XOLREMDI Hard capsule Ref.[116582] Active ingredients: Mavorixafor
Source: European Medicines Agency (EU) Revision Year: 2026 Publisher: X4 Pharmaceuticals (Austria) GmbH, Hohenstaufengasse 9/DG, 1010 Vienna, Austria
5.1. Pharmacodynamic properties
Pharmacotherapeutic group: Immunostimulants, Other immunostimulants
ATC code: L03AX24
Mechanism of action
Mavorixafor is a CXC Chemokine Receptor 4 (CXCR4) antagonist that blocks the binding of the CXCR4 ligand, stromal-derived factor-1α (SDF-1α)/CXC Chemokine Ligand 12 (CXCL12). SDF-1/CXCR4 plays a role in trafficking and homing of leukocytes to and from the bone marrow compartment. Gain of function mutations in the CXCR4 receptor gene that occur in patients with WHIM syndrome lead to increased responsiveness to CXCL12 and retention of leukocytes in the bone marrow. Mavorixafor inhibits the response to CXCL12 in both wild‑type and mutated CXCR4 variants associated with WHIM syndrome. Treatment with mavorixafor results in increased mobilisation of neutrophils and lymphocytes and monocytes from the bone marrow into peripheral circulation.
Pharmacodynamic effects
Absolute neutrophil count (ANC) and absolute lymphocyte count (ALC) peaked at 4 hours after Xolremdi dosing and returned towards baseline within 24 h after dosing. Over mavorixafor doses of 50 mg (0.125 times the maximum recommended dose) to 400 mg once daily, higher mavorixafor exposure at steady state was associated with longer mean time (hours) above ANC threshold (TATANC) of 500 cells/μL and longer mean time (hours) above ALC threshold (TATALC) of 1 000 cells/μL over a 24-hour period.
Cardiac electrophysiology
In a QT study, the maximum mean increase in the QTc interval was 15.6 ms (upper bound of the 90% confidence interval = 19.8 ms) after administration of Xolremdi 800 mg (2 times the maximum recommended dose) in healthy volunteers. See section 4.4.
Clinical efficacy and safety
Xolremdi was assessed in two clinical studies. Study X4P-001-103 (hereafter, study 1) was a pivotal Phase 3 randomised, double-blind, placebo-controlled, multicentre clinical study in adult and adolescent (aged 12 years and older) patients with WHIM syndrome. Study X4P-001-MKKA (hereafter, study 2) was a supportive open-label Phase 2 study in adult patients with WHIM syndrome.
Phase 3 study (pivotal)
The efficacy of Xolremdi in adult and adolescent patients aged 12 to <18 years with WHIM syndrome was evaluated in the 52-week, randomised, double-blind, placebo-controlled period of study 1. All enrolled patients had a genotype-confirmed variant of CXCR4 consistent with WHIM syndrome, and a confirmed ANC ≤400 cells/μL. Mavorixafor 400 mg was administered orally once daily to adults and adolescents weighing >50 kg and 200 mg once daily to adolescents weighing ≤50 kg. Patients were permitted to continue (but not initiate) immunoglobulin therapy at the same dose. Use of other CXCR4 antagonists or granulocyte-colony stimulating factor (G-CSF) was not permitted.
Thirty-one patients were randomised 1:1 to receive either placebo (n=17) or mavorixafor (n=14) once daily for 52 weeks. Baseline patient demographics and disease characteristics are shown in Table 5.
Table 5. Baseline demographic and baseline characteristics in patients with WHIM syndrome (study 1):
| Demographics and disease characteristics | Xolremdi (N=14) | Placebo (N=17) |
| Demographics | ||
| Age (years) mean (SD) | 22.1 (12.20) | 30.9 (21.25) |
| Age group, n (%) | - | - |
| 12 to <18 years | 7 (50.0) | 8 (47.1) |
| ≥18 years | 7 (50.0) | 9 (52.9) |
| Sex, n (%) | - | - |
| Male | 5 (35.7) | 8 (47.1) |
| Female | 9 (64.3) | 9 (52.9) |
| Race, n (%) | - | - |
| White | 13 (93) | 16 (94) |
| Asian | 0 | 1 (6) |
| Other | 1 (7) | 0 |
| Disease characteristics | ||
| Baseline Ig use, n (%) | - | - |
| Yes | 6 (42.9) | 8 (47.1) |
| Baseline mean absolute neutrophil count (ANC) (cells/μL) mean (SD) | 155 (93.8) | 281 (232.7) |
| Baseline mean absolute lymphocyte count (ALC) (cells/μL) mean (SD) | 501 (204.8) | 563 (199.1) |
Abbreviations: SD = standard deviation; Ig = immunoglobulin.
