XULTOPHY Solution for injection Ref.[50802] Active ingredients: Insulin degludec Insulin degludec and Liraglutide Liraglutide
Source: European Medicines Agency (EU) Revision Year: 2023 Publisher: Novo Nordisk A/S, Novo Allé, DK-2880 Bagsværd, Denmark
5.1. Pharmacodynamic properties
Pharmacotherapeutic group: Drugs used in diabetes. Insulins and analogues for injection, long-acting
ATC code: A10AE56
Mechanism of action
Xultophy is a combination product consisting of insulin degludec and liraglutide having complementary mechanisms of action to improve glycaemic control.
Insulin degludec is a basal insulin that forms soluble multi-hexamers upon subcutaneous injection, resulting in a depot from which insulin degludec is continuously and slowly absorbed into the circulation leading to a flat and stable glucose-lowering effect of insulin degludec with a low day-today variability in insulin action.
Insulin degludec binds specifically to the human insulin receptor and results in the same pharmacological effects as human insulin.
The blood glucose-lowering effect of insulin degludec is due to the facilitated uptake of glucose following the binding of insulin to receptors on muscle and fat cells and to the simultaneous inhibition of glucose output from the liver.
Liraglutide is a Glucagon-Like Peptide-1 (GLP-1) analogue with 97% sequence homology to human GLP-1 that binds to and activates the GLP-1 receptor (GLP-1R). Following subcutaneous administration, the protracted action profile is based on three mechanisms: self-association, which results in slow absorption; binding to albumin; and higher enzymatic stability towards the dipeptidyl peptidase IV (DPP-IV) and neutral endopeptidase (NEP) enzymes, resulting in a long plasma half-life.
Liraglutide action is mediated via a specific interaction with GLP-1 receptors and improves glycaemic control by lowering fasting and postprandial blood glucose. Liraglutide stimulates insulin secretion and lowers inappropriately high glucagon secretion in a glucose-dependent manner. Thus, when blood glucose is high, insulin secretion is stimulated and glucagon secretion is inhibited. Conversely, during hypoglycaemia liraglutide diminishes insulin secretion and does not impair glucagon secretion. The mechanism of blood glucose-lowering also involves a minor delay in gastric emptying.
Liraglutide reduces body weight and body fat mass through mechanisms involving reduced hunger and lowered energy intake.
GLP-1 is a physiological regulator of appetite and food intake, but the exact mechanism of action is not entirely clear. In animal studies, peripheral administration of liraglutide led to uptake in specific brain regions involved in regulation of appetite, where liraglutide, via specific activation of the GLP1R, increased key satiety and decreased key hunger signals, thereby leading to lower body weight.
GLP-1 receptors are also expressed in specific locations in the heart, vasculature, immune system, and kidneys. In mouse models of atherosclerosis, liraglutide prevented aortic plaque progression and reduced inflammation in the plaque. In addition, liraglutide had a beneficial effect on plasma lipids. Liraglutide did not reduce the plaque size of already established plaques.
Pharmacodynamic effects
Xultophy has a stable pharmacodynamic profile with a duration of action reflecting the combination of the individual action profiles of insulin degludec and liraglutide that allows for administration of Xultophy once daily at any time of the day with or without meals. Xultophy improves glycaemic control through the sustained lowering of fasting plasma glucose levels and postprandial glucose levels after all meals.
Postprandial glucose reduction was confirmed in a 4 hour standardised meal test substudy in patients uncontrolled on metformin alone or in combination with pioglitazone. Xultophy decreased the postprandial plasma glucose excursion (mean over 4 hours) significantly more than insulin degludec. The results were similar for Xultophy and liraglutide.
