TRYNGOLZA Solution for injection Ref.[115519] Active ingredients: Olezarsen
Source: FDA, National Drug Code (US) Revision Year: 2025
12.1. Mechanism of Action
Olezarsen is an ASO-GalNAc3 conjugate that binds to apoC-III mRNA leading to mRNA degradation and resulting in a reduction of serum apoC-III protein. Reduction of apoC-III protein leads to increased clearance of plasma TG and VLDL.
12.2. Pharmacodynamics
Fasting apoC-III
Olezarsen decreased fasting apoC-III following administration of TRYNGOLZA 80 mg dosage every 4 weeks to patients with FCS [See Clinical Studies (14)]. The placebo-corrected percent change in fasting apoC-III from baseline was -57% at 1 month, -69% at 3 months, -72% at 6 months, and -80% at 12 months.
Cardiac Electrophysiology
At a dose 1.5-times the maximum approved recommended dosage, TRYNGOLZA does not prolong the QT interval to any clinically relevant extent.
12.3. Pharmacokinetics
Olezarsen steady state mean (SD) maximum concentrations (Cmax) is 883 (662) ng/mL and area under the curve (AUCτ) is 7440 (3880) ng*h/mL at the approved recommended dosage in patients with FCS. Olezarsen Cmaxand AUC increase dose-proportionally following single subcutaneous doses ranging from 10 to 120 mg (i.e., 0.13- to 1.5-times the approved recommended dose) in healthy volunteers. No olezarsen accumulation occurs with repeat dosing.
Absorption
Olezarsen time to Cmax (Tmax) is approximately 2 hours following subcutaneous administration.
Distribution
The population estimates for the apparent central volume of distribution is 91.9 L and the apparent peripheral volume of distribution is 2960 L for olezarsen. Olezarsen is greater than 99% bound to plasma proteins, in vitro.
Olezarsen distributes primarily to the liver and kidney after subcutaneous dosing.
Elimination
Olezarsen terminal elimination half-life is approximately 4 weeks.
Metabolism
Olezarsen is metabolized by endo- and exonucleases to short oligonucleotide fragments of varying sizes within the liver.
Excretion
Less than 1% of olezarsen administered dose is eliminated unchanged in urine within 24 hours.
Specific Populations
No clinically significant differences in the pharmacokinetics of olezarsen were observed based on age (<65 to ≥75 years), body weight, sex, race (White, Black or African American, Asian, Japanese, American Indian or Alaska Native, Native Hawaiian or Pacific Islander), mild-to-moderate renal impairment (eGFR ≥30 to <90 mL/min) [CKD-EPI], or mild hepatic impairment (total bilirubin ≤ ULN and AST > ULN, or total bilirubin >1 to 1.5 x ULN and any AST, National Cancer Institute Organ Dysfunction Working Group criteria). The effect of severe renal impairment (eGFR <30 mL/min), end-stage renal disease, moderate or severe hepatic impairment (total bilirubin >1.5 x ULN with any AST) on olezarsen pharmacokinetics is unknown.
Drug Interaction Studies
In vitro Studies
CYP450 Enzymes: Olezarsen is not an inhibitor or inducer of CYP450 enzymes.
Transporter Systems: Olezarsen is not a substrate or inhibitor of OAT1, OAT3, OCT1, OCT2, OATP1B1, OATP1B3, MATE1, MATE2-K, BCRP, P-gp, and BSEP.
Protein Binding: Olezarsen does not displace warfarin and ibuprofen from plasma protein binding sites.