Note: Percentages are calculated based on the number of patients within each characteristic as denominator.
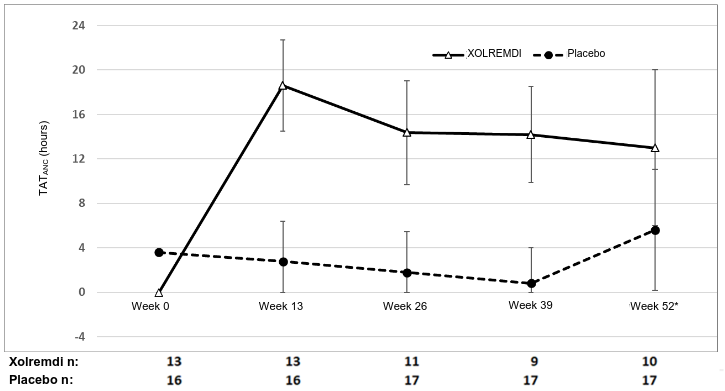
The primary efficacy endpoint was improvement in ANC as measured by the mean time (hours) above ANC threshold (TATANC) of 500 cells/μL which was assessed over a 24-hour period 4 times throughout the study (every 3 months for 12 months). Over the 52-week period, TATANC was statistically significantly greater in patients treated with mavorixafor compared with placebo. See Table 6 and Figure 1.
Table 6. Mean time (hours) above ANC threshold (TATANC) in study 1:
| Xolremdi (n=14) | Placebo (n=17) | ||
| TATANC (hours) | |||
| Baseline | Mean (SD) | 0.0 (0.0) | 3.6 (5.7) |
| Overall MMRM results | LS mean (SE) | 15.0 (1.89) | 2.8 (1.52) |
| LS mean 95% CI | (11.2, 18.9) | (0.0, 5.9) | |
| Difference from placebo: | |||
| LS mean difference (SE) | 12.30 (2.5) | - | |
| LS mean difference 95% CI | (7.2, 17.4) | - | |
| P-value1 | <0.0001 | - | |
Abbreviations: ANC = absolute neutrophil count; CI = confidence interval; LS = least squares; MMRM = mixed-model repeated measures; SD = standard deviation; SE = standard error; TAT = time above threshold of 500 cells/μL.
1 The results are based on an MMRM analysis with time above threshold as a dependent variable; treatment, visit (weeks 13, 26, 39 and 52), treatment × visit, Ig use (randomisation strata), and baseline time above threshold as covariates; and patient as the repeated random effect.
Figure 1. TATANC over time (hours) (LS Mean ± 95% CI) by treatment Group (study 1):
Abbreviations: ANC = absolute neutrophil count; CI = confidence interval; LS = least squares; TAT = total time (hours) above threshold (500 cells/μL) in 24 hours.
* At week 52, 3 of 17 placebo patients were given mavorixafor in advance of their TAT measurement as they entered the open-label period of the study; one mavorixafor patient did not take mavorixafor. All data were included in the ITT analysis.
The key secondary efficacy endpoint was improvement in ALC as measured by the mean time (hours) above ALC threshold (TATALC) of 1 000 cells/μL over a 24-hour period. Over the 52-week period, TATALC was statistically significantly greater in patients treated with mavorixafor compared with placebo. See Table 7.