Clinical efficacy and safety
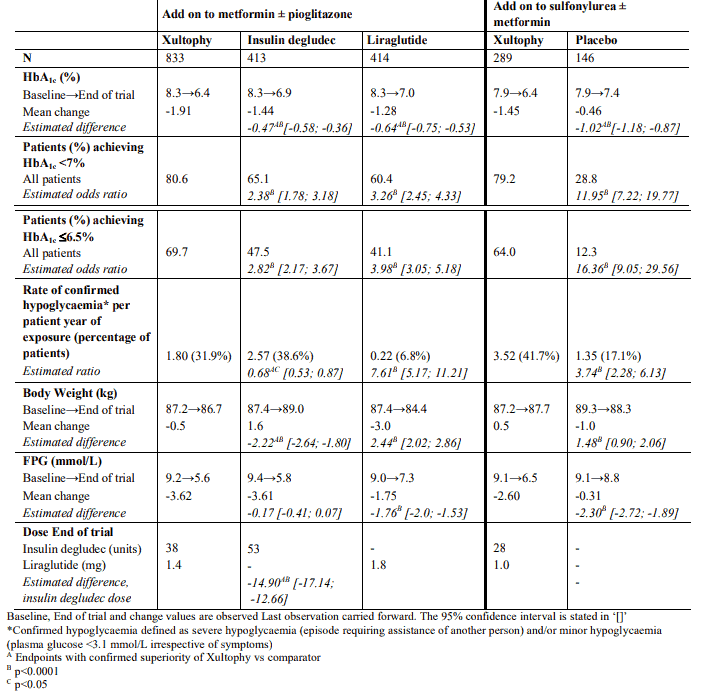
The safety and efficacy of Xultophy were evaluated in seven randomised, controlled, parallel group phase 3 trials in different populations of subjects with type 2 diabetes defined by previous antidiabetes treatment. Comparator treatments comprised basal insulin, GLP-1 RA therapy, placebo and a basal bolus regimen. The trials were of 26 weeks duration randomising between 199 and 833 patients to Xultophy. One study was further extended to 52 weeks. In all trials, the starting dose was given according to label and a twice-weekly titration regimen for Xultophy was used (see Table 2). The same titration algorithm was applied for basal insulin comparators. In six studies, Xultophy produced clinically and statistically significant improvements in glycaemic control versus comparators as measured by glycated haemaglobin A1c (HbA1c), whereas one study demonstrated a similar reduction of HbA1c in both treatment arms.
Table 2. Titration of Xultophy:
| Pre-breakfast plasma glucose* | Dose adjustment (twice weekly) | |
|---|---|---|
| mmol/L | mg/dL | Xultophy (dose steps) |
| <4.0 | <72 | -2 |
| 4.0-5.0 | 72-90 | 0 |
| >5.0 | >90 | +2 |
* Self-measured plasma glucose. In the trial investigating Xultophy as add on to sulfonylurea the target was 4.0-6.0 mmol/L
Glycaemic control
Add-on to oral glucose-lowering medicinal products
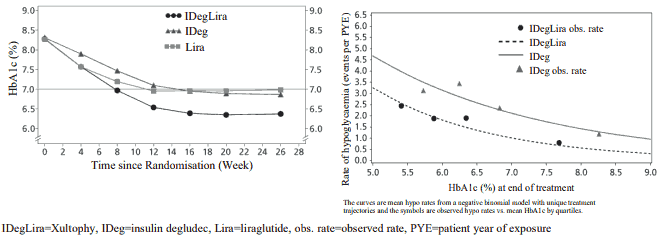
Adding Xultophy to metformin alone or in combination with pioglitazone in a 26-week randomised, controlled, open-label trial resulted in 60.4% of patients treated with Xultophy reaching a target of HbA1c <7% without confirmed hypoglycaemic episodes after 26 weeks of treatment. The proportion was significantly larger than observed with insulin degludec (40.9%, odds ratio 2.28, p <0.0001) and similar to that observed with liraglutide (57.7%, odds ratio 1.13, p=0.3184). The key results of the trial are listed in Figure 1 and Table 3.
Rates of confirmed hypoglycaemia were lower with Xultophy than with insulin degludec irrespective of the glycaemic control, see Figure 1. The rate per patient year of exposure (percentage of patients) of severe hypoglycaemia defined as an episode requiring assistance of another person was 0.01 (2 patients out of 825) for Xultophy, 0.01 (2 patients out of 412) for insulin degludec and 0.00 (0 patients out of 412) for liraglutide. The rate of nocturnal hypoglycaemic events was similar with Xultophy and insulin degludec treatment.
Patients treated with Xultophy overall experienced less gastrointestinal side effects than patients treated with liraglutide. This might be due to the slower increase in the dose of the liraglutide component during treatment initiation when using Xultophy as compared to using liraglutide alone.
The efficacy and safety of Xultophy were sustained up to 52 weeks of treatment. The reduction in HbA1c from baseline to 52 weeks was 1.84% with Xultophy with an estimated treatment difference of -0.65% compared to liraglutide (p<0.0001) and -0.46% compared to insulin degludec (p<0.0001). Body weight was reduced by 0.4 kg with an estimated treatment difference between Xultophy and insulin degludec of -2.80 kg (p<0.0001), and the rate of confirmed hypoglycaemia remained 1.8 events per patient year of exposure maintaining a significant reduction in overall risk of confirmed hypoglycaemia compared to insulin degludec.
