TEIZEILD Concentrate for solution for infusion Ref.[116513] Active ingredients: Teplizumab
Source: European Medicines Agency (EU) Revision Year: 2026 Publisher: Sanofi Winthrop Industrie, 82 avenue Raspail, 94250 Gentilly, France
5.1. Pharmacodynamic properties
Pharmacotherapeutic Group: other drugs in diabetes
ATC Code: A10XX01
Mechanism of action
Teplizumab binds to CD3 (a cell surface antigen present on T lymphocytes) and delays disease progression in patients with stage 2 T1D. The mechanism may involve partial agonistic signalling leading to deactivation of autoreactive CD8+ T lymphocytes and reduced immune-mediated beta-cell destruction. Teplizumab leads to an increase in the proportion of CD8+ T cells with signs of exhaustion in peripheral blood.
Pharmacodynamic effects
Clinical studies have shown that teplizumab binds to CD3 molecules on the surface of both CD4+ and CD8+ T cells during treatment, with internalisation of the teplizumab/CD3 complex from the surface of T cells. Pharmacodynamic effects include transient lymphopenia with a reduction in circulating T cells with a nadir on the 5th day of dosing, during a 14-day course of teplizumab treatment (see section 4.4). Teplizumab exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of teplizumab have not been fully characterised.
Target patient population
Teplizumab is indicated in adult and paediatric patients 8 years of age and older who have a diagnosis of stage 2 T1D.
Stage 2 T1D confirmed by:
- At least two positive pancreatic islet autoantibodies
- Dysglycaemia without overt hyperglycaemia
Clinical efficacy and safety
The effectiveness of teplizumab was investigated in the following clinical study:
Study TN-10
A randomised, double-blind, event-driven, placebo-controlled study in 76 patients, 8 to 49 years of age with stage 2 T1D. Stage 2 T1D was defined as having both of the following:
1. Two or more of the following pancreatic islet autoantibodies:
- Glutamic acid decarboxylase 65 (GAD) autoantibodies
- Insulin autoantibody (IAA)
- Insulinoma-associated antigen 2 autoantibody (IA-2A)
- Zinc transporter 8 autoantibody (ZnT8)
- Islet cell autoantibody (ICA)
2. Dysglycaemia on oral glucose tolerance testing
In this study, patients were randomised 1:1 to receive teplizumab or placebo once daily by intravenous infusion for 14 days. The 2 treatment groups were:
- Group 1: Daily intravenous doses of 51 micrograms/m², 103 micrograms/m², 207 micrograms/m², and 413 micrograms/m² on study days 0 to 3, respectively, and 1 dose of 826 micrograms/m² on each of study days 4 to 13. The total dose for the 14-day course was approximately 9 034 micrograms/m².
- Group 2: Intravenous placebo only.
Patients in the teplizumab group had a total drug exposure that was comparable to the total drug exposure achieved with the recommended total teplizumab dose (see section 4.2). The primary efficacy endpoint in this study was the time from randomisation to development of stage 3 T1D diagnosis.
Baseline patient characteristics
In this study, 45% were female and the median age was 14 years (72% were <18 years old). Patients' characteristics are displayed in Table 2.
Table 2. Baseline characteristics of adult and paediatric patients 8 years of age and older with stage 2 T1D (Study TN-10)1:
| Teplizumab N=44 | Placebo N=32 | |
| Age group | ||
| ≥18 years | 34% | 19% |
| <18 years | 66% | 81% |
| Paediatric age group quartiles | ||
| 8 to <11 years | 21% | 25% |
| 11 to <14 years | 27% | 31% |
| 14 to <18 years | 18% | 25% |
| Glucose, mg/dL2 | ||
| median (min, max) | 165 (115, 207) | 154 (103, 200) |
| OGTT 30 minutes, median (min, max) | 161 (99, 237) | 165 (121, 223) |
| OGTT 60 minutes, median (min, max) | 186 (97, 244) | 173 (77, 233) |
| OGTT 90 minutes, median (min, max) | 175 (98, 242) | 159 (82, 244) |
| OGTT 120 minutes, median (min, max) | 152 (87, 240) | 144 (81, 217) |
| HbA1c, % | ||
| median (min, max) | 5.2 (4.6, 6.1) | 5.3 (4.3, 5.6) |
| HLA-DR3/DR4 | ||
| Both DR3 and DR4 | 25% | 22% |
| DR3 only | 23% | 25% |
| DR4 only | 36% | 44% |
| Neither DR3 nor DR4 | 11% | 9% |
| Not analysed | 5% | 0 |
| Autoantibody type positive | ||
| GAD65 | 91% | 88% |
| IAA | 43% | 34% |
| IA-2A | 59% | 75% |
| ICA | 66% | 88% |
| ZnT8 | 73% | 75% |
| Autoantibodies positive (N) | ||
| 1 | 2% | 0 |
| 2 | 27% | 22% |
| 3 | 25% | 16% |
| 4 | 27% | 44% |
| 5 | 18% | 19% |
1 Intent to treat (ITT) population
2 The glucose data are area under the time-concentration curve (AUC) values from the oral glucose tolerance test.
Abbreviations: HbA1c=haemoglobin A1c, SD=standard deviation, HLA=human leukocyte antigen, GAD65=glutamic acid decarboxylase 65 (GAD) autoantibody, IAA=insulin autoantibody, IA-2A=insulinoma-associated antigen 2 autoantibody, ZnT8=zinc transporter 8 autoantibody, ICA=islet cell autoantibody.