Table 7. Mean time (hours) above ALC threshold (TATALC) in study 1:
| Xolremdi (n=14) | Placebo (n=17) | ||
| TATALC (hours) | |||
| Baseline | Mean (SD) | 2.2 (5.07) | 2.8 (5.86) |
| Overall MMRM results | LS mean (SE) | 15.8 (1.39) | 4.6 (1.15) |
| LS mean 95% CI | (13.0, 18.7) | (2.2, 6.9) | |
| Difference from placebo: | |||
| LS mean difference (SE) | 11.3 (1.80) | - | |
| LS mean difference 95% CI | (7.5, 15.0) | - | |
| P-value1 | <0.0001 | - | |
Abbreviations: ALC = absolute lymphocyte count; CI = confidence interval; LS = least squares; MMRM = mixed-model repeated measures; SD = standard deviation; SE = standard error; TAT = time above threshold of 1 000 cells/μL.
1 The results are based on an MMRM analysis with time above threshold as a dependent variable; treatment, visit (weeks 13, 26, 39 and 52), treatment × visit, Ig use (randomisation strata), and baseline time above threshold as covariates; and patient as the repeated random effect.
The efficacy of mavorixafor was further assessed on total infection score and total wart change score. Across the 52-week treatment period, the severity-weighted total infection score was numerically lower in mavorixafor-treated patients [LS mean (SE) 7.41 (2.805)] compared with placebo-treated patients [LS mean (SE) 12.27 (2.443)] with a mean difference of -4.85 [95% CI (-12.57, 2.86)]. Similarly, the annualised infection rate was numerically lower in mavorixafor-treated patients [LS mean (SE) 1.7 (0.5)] compared with placebo-treated patients [LS mean (SE) 4.2 (0.7)] with a rate ratio of 0.417 [95% CI (0.220, 0.789)]. There was no difference in total wart change scores between the mavorixafor and placebo treatment arms over the 52-week period.
Phase 2 study (supportive)
In an open-label Phase 2 (study 2) in 8 adult patients with WHIM syndrome, the pharmacodynamics of mavorixafor over the dose range 50 to 400 mg administered orally once daily, were evaluated. Doses of 300 to 400 mg achieved sustained increases in ANC ≥600 cells/μL and ALC ≥1 000 cells/μL.
Paediatric population
The European Medicines Agency has deferred the obligation to submit the results of studies with Xolremdi in one or more subsets of the paediatric population for the treatment of WHIM (warts, hypogammaglobulinemia, infections, and myelokathexis) syndrome (see section 4.2 for information on paediatric use).
Other information
This medicinal product has been authorised under 'exceptional circumstances'. This means that due to the rarity of the disease it has not been possible to obtain complete information on this medicinal product. The European Medicines Agency will review any new information which may become available every year and this SmPC will be updated as necessary.
5.2. Pharmacokinetic properties
Mavorixafor pharmacokinetic parameters are presented as geometric mean (CV%) in adults with WHIM syndrome unless otherwise specified. Mavorixafor steady state Cmax is 3 304 (58.6%) ng/mL and the AUC from 0 to 24 hours (AUC0-24h) is 13 970 (58.4%) ng×h/mL following 400 mg once daily.
Absorption
Mavorixafor median (range) time to Cmax (tmax) is 2.8 hours (1.9 to 4 hours) at the highest approved recommended dose.
Effect of food
High fat meal: Mavorixafor Cmax decreased by 66% and AUC decreased by 55% following single-dose administration of Xolremdi 400 mg with a high‑fat meal (1 000 calories, 50% fat) to healthy subjects.
Low fat meal: Mavorixafor Cmax decreased by 55% and AUC decreased by 51% following single-dose administration of Xolremdi 400 mg with a low-fat meal (500 calories, 25% fat) to healthy subjects. In addition, a 14% higher mavorixafor Cmax and 18% lower AUC was observed following single-dose administration of Xolremdi 400 mg with a low-fat meal to healthy subjects after an overnight fast compared to fasting for an additional 4 hours after the Xolremdi dose (see section 4.2).
Distribution
Mavorixafor volume of distribution is 120 L/kg. Mavorixafor is > 93% bound to human plasma proteins in vitro.
Biotransformation
CYP3A4 and, to a lesser extent, CYP2D6 are primarily responsible for mavorixafor metabolism.
Elimination
Mavorixafor's terminal half-life was 82 h with an apparent clearance of 62 L/h following single-dose administration of Xolremdi 400 mg in healthy subjects. Mavorixafor exhibits at least partial nonlinear apparent clearance; however, this is not clinically significant at the approved recommended dose.