Figure 1. Mean HbA1c ( % ) by treatment week (left) and rate of confirmed hypoglycaemia per patient year of exposure vs mean HbA1c ( % ) (right) in patients with type 2 diabetes mellitus inadequately controlled on metformin alone or in combination with pioglitazone:
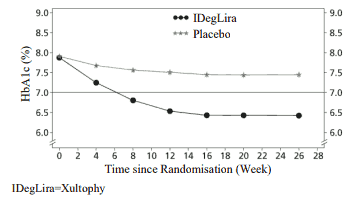
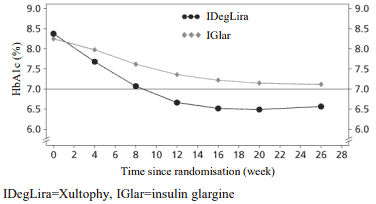
Xultophy as add-on to sulfonylurea alone or in combination with metformin were studied in a 26-week randomised, placebo-controlled, double-blind trial. The key results of the trial are listed in Figure 2 and Table 3.
Figure 2. Mean HbA1c ( % ) by treatment week in patients with type 2 diabetes mellitus inadequately controlled on sulfonylurea alone or in combination with metformin:
The rate per patient year of exposure (percentage of patients) of severe hypoglycaemia was 0.02 (2 patients out of 288) for Xultophy and 0.00 (0 patients out of 146) for placebo.
Table 3. Results at 26-weeks - Add on to oral glucose-lowering medicinal products:
In an open label trial comparing the efficacy and safety of Xultophy and insulin glargine 100 units/mL, both as add-on to SGLT2i ± OAD, Xultophy was superior to insulin glargine in reducing mean HbA1c after 26 weeks by 1.9% (from 8.2% to 6.3%) versus 1.7% (from 8.4% to 6.7%) with an estimated treatment difference of -0.36% [-0.50; -0.21]. Compared to baseline, Xultophy resulted in an unchanged mean body weight compared to a mean weight increase of 2.0 kg for patients treated with insulin glargine (estimated treatment difference -1.92 kg [95% CI: -2.64; -1.19]). The percentage of patients experiencing severe or blood-glucose confirmed symptomatic hypoglycaemia was 12.9% in the Xultophy group and 19.5% in the insulin glargine group (estimated treatment ratio 0.42 [95% CI: 0.23; 0.75]). The mean daily insulin dose at end of trial was 36 units for patients treated with Xultophy and 54 units for patients treated with insulin glargine.
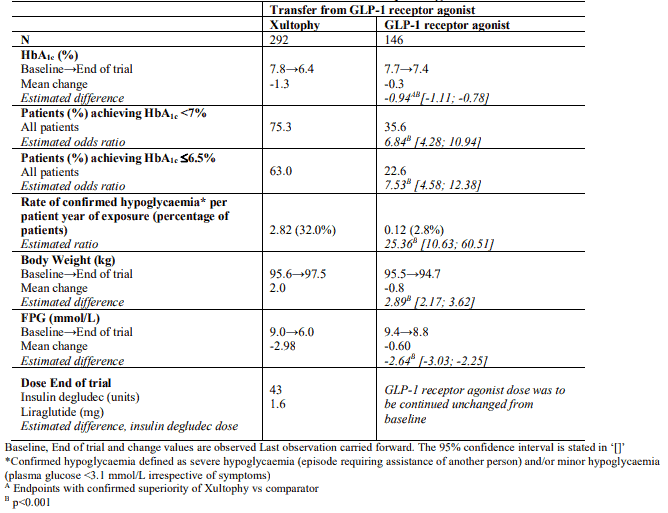
Transfer from GLP-1 receptor agonist therapy
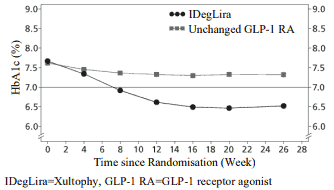
Transfer from GLP-1 receptor agonist to Xultophy compared to unchanged GLP-1 receptor agonist therapy (dosed according to label) were studied in a 26-weeks randomised, open-label trial in patients with type 2 diabetes mellitus inadequately controlled on a GLP-1 receptor agonist and metformin alone (74.2%) or in combination with pioglitazone (2.5%), sulfonylurea (21.2%) or both (2.1%). The key results of the trial are listed in Figure 3 and Table 4.
