SKILARENCE Gastro-resistant tablet Ref.[9768] Active ingredients: Dimethyl fumarate
Source: European Medicines Agency (EU) Revision Year: 2020 Publisher: Almirall, S.A., Ronda General Mitre, 151, 08022, Barcelona, Spain
Pharmacodynamic properties
Pharmacotherapeutic group: Other immunosuppressants
ATC code: L04AX07
Mechanism of action
The anti-inflammatory and immunomodulating effects of dimethyl fumarate and its metabolite monomethyl fumarate are not fully elucidated but are thought to be mainly due to the interaction with the intracellular reduced glutathione of cells directly involved in the pathogenesis of psoriasis. This interaction with glutathione leads to the inhibition of translocation into the nucleus and the transcriptional activity of the nuclear factor kappa-light-chain-enhancer of activated B-cells (NF-κB).
The main activity of dimethyl fumarate and monomethyl fumarate is considered to be immunomodulatory, resulting in a shift in T helper cells (Th) from the Th1 and Th17 profile to a Th2 phenotype. The inflammatory cytokine production is reduced with induction of proapoptotic events, inhibition of keratinocyte proliferation, reduced expression of adhesion molecules, and diminished inflammatory infiltrate within psoriatic plaques.
Clinical efficacy and safety
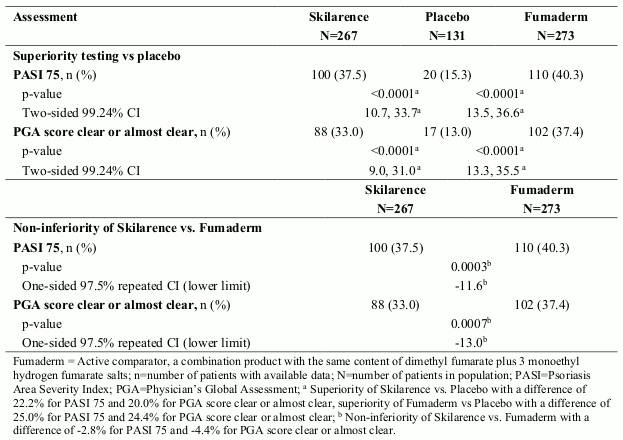
The safety and efficacy of Skilarence was assessed in one double-blind, 3-arm, placebo- and active comparator-controlled Phase III study (1102) in patients with moderate to severe plaque psoriasis (Study 1102). 704 patients were randomised to receive Skilarence, an active comparator (Fumaderm, a combination product with the same content of dimethyl fumarate plus 3 monoethyl fumarate salts) and placebo in a ratio of 2:2:1. Patients began treatment with tablets containing 30 mg/day dimethyl fumarate or placebo, titrating up to a maximum of 720 mg/day in both active treatment arms as described in section 4.2. If treatment success was observed before the maximum dose of 720 mg/day of dimethyl fumarate was reached, no further increase of dosage was necessary and the dosage was to be steadily reduced to an individual maintenance dose. In case of individual intolerability of the increased dosage during weeks 4 to 16, the patient was to return to the last tolerated dose taken since the start of week 4, which was to be maintained until end of the treatment period (week 16). Patients received treatment for up to 16 weeks and follow-up visits were planned for up to 12 months after treatment was stopped.
The demographic and baseline characteristics were well balanced between the treatment groups. Of the 699 patients, most were Caucasian (99%) and male (65%), and the mean age was 44 years. Most patients (91%) were <65 years of age. Most patients had moderate psoriasis based on Psoriasis Area and Severity Index (PASI) and Physician’s Global Assessment (PGA) scores at baseline: the mean PASI score at baseline was 16.35 and 60% of patients scored as moderate on the PGA. The majority of patients reported a “very large” or “extremely large” effect of psoriasis on their life based on the Dermatology Life Quality Index (DLQI), with a mean DLQI score of 11.5.
After 16 weeks of treatment, Skilarence was found to be superior to placebo (p<0.0001) based on PASI 75 and PGA score clear or almost clear and non-inferior (using a non-inferiority margin of -15%) to the active comparator (p<0.0003) based on PASI 75.
Summary of clinical efficacy after 16 weeks treatment in Study 1102:
There was a trend in the efficacy endpoint PASI score mean % change from baseline, indicating the onset of a clinical response to Skilarence as early as week 3 (-11.8%) which became statistically significant compared to placebo by week 8 (-30.9%). Further improvement was seen by week 16 (-50.8%).
The benefits of treatment with Skilarence were also supported by patient self-perceived improvements in their quality of life. At week 16, patients treated with Skilarence had a lower mean DLQI compared to placebo (5.4 vs 8.8).
Rebound (defined as worsening of ≥125% of baseline PASI value) was assessed after 2 months off treatment and was shown not to be a clinical concern with fumaric acid esters, as it was documented in very few patients (Skilarence 1.1% and active comparator 2.2%, compared to 9.3% in the placebo group).
Long-term efficacy data are currently not available for Skilarence, however, in the pharmacokinetic and clinical studies the systemic exposure, efficacy and safety of Skilarence were shown to be comparable to the active comparator containing dimethyl fumarate. Hence it is reasonable to expect the long-term efficacy of Skilarence to also be comparable to dimethyl fumarate-containing products. Maintenance of long term efficacy has been well described for other dimethyl fumarate-containing products, and therefore the treatment benefits seen with Skilarence at 16 weeks can be expected to be maintained in patients treated over the long term for at least 24 months.