13.1. Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
No long-term carcinogenicity studies were conducted with olezarsen in animals. However, the unconjugated antisense oligonucleotide (ASO) lacking GalNAc was administered weekly in mice and rats at subcutaneous doses of 0, 6, 25 and 40 mg/kg/week (along with a mouse-specific surrogate ASO at 25 mg/kg/week) and 0, 0.2, 1 and 5 mg/kg/week, respectively, for 2 years. In male mice, there were statistically significant increases in the incidences of hepatocellular adenomas and carcinomas at ≥25 mg/kg/week and hemangiomas and hemangiosarcomas at all doses. In female mice, there were statistically significant increases in the incidences of histiocytic sarcomas at all doses (including the mouse-specific surrogate) and pituitary gland adenomas at 25 mg/kg/week. In rats, the incidence of malignant fibrous histiocytoma at the injection site was increased in both sexes at doses ≥1 mg/kg/week. These tumors are considered a response to chronic tissue irritation and inflammation caused by repeated subcutaneous injection. The clinical significance of these findings is uncertain.
Mutagenesis
Olezarsen was negative for genotoxicity in vitro (bacterial reverse mutation assay and chromosome aberration assay in Chinese hamster lung cells) and in vivo (mouse bone marrow micronucleus assay).
Impairment of Fertility
Olezarsen was administered at doses of 0, 5, 10, or 20 mg/kg given every other week to male and female mice prior to mating, followed by every other day dosing after mating and until gestation day 6 in females. There was no effect on fertility in mice administered olezarsen at doses up to 20 mg/kg (approximately 2-times the monthly maximum recommended human dose based on body surface area).
14. Clinical Studies
The efficacy of TRYNGOLZA was demonstrated in a randomized, placebo-controlled, double-blind clinical trial in adult patients with genetically identified FCS and fasting triglyceride (TG) levels ≥880 mg/dL (Trial 1; NCT04568434). After a ≥4-week run-in period where patients continued to follow a low-fat diet with ≤20 grams fat per day, patients were randomly assigned to receive doses every 4 weeks of TRYNGOLZA 80 mg (n=22) or matching volume of placebo (n=23) via subcutaneous injection over a 53-week treatment period.
Patient demographic and baseline characteristics were generally similar across the treatment groups [see Adverse Reactions (6.1)]. The proportion of patients with diabetes at enrollment was 32% in the TRYNGOLZA 80 mg group compared with 26% in the placebo group. Patients in the TRYNGOLZA 80 mg and placebo groups were treated with statins (27%), omega-3 fatty acids (42%), fibrates (49%), or other lipid lowering therapies (13%) at study entry. Seventy-one percent (71%) of patients in the TRYNGOLZA 80 mg and placebo groups combined had a history of documented acute pancreatitis in the prior 10 years. Mean (SD) and median fasting TG levels at baseline were 2,604 (1,364) mg/dL and 2,303 mg/dL, respectively (range of 334 to 6,898 mg/dL).
The primary endpoint was percent change in fasting triglycerides from baseline to Month 6 (average of Weeks 23, 25, and 27) compared to placebo. The difference between TRYNGOLZA 80 mg group and the placebo group in percent change in fasting triglycerides from baseline to Month 6 was -42.5% (95% CI: -74.1%, -10.9%; p=0.0084). For additional results see Table 2.
Table 2. Mean Baseline (BL) and Mean Percent (%) Changes from Baseline in Lipid/Lipoprotein Parameters in Patients with FCS at Month 6 in Trial 1:
| Parameter (mg/dL) | TRYNGOLZA 80 mg N=22 | Placebo N=23 | TRYNGOLZA 80 mg vs. Placebo | ||
|---|---|---|---|---|---|
| BL | % change Month 6 | BL | % change Month 6 | Treatment Difference % change (95% CI) at Month 6 | |
| Triglycerides | 2613.1 | -30 | 2595.7 | +12 | -42.5* (-74.1, -10.9) |
| Non-HDL-C | 262.9 | -18 | 271.3 | +5.7 | -23.4 (-45.3, -1.5) |
| LDL-C | 22.8 | +64 | 16.7 | +9 | +55.0† (0.7, 109.4) |
| Total ApoB | 58.4 | +20 | 59.7 | +9 | +11.7 (-12.6, 35.9) |
| ApoB-48 | 11.6 | -51 | 14.2 | +25 | -75.9 (-149.8, -2.0) |
Abbreviations: apoB = apolipoprotein B; non-HDL-C = non high-density lipoprotein cholesterol; LDL-C = low-density lipoprotein cholesterol.