Figure 3. Mean HbA1c ( % ) by treatment week in patients with type 2 diabetes mellitus inadequately controlled on GLP-1 receptor agonists:
The rate per patient year of exposure (percentage of patients) of severe hypoglycaemia was 0.01 (1 patient out of 291) for Xultophy and 0.00 (0 patients out of 199) for GLP-1 receptor agonists.
Table 4. Results at 26-weeks – Transfer from GLP-1 receptor agonists:
Transfer from basal insulin therapies
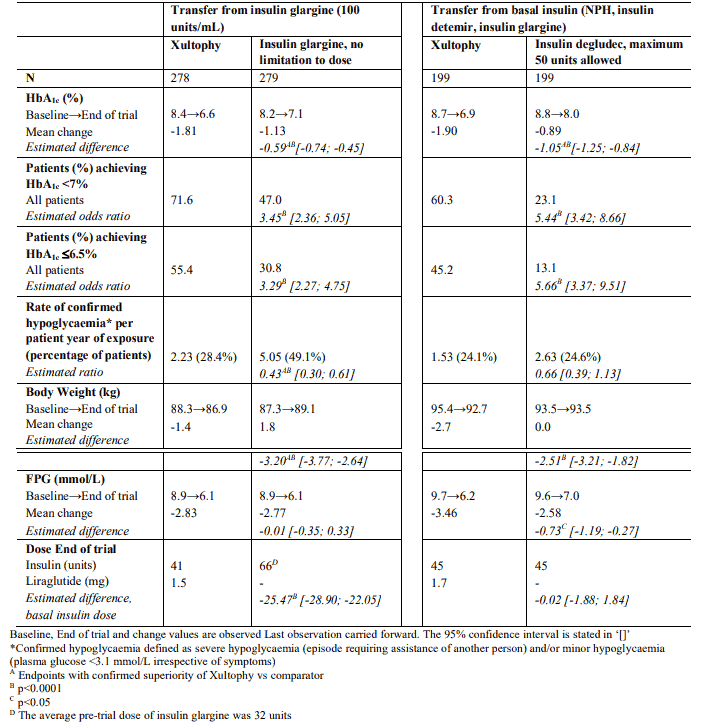
Transfer of patients from insulin glargine (100 units/mL) to Xultophy or intensification of insulin glargine in patients inadequately controlled on insulin glargine (20-50 units) and metformin were studied in a 26 week trial. The maximum allowed dose in the trial was 50 dose steps for Xultophy whereas there was no maximum dose for insulin glargine. 54.3% of patients treated with Xultophy reached the HbA1c target of <7% without confirmed hypoglycaemic episodes compared to 29.4% of patients treated with insulin glargine (odds ratio 3.24, p<0.001).
The key results of the trial are listed in Figure 4 and Table 5.
Figure 4. Mean HbA1c ( % ) by treatment week in patients with type 2 diabetes mellitus inadequately controlled on insulin glargine:
The rate per patient year of exposure (percentage of patients) of severe hypoglycaemia was 0.00 (0 patients out of 278) for Xultophy and 0.01 (1 patient out of 279) for insulin glargine. The rate of nocturnal hypoglycaemic events was significantly lower with Xultophy compared to insulin glargine (estimated treatment ratio 0.17, p<0.001).
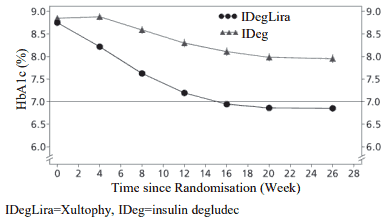
In a second trial, the transfer from basal insulin to Xultophy or insulin degludec was investigated in a 26-week randomised, double-blind trial in patients inadequately controlled on basal insulin (20-40 units) and metformin alone or in combination with sulfonylurea/glinides. Basal insulin and sulfonylurea/glinides were discontinued at randomisation. The maximum allowed dose was 50 dose steps for Xultophy and 50 units for insulin degludec. 48.7% of patients treated with Xultophy reached the HbA1c target of <7% without confirmed hypoglycaemic episodes. This was a significantly higher proportion than observed with insulin degludec (15.6%, odds ratio 5.57, p<0.0001). The key results of the trial are listed in Figure 5 and Table 5.