After a single oral dose of radiolabelled mavorixafor, 74.2% of the administered dose was recovered out of which 61.0% of administered radioactivity was recovered in faeces and 13.2% (3% unchanged) was recovered in the urine over the 240-hour collection period in healthy subjects.
Linearity/non-linearity
Mavorixafor demonstrates nonlinear pharmacokinetics with greater than dose-proportional increases in Cmax and AUC0-24h over a dose range of 50 mg (0.125 times the recommended dose) to 400 mg. Mavorixafor steady state is reached after approximately 9 to 12 days in healthy subjects at the highest approved recommended dose.
Pharmacokinetic/pharmacodynamic relationship(s)
In study 2, mean ANC levels for doses 50 to 200 mg generally remained below the clinical benefit threshold of 500 cells/μL during the 24-hour dosing interval. For 300 mg and 400 mg, mean ANC levels rose above the threshold by approximately 1 hour post-dose and remained above or at the threshold over the entire dosing interval. A mavorixafor dose of 300/400 mg QD was required to achieve AUCANC ≥600/μL and AUCALC ≥1 000/μL.
Drug interaction studies
For information regarding drug interactions with other medicinal products please refer to section 4.5.
Other medicinal products: No clinically significant differences in the pharmacokinetics of caffeine (CYP1A2 substrate), losartan (CYP2C9 substrate), omeprazole (CYP2C19 substrate), furosemide (OAT1 and OAT3 substrate) and oral contraceptives were observed following concomitant use with mavorixafor.
Special populations
Hepatic impairment
Mavorixafor is metabolised by the liver. The effect of moderate to severe hepatic impairment on the pharmacokinetics of mavorixafor has not been studied (see section 4.2).
Renal impairment
Renal clearance is a minor excretion pathway for mavorixafor. No clinically significant differences in the pharmacokinetics of mavorixafor were observed in mild to moderate renal impairment (CLcr 30 to less than 90 mL/min). The pharmacokinetics of mavorixafor have not been studied in subjects with severe renal impairment or end-stage renal disease (see section 4.2).
Elderly
In clinical studies of Xolremdi in patients with WHIM syndrome, 2 (5%) patients were aged 65 years and older, and no patients were aged 75 years and older. Clinical studies did not include sufficient numbers of patients aged 65 and older to determine whether they respond differently from younger patients.
Race/Ethnicity
The effect of race/ethnicity on mavorixafor systemic exposure is unknown.
Gender
The effect of gender on mavorixafor systemic exposure is unknown.
5.3. Preclinical safety data
Adverse reactions not observed in clinical studies, but seen in animals at exposure levels comparable to clinical exposure levels and with possible relevance to clinical use were as follows: testicular toxicity, hepatotoxicity, retinal degeneration and atrophy.
Genotoxicity
Mavorixafor was not genotoxic in an in vitro bacterial reverse mutation assay (Ames test), in an in vitro human lymphocyte culture chromosome aberration assay, or in an in vivo rat bone marrow micronucleus assay.
Reproductive toxicity
Animal reproduction studies have not been conducted with mavorixafor to evaluate effects on reproduction and embryo-foetal development. CXCR4/SDF-1 signalling plays an important role in mammalian embryo-foetal and placental development. In mice, CXCR4-/- knockout is embryo lethal and causes multiple developmental toxicities, most notably in the hematopoietic, cardiovascular and nervous systems. CXCR4/SDF-1 levels also have a key role in stimulating trophoblast proliferation and differentiation necessary for appropriate placental growth and function in humans. Based on its mechanism of action, Xolremdi may cause foetal harm when administered to a pregnant woman.
The effect of mavorixafor on male or female fertility was not studied in designated reproductive toxicology studies.
In a 39-week study with initiation of treatment in young prepubertal dogs, testicular changes of seminiferous tubule degeneration/atrophy including spermatogonial stem cell depletion were observed at exposure levels equivalent to the human exposure at MRHD; similar changes were not observed in a 13-week dog study in sexually mature male dogs and in a 26-week dog study in juvenile dogs covering the period of puberty. The mechanism by which mavorixafor may exert this effect is unknown, but a relation to pharmacological action of mavorixafor cannot be excluded. There are no data on the recovery of this effect.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.