Note: Analyses results were based on an analysis of covariance model with treatment, the two randomization stratification factors, prior history of pancreatitis within 10 years prior to Screening (yes vs. no), previous treatment with the unconjugated ASO (yes vs. no) as the fixed effects and log-transformed Baseline value as a covariate. Missing data was imputed using placebo washout imputation. The 95% CIs of treatment differences were calculated using a robust variance estimator. For triglycerides and non-HDL, a test of residual normality did not indicate significant departure from normal distribution.
* Reached statistical significance (p value <0.05).
† Mean LDL-C levels increased but remained within normal range (i.e., <70 mg/dL for 74% of patients treated with TRYNGOLZA).
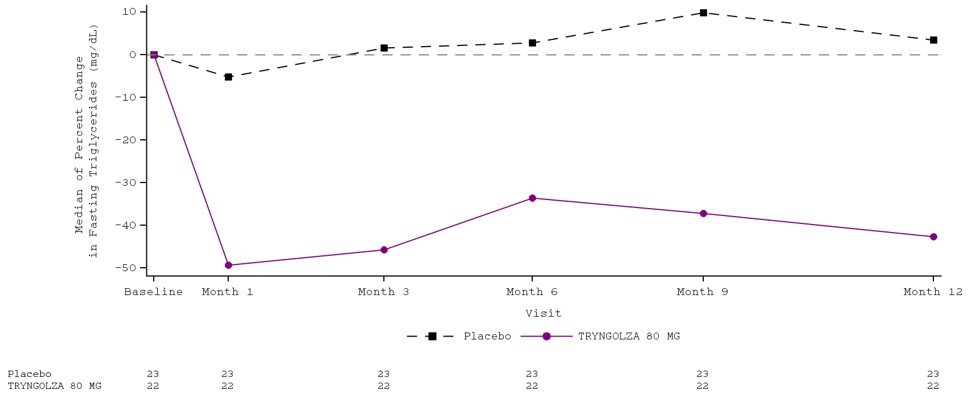
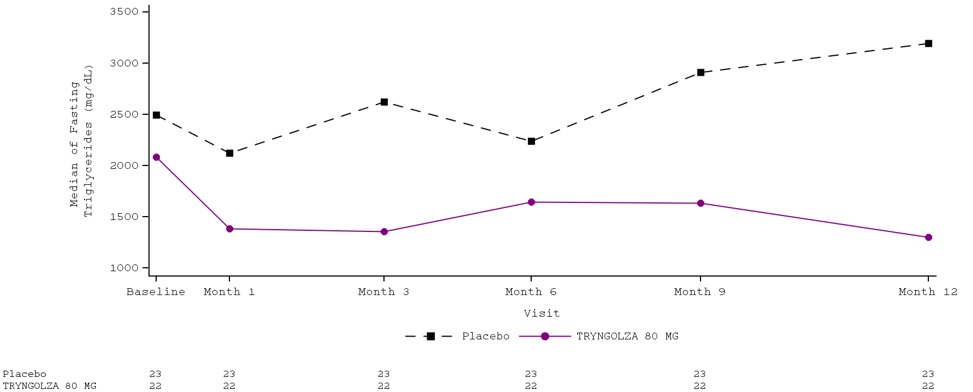
Median percent change from baseline (Figure 1) and median absolute TG values (Figure 2) over time demonstrated a consistent lowering effect during the 12-month treatment period.
Figure 1. Percent Change in Fasting Triglyceride (mg/dL) Over Time:
Figure 2. Fasting Triglyceride (mg/dL) Over Time:
Over the 12-month treatment period, the numerical incidence of acute pancreatitis in patients treated with TRYNGOLZA 80 mg was lower compared with placebo [1 (5%) patient in the TRYNGOLZA 80 mg group compared with 7 (30%) patients in the placebo group]; all of these patients had a prior history of pancreatitis within 10 years prior to screening.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.