Figure 5. Mean HbA1c ( % ) by treatment week in patients with type 2 diabetes mellitus inadequately controlled on basal insulin:
The rate per patient year of exposure (percentage of patients) of severe hypoglycaemia was 0.01 (1 patient out of 199) for Xultophy and 0.00 (0 patients out of 199) for insulin degludec. The rate of nocturnal hypoglycaemic events was similar with Xultophy and insulin degludec treatment.
Table 5. Results at 26-weeks - Transfer from basal insulin:
Treatment with Xultophy compared to a basal-bolus insulin regimen consisting of basal insulin (insulin glargine 100 units/mL) in combination with bolus insulin (insulin aspart) studied in a 26-week trial in patients with type 2 diabetes mellitus inadequately controlled on insulin glargine and metformin demonstrated a similar reduction of HbA1c in the two groups (mean value from 8.2% to 6.7% in both groups). In both groups 66%–67% achieved HbA1c < 7%. Compared to baseline, there was a mean reduction in body weight of 0.9 kg for Xultophy and a mean increase of 2.6 kg for patients treated with a basal-bolus regimen and the estimated treatment difference was -3.57 kg [95% CI: - 4.19; -2.95]. The percentage of patients experiencing severe or blood-glucose confirmed symptomatic hypoglycaemia was 19.8% in the Xultophy group and 52.6% in the basal-bolus insulin group, and the estimated rate ratio was 0.11 [95% CI: 0.08-0.17]. The total daily insulin dose at end of trial was 40 units for patients treated with Xultophy and 84 units (52 units of basal insulin and 32 units of bolus insulin) for patients treated with a basal-bolus insulin regimen.
Cardiovascular Safety
No cardiovascular outcomes trials have been performed with Xultophy.
Liraglutide (Victoza)
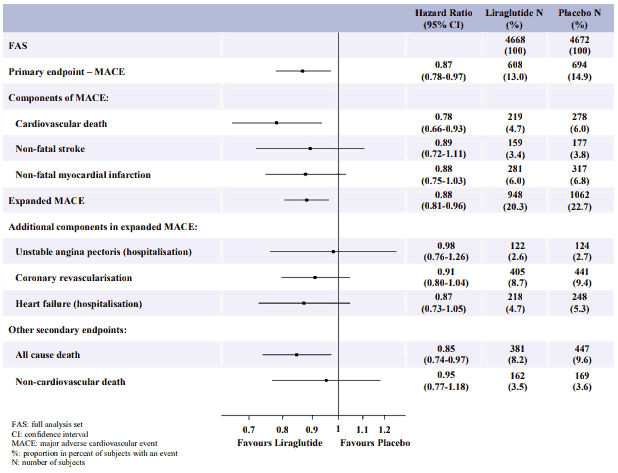
The Liraglutide Effect and Action in Diabetes Evaluation of Cardiovascular Outcome Results (LEADER) trial, was a multicentre, placebo-controlled, double-blind clinical trial. 9,340 patients were randomly allocated to either liraglutide (4,668) or placebo (4,672), both in addition to standards of care for HbA1c and cardiovascular (CV) risk factors. Primary outcome or vital status at end of trial was available for 99.7% and 99.6% of participants randomised to liraglutide and placebo, respectively. The duration of observation was minimum 3.5 years and up to a maximum of 5 years. The study population included patients ≥65 years (n=4,329) and ≥75 years (n=836) and patients with mild (n=3,907), moderate (n=1,934) or severe (n=224) renal impairment. The mean age was 64 years and the mean BMI was 32.5 kg/m². The mean duration of diabetes was 12.8 years.
The primary endpoint was the time from randomisation to first occurrence of any major adverse cardiovascular events (MACE): CV death, non-fatal myocardial infarction or non-fatal stroke. Liraglutide was superior in preventing MACE vs placebo (Figure 6).
Figure 6. Forest plot of analyses of individual cardiovascular event types – FAS population:
A reduction in HbA1c from baseline to month 36 was observed with liraglutide vs placebo, in addition to standard of care (-1.16% vs -0.77%; estimated treatment difference [ETD] -0.40% [-0.45; -0.34]).