Paediatric population
The European Medicines Agency has waived the obligation to submit the results of studies with Skilarence in all subsets of the paediatric population in this indication (see section 4.2 for information on paediatric use).
Pharmacokinetic properties
Absorption
After oral administration, dimethyl fumarate is not detected in plasma because it is rapidly hydrolysed by esterases to its active metabolite monomethyl fumarate. After oral administration of a single Skilarence 120 mg tablet in healthy subjects, monomethyl fumarate reached plasma peak concentrations of around 1325 ng/mL and 1311 ng/mL under fasted or fed conditions, respectively. Taking Skilarence with food delayed the tmax of monomethyl fumarate from 3.5 to 9.0 hours.
Distribution
The plasma protein binding of monomethyl fumarate is around 50%. Dimethyl fumarate does not show any binding affinity to serum proteins which may further contribute to its rapid elimination from the circulation. Biotransformation The biotransformation of dimethyl fumarate does not involve cytochrome P450 isoenzymes. In vitro studies have shown that monomethyl fumarate at the therapeutic dose does not inhibit or induce any of the cytochrome P450 enzymes, it is not a substrate or inhibitor of P-glycoprotein and is not an inhibitor of the most common efflux and uptake transporters. In vitro studies have shown that dimethyl fumarate at a therapeutic dose does not inhibit CYP3A4/5 and BCRP and is a weak P-glycoprotein inhibitor.
In vitro studies have shown that hydrolysis of dimethyl fumarate to monomethyl fumarate occurs rapidly at pH 8 (pH in the small intestine), but not at pH 1 (pH in the stomach). A part of the total dimethyl fumarate is hydrolysed by esterases and the alkaline milieu of the small intestine, while the remainder enters the portal vein blood. Further studies have shown that dimethyl fumarate (and to a lesser extent monomethyl fumarate) reacts partially with reduced glutathione forming a glutathioneadduct. These adducts were detected in animal studies in the intestinal mucosa of rats and to a smaller extent in portal vein blood. Unconjugated dimethyl fumarate, however, cannot be detected in the plasma of animals or psoriatic patients following oral administration. By contrast, unconjugated monomethyl fumarate is detectable in plasma. Further metabolism occurs through oxidation via the tricarboxylic acid cycle forming carbon dioxide and water.
Elimination
Exhalation of CO2 resulting from the metabolism of monomethyl fumarate is the primary route of elimination; only small amounts of intact monomethyl fumarate are excreted through urine or faeces. The portion of dimethyl fumarate that reacts with glutathione, forming a glutathione-adduct, is metabolised further to its mercapturic acid, which is excreted in the urine.
The apparent terminal elimination half-life of monomethyl fumarate is about 2 hours.
Linearity/non-linearity
Despite the high inter-subject variability, the exposure measured as AUC and Cmax was generally dose-proportional after single dose administration of 4 × 30 mg dimethyl fumarate tablets (total dose of 120 mg) and 2 × 120 mg dimethyl fumarate tablets (total dose of 240 mg).
Renal impairment
No specific studies have been performed in patients with renal impairment. However, because renal elimination plays a minor role in the total clearance from plasma, it is unlikely that renal impairment may affect the pharmacokinetic characteristics of Skilarence (see section 4.2).
Hepatic impairment
No specific studies have been performed in patients with hepatic impairment. However, as dimethyl fumarate is metabolised by esterases and the alkaline milieu of the small intestine without the involvement of cytochrome P450, hepatic impairment is not expected to influence exposure (see section 4.2).
Preclinical safety data
Non-clinical safety pharmacology and genotoxicity data reveal no special hazard for humans.
Toxicology
The kidney was identified as a major target organ of toxicity in non-clinical studies. Renal findings in dogs included minimal to moderate tubular hypertrophy, increased incidence and severity of tubular vacuolation and minimal to slight tubular degeneration, which were considered toxicologically relevant. The no-observed adverse-effect-level (NOAEL) after 3 months of treatment was 30 mg/kg/day, which corresponds to 2.9-fold and 9.5-fold the human systemic exposure at the highest recommended dose (720 mg/day), as AUC and Cmax values, respectively.
Reproduction toxicity
No fertility or pre- and post-natal development studies have been conducted with Skilarence.
There were no effects on foetal body weights or malformations attributed to maternal administration of dimethyl fumarate during the embryo-foetal development study in rats. However, there was an increased number of foetuses with the variations “supernumerary liver lobe” and “abnormal iliac alignment” at maternally toxic doses. The NOAEL for maternal and embryo-foetal toxicity was 40 mg/kg/day, corresponding to 0.2-fold and 2.0-fold the human systemic exposure at the highest recommended dose (720 mg/day), as AUC and Cmax values, respectively.
Dimethyl fumarate has been shown to cross the placental membrane into foetal blood in rats.
Carcinogenicity
No carcinogenicity studies have been performed for Skilarence. Based on available data suggesting that fumaric acid esters may activate cellular pathways related to the development of renal tumours, a potential tumorigenic activity of exogenously administered dimethyl fumarate on the kidneys cannot be excluded.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.