days | Percent Remaining |
|---|---|---|---|
| 0* | 100 | 10 | 8 |
| 1 | 77.7 | 11 | 6.3 |
| 2 | 60.4 | 12 | 4.9 |
| 3 | 46.9 | 13 | 3.8 |
| 4 | 36.5 | 14 | 2.9 |
| 5 | 28.4 | 15 | 2.3 |
| 6 | 22.0 | 20 | 0.6 |
| 7 | 17.1 | 25 | 0.2 |
| 8 | 13.3 | 30 | 0.1 |
| 9 | 10.3 |
* calibration time
The physical decay characteristics of molybdenum Mo-99 are such that 88.6% of the decaying molybdenum Mo-99 atoms form Technetium Tc-99m. RadioGenix System elutions may be made at any time, but the amount of technetium Tc-99m available will depend on the time interval measured from the last elution cycle. Eluting the RadioGenix System every 24 hours will provide the maximal yield of Sodium Pertechnetate Tc-99m.
To correct for physical decay of technetium Tc-99m, the fractions that remain at selected intervals of time are shown in Table 11.
Table 11. Physical Decay Chart. Technetium Tc-99m, half-life6.01 Hours:
| Hours | Percent Remaining | Hours | Percent Remaining |
|---|---|---|---|
| 0* | 100 | 7 | 44.7 |
| 1 | 89.1 | 8 | 39.8 |
| 2 | 79.4 | 9 | 35.5 |
| 3 | 70.8 | 10 | 31.6 |
| 4 | 63.1 | 11 | 28.2 |
| 5 | 56.2 | 12 | 25.1 |
| 6 | 50.1 |
* calibration time
| Dosage Forms and Strengths |
|---|
|
The RadioGenix System provides Sodium Pertechnetate Tc 99m Injection, USP from a non-highly enriched uranium source of potassium molybdate Mo-99, as a clear, colorless solution containing 30 mCi/mL to 1,153 mCi/mL (1,110 to 42,661 MBq/mL) of technetium Tc-99m radioactivity in approximately 5 mL of volume. The amount of Tc-99m radioactivity depends on the radioactivity in the potassium molybdate Mo-99 source. The source is supplied in vessels containing 6 Ci (222 GBq) at the date and time of calibration. RadioGenix System 1.2The RadioGenix System provides Sodium Pertechnetate Tc 99m Injection, USP, from a non-highly enriched uranium source of potassium molybdate Mo-99, as a clear, colorless solution containing 25 mCi/mL to 1,015 mCi/mL (925 to 37,555 MBq/mL) of technetium Tc 99m radioactivity in approximately 5 mL of volume. The amount of Tc-99m radioactivity depends on the radioactivity in the potassium molybdate Mo-99 source. The source is supplied in vessels containing 6 Ci (222 GBq) at the date and time of calibration. |
| How Supplied | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
The RadioGenix System is a technetium tc-99m generator supplied and installed by NorthStar Medical Radioisotopes, LLC. It produces Sodium Pertechnetate Tc 99m Injection from a non-uranium potassium molybdate Mo-99 source solution. The potassium molybdate Mo-99 source solution is shielded within a source container which completely encases a vial that contains 29 mL of solution. NorthStar supplies potassium molybdate Mo-99 solution with the referenced calibration date and time specified on the container label (Table 12). Table 12. Potassium Molybdate Mo-99 Solution Containers:
The following kits (Tables 13-17) are used in the operation of the RadioGenix System as described in the RadioGenix System Operator Guide 1.0a and 1.1, (94S05058). Table 13. Materials Supplied in Source Vessel Kit for RadioGenix System, p/n 40P05047:
Table 14. Materials Supplied in Reagent Kit for RadioGenix System, p/n 40P05044:
Table 15. Materials Supplied in Tc-99m Product Kit for RadioGenix System, p/n 40P05045:
Table 16. Materials Supplied in Sterilization Kit for RadioGenix System, p/n 40P05043:
Table 17. Materials Supplied in Discarded Material Kit for RadioGenix System, p/n 40P05046:
RadioGenix System 1.2The RadioGenix System is a technetium Tc 99m generator supplied and installed by NorthStar Medical Radioisotopes, LLC. It produces Sodium Pertechnetate Tc 99m Injection, USP from a non-uranium potassium molybdate Mo-99 source solution. The potassium molybdate Mo-99 source solution is shielded within a source container which completely encases a vial that contains 29 mL of solution. NorthStar supplies potassium molybdate Mo-99 solution with the referenced calibration date and time specified on the container label (Table 12). Table 12. Potassium Molybdate Mo-99 Solution Containers:
The following kit and packs and consumables (Tables 13-22) are used in the operation of the RadioGenix System as described in the Operator Guide, RadioGenix System 1.2 (SYS-0028) or Operator Guide, RadioGenix System 1.2 (SYS-0060) P/N 40010570. Table 13. Materials Supplied in Source Vessel Kit for RadioGenix System, PN 40P07954:
Table 14. Materials Supplied in Primary Separation Cartridge (PSC) Pack for RadioGenix System 1.2, PN 40P09451:
Table 15. Materials Supplied in Elution Pack for RadioGenix System 1.2, PN 40P09452:
Table 16. Materials Supplied in Sterilization Pack for RadioGenix System 1.2, PN 40P09444:
Table 17. Materials Supplied in Discarded Material Pack for RadioGenix System 1.2, PN 40P09854:
Table 18. Materials Supplied in Discarded Material Type A Pack for RadioGenix System 1.2, PN 40P09855:
Table 19. Materials Supplied in Saline Tubing Pack for RadioGenix System 1.2, PN 40P09453:
The following consumables are shipped in bulk to the customer from NorthStar: Table 20. SWFI for RadioGenix System 1.2, PN 16C04488:
Table 21. NaOH for RadioGenix System 1.2, PN 16P09302:
Table 22. Saline for RadioGenix System 1.2, PN 16C09849:
* Indicates sterile components |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drugs
| Drug | Countries | |
|---|---|---|
| RADIOGENIX SYSTEM | United States |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.