Efficacy results
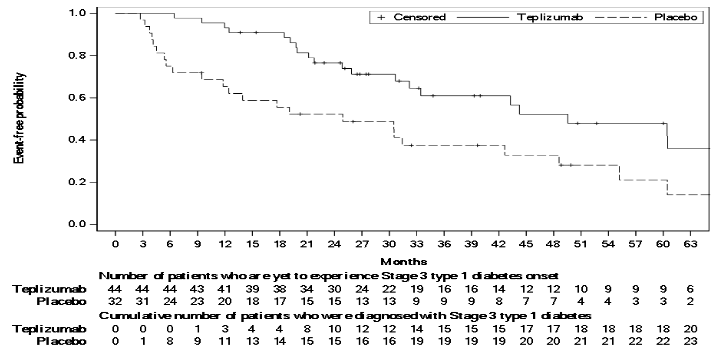
In study TN-10, stage 3 T1D was diagnosed in 20 (45%) of the patients treated with teplizumab and in 23 (72%) of the patients treated with placebo. A Cox proportional hazards model, stratified by age and oral glucose tolerance test status at randomisation, showed that the median time from randomisation to stage 3 T1D diagnosis was 50 months in the teplizumab group and 25 months in the placebo group, for a difference of 25 months. With a median follow-up time of 51 months, therapy with teplizumab resulted in a statistically significant delay in the development of stage 3 T1D, hazard ratio 0.41 (95% CI: 0.22 to 0.78; p=0.0066) (Figure 1).
Study TN-10 was not designed to assess whether there were differences in the effectiveness between subgroups based on demographic characteristics or baseline disease characteristics.
Figure 1. Kaplan-Meier curve of time to diagnosis of stage 3 T1D in adult and paediatric patients 8 years of age and older with stage 2 T1D by treatment group (Study TN-10)1:
1 ITT population
Paediatric population
The European Medicines Agency has deferred the obligation to submit the results of studies with Teizeild containing teplizumab in one or more subsets of the paediatric population in prevention of stage 3 type 1 diabetes mellitus as per paediatric investigation plan (PIP) EMEA-000524-PIP02-24, for the granted indication (see section 4.2 for information on paediatric use).
5.2. Pharmacokinetic properties
Steady state concentrations of teplizumab are not expected to be achieved during the 14-day course of teplizumab.
Absorption
There is no information about absorption since teplizumab is administered intravenously.
Distribution
No protein binding studies were conducted as teplizumab is a monoclonal antibody.
Metabolism
Teplizumab is expected to be metabolised into small peptides by catabolic pathways.
Elimination
The apparent elimination half-life of teplizumab is approximately 3 days.
Special populations
Age
No clinically significant differences in the pharmacokinetics of teplizumab were observed based on age (8 to 35 years old).
Gender
No clinically significant differences in the pharmacokinetics of teplizumab were observed based on gender.
Race
No clinically significant differences in the pharmacokinetics of teplizumab were observed based on racial groups (White, Asian).
Body weight
BSA-based dosing normalises the exposure to teplizumab across body weight.
Renal impairment
No specific studies to evaluate the pharmacokinetics of teplizumab in patients with renal impairment have been performed.
Hepatic impairment
No specific studies to evaluate the pharmacokinetics of teplizumab in patients with hepatic impairment have been performed.
Paediatric population
The pharmacokinetics of teplizumab in children younger than 8 years of age have not been established.
5.3. Preclinical safety data
No studies have been performed to assess the genotoxic, including mutagenic, potential of teplizumab. As an antibody, teplizumab is not expected to interact directly with DNA. No long-term studies have been performed to assess the carcinogenic potential of teplizumab. Based on the weight of evidence assessment and the proposed long term immunomodulatory mode of action a very low potential carcinogenicity risk cannot fully be excluded.
Developmental and reproductive toxicity
Non-clinical studies conducted in mice using a surrogate antibody directed towards murine CD3 indicate direct or indirect harmful with respect to pregnancy and embryonic/foetal development.
In an embryo-foetal developmental toxicity study in pregnant mice by subcutaneous injection at dose levels of 0, 0.03, 0.3, or 20 mg/kg on gestation days 6, 10, and 14, increase in post-implantation loss occurred in the 20 mg/kg group in the presence of maternal toxicity.
In a pre- and postnatal development toxicity study in pregnant mice administered every 3 days from gestation day 6 through lactation day 19 at doses of 0, 0.3, 3, or 20 mg/kg, no maternal toxicity or increased incidence of post-implantation loss was observed. Reductions in T cell populations and increases in B cells, and a reduction in the adaptive immune response to keyhole limpet hemocyanin (KLH) were observed in the offspring on postnatal days 35 and 84 at 20 mg/kg. The surrogate antibody was present in the offspring serum at level less than 1.5% that of maternal serum at the high dose. A trend towards reduction in fertility was observed in the offspring of dams at 20 mg/kg.
Fertility and reproductive performance were unaffected in female and male mice that received a murine surrogate anti-mouse CD3 antibody administered by the subcutaneous route at doses up to 20 mg/kg.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.