Insulin degludec (Tresiba)
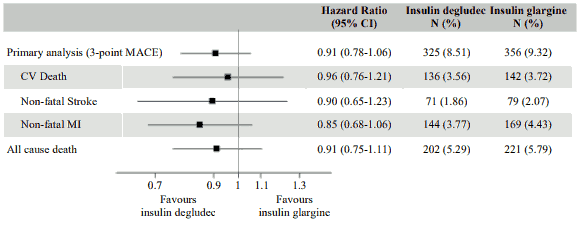
DEVOTE was a randomised, double-blind, and event-driven clinical trial with a median duration of 2 years comparing the cardiovascular safety of insulin degludec versus insulin glargine (100 units/mL) in 7,637 patients with type 2 diabetes mellitus at high risk of cardiovascular events. The primary analysis was time from randomisation to first occurrence of a 3-component major adverse cardiovascular event (MACE) defined as cardiovascular death, non-fatal myocardial infarction, or non-fatal stroke. The trial was designed as a non-inferiority trial to exclude a pre-specified risk margin of 1.3 for the hazard ratio (HR) of MACE comparing insulin degludec to insulin glargine. The cardiovascular safety of insulin degludec as compared to insulin glargine was confirmed (HR 0.91 [0.78; 1.06]) (Figure 7).
At baseline, HbA1c was 8.4% in both treatment groups and after 2 years HbA1c was 7.5% both with insulin degludec and insulin glargine.
Figure 7. Forest plot of analysis of the composite 3-point MACE and individual cardiovascular endpoints in DEVOTE:
Insulin secretion/beta-cell function
Xultophy improves beta-cell function compared to insulin degludec as measured by the homeostasis model assessment for beta-cell function (HOMA-β). Improved insulin secretion compared to insulin degludec in response to a standardised meal test was demonstrated in 260 patients with type 2 diabetes after 52 weeks of treatment. No data is available beyond 52 weeks of treatment.
Blood pressure
In patients inadequately controlled on metformin alone or in combination with pioglitazone, Xultophy reduced mean systolic blood pressure by 1.8 mmHg compared to a reduction of 0.7 mmHg with insulin degludec and 2.7 mmHg with liraglutide. In patients inadequately controlled on sulfonylurea alone or in combination with metformin, the reduction was 3.5 mmHg with Xultophy and 3.2 mmHg with placebo. The differences were not statistically significant. In three trials with patients inadequately controlled on basal insulin, systolic blood pressure was reduced by 5.4 mmHg with Xultophy and 1.7 mmHg with insulin degludec, with a statistically significant estimated treatment difference of -3.71 mmHg (p=0.0028), reduced by 3.7 mmHg with Xultophy vs 0.2 mmHg with insulin glargine, with a statistically significant estimated treatment difference of -3.57 mmHg (p<0.001) and reduced by 4.5 mmHg with Xultophy vs 1.16 mmHg with insulin glargine U100 plus insulin aspart, with a statistically significant estimated treatment difference of -3.70 mmHg (p=0.0003).
Paediatric population
The European Medicines Agency has waived the obligation to submit the results of studies with Xultophy in all subsets of the paediatric population for treatment of type 2 diabetes mellitus (see section 4.2 for information on paediatric use).
5.2. Pharmacokinetic properties
Overall, the pharmacokinetics of insulin degludec and liraglutide were not affected in a clinically relevant manner when administered as Xultophy compared with independent injections of insulin degludec and liraglutide.
The following reflects the pharmacokinetic properties of Xultophy unless stated that the presented data is from administration of insulin degludec or liraglutide alone.
Absorption
The overall exposure of insulin degludec was equivalent following administration of Xultophy versus insulin degludec alone while the Cmax was higher by 12%. The overall exposure of liraglutide was equivalent following administration of Xultophy versus liraglutide alone while Cmax was lower by 23%. The differences are considered of no clinical relevance since Xultophy is initiated and titrated according to the individual patient's blood glucose targets.
Insulin degludec and liraglutide exposure increased proportionally with the Xultophy dose within the full dose range based on a population pharmacokinetic analysis.
The pharmacokinetic profile of Xultophy is consistent with once-daily dosing and steady-state concentration of insulin degludec and liraglutide is reached after 2–3 days of daily administration.
Distribution
Insulin degludec and liraglutide are extensively bound to plasma proteins (>99% and >98%, respectively).
Biotransformation
Insulin degludec
Degradation of insulin degludec is similar to that of human insulin; all metabolites formed are inactive.
Liraglutide
During 24 hours following administration of a single radiolabelled [3H]-liraglutide dose to healthy subjects, the major component in plasma was intact liraglutide. Two minor plasma metabolites were detected (≤9% and ≤5% of total plasma radioactivity exposure). Liraglutide is metabolised in a similar manner to large proteins without a specific organ having been identified as major route of elimination.
Elimination
The half-life of insulin degludec is approximately 25 hours and the half-life of liraglutide is approximately 13 hours.
Special populations
Elderly patients
Age had no clinically relevant effect on the pharmacokinetics of Xultophy based on results from a population pharmacokinetic analysis including adult patients up to 83 years treated with Xultophy.
Gender
Gender had no clinically relevant effect on the pharmacokinetics of Xultophy based on results from a population pharmacokinetic analysis.
Ethnic origin
Ethnic origin had no clinically relevant effect on the pharmacokinetics of Xultophy based on results from a population pharmacokinetic analysis including White, Black, Indian, Asian and Hispanic groups.
Renal impairment
Insulin degludec:
There is no difference in the pharmacokinetics of insulin degludec between healthy subjects and patients with renal impairment.
Liraglutide:
Liraglutide exposure was reduced in patients with renal impairment compared to individuals with normal renal function. Liraglutide exposure was lowered by 33%, 14%, 27% and 26%, in patients with mild (creatinine clearance, CrCl 50-80 mL/min), moderate (CrCl 30-50 mL/min), and severe (CrCl <30 mL/min) renal impairment and in end-stage renal disease requiring dialysis, respectively. Similarly, in a 26-week clinical trial, patients with type 2 diabetes and moderate renal impairment (CrCl 30-59 mL/min) had 26% lower liraglutide exposure when compared with a separate trial including patients with type 2 diabetes with normal renal function or mild renal impairment.
Hepatic impairment
Insulin degludec:
There is no difference in the pharmacokinetics of insulin degludec between healthy subjects and patients with hepatic impairment.
Liraglutide:
The pharmacokinetics of liraglutide was evaluated in patients with varying degrees of hepatic impairment in a single-dose trial. Liraglutide exposure was decreased by 13–23% in patients with mild to moderate hepatic impairment compared to healthy subjects. Exposure was significantly lower (44%) in patients with severe hepatic impairment (Child Pugh score >9).
Paediatric population
No studies have been performed with Xultophy in children and adolescents below 18 years of age.
5.3. Preclinical safety data
The non-clinical development programme for insulin degludec/liraglutide included pivotal combination toxicity studies of up to 90 days duration in a single relevant species (Wistar rats) to support the clinical development programme. Local tolerance was assessed in rabbits and pigs.
Non-clinical safety data revealed no safety concern for humans based on repeated dose toxicity studies.
The local tissue reactions in the two studies in rabbits and pigs, respectively, were limited to mild inflammatory reactions.
No studies have been conducted with the insulin degludec/liraglutide combination to evaluate carcinogenesis, mutagenesis or impairment of fertility. The following data are based upon studies with insulin degludec and liraglutide individually.
Insulin degludec
Non-clinical data reveal no safety concern for humans based on studies of safety pharmacology, repeated dose toxicity, carcinogenic potential, and toxicity to reproduction. The ratio of mitogenic relative to metabolic potency for insulin degludec is unchanged compared to human insulin.
Liraglutide
Non-clinical data reveal no special hazards for human based on conventional studies of safety pharmacology, repeat-dose toxicity, or genotoxicity. Non-lethal thyroid C-cell tumours were seen in 2-year carcinogenicity studies in rats and mice. In rats, a no observed adverse effect level (NOAEL) was not observed. These tumours were not seen in monkeys treated for 20 months. These findings in rodents are caused by a non-genotoxic, specific GLP-1 receptor-mediated mechanism to which rodents are particularly sensitive. The relevance for humans is likely to be low but cannot be completely excluded. No other treatment-related tumours have been found.
Animal studies did not indicate direct harmful effects with respect to fertility but slightly increased early embryonic deaths at the highest dose. Dosing with liraglutide during mid-gestation caused a reduction in maternal weight and foetal growth with equivocal effects on ribs in rats and skeletal variation in the rabbit. Neonatal growth was reduced in rats while exposed to liraglutide, and persisted in the post-weaning period in the high dose group. It is unknown whether the reduced pup growth is caused by reduced pup milk intake due to a direct GLP-1 effect or reduced maternal milk production due to decreased caloric intake.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.