YUVIWEL Powder for solution for injection Ref.[116347] Active ingredients: Navepegritide
Source: FDA, National Drug Code (US) Revision Year: 2026
Product description
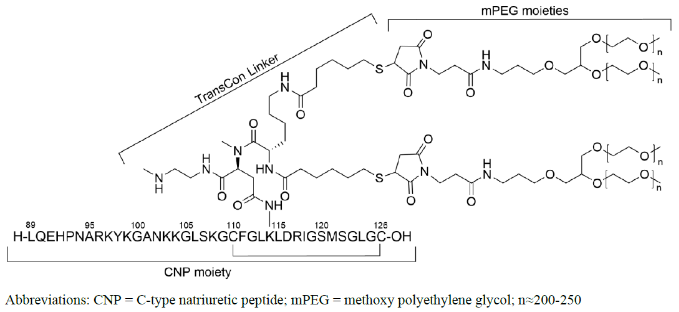
YUVIWEL for injection contains navepegritide, a C-type natriuretic peptide (CNP) analog. Navepegritide is a prodrug of active CNP consisting of a CNP moiety transiently conjugated to two branched 20 kDa methoxy polyethylene glycol (mPEG) moieties via a proprietary TransCon Linker. The amino acid sequence of the CNP moiety is identical to the 38 amino acid sequence of residues 89-126 of human CNP. The structural formula of navepegritide is as follows:
The molecular formula is C231H386N64O67S5 + 4 × (C2H4O)n, where n is between 200 and 250. The average molecular weight is approximately 45 kDa.
YUVIWEL is provided as a sterile, lyophilized white to off-white powder for reconstitution to a colorless solution with Sterile Water for Injection, USP (diluent). The diluent for reconstitution of YUVIWEL is provided in a prefilled syringe.
The compositions of YUVIWEL are shown in Table 3.
Table 3. Content of YUVIWEL:
| Strength | Composition of YUVIWEL (gross content per vial)* | CNP(89-126) Concentration After Reconstitution |
| 1.3 mg/vial | Navepegritide equivalent to 1.9 mg CNP(89-126), succinic acid (1.0 mg), trehalose dihydrate (72.2 mg), tromethamine and hydrochloric acid (q.s. for adjustment to pH 5.0) | 2.2 mg/mL |
| 2.8 mg/vial | Navepegritide equivalent to 4.0 mg CNP(89-126), succinic acid (1.0 mg), trehalose dihydrate (66.7 mg), tromethamine and hydrochloric acid (q.s. for adjustment to pH 5.0) | 4.6 mg/mL |
| 5.5 mg/vial | Navepegritide equivalent to 6.9 mg CNP(89-126), succinic acid (1.5 mg), trehalose dihydrate (91.9 mg), tromethamine and hydrochloric acid (q.s. for adjustment to pH 5.0) | 5.5 mg/mL |
* An overfill is included in the vial to compensate for loss in the vial and during transfer.
| Dosage Forms and Strengths |
|---|
|
For injection: 1.3 mg, 2.8 mg, and 5.5 mg of CNP(89-126) as a white to off-white lyophilized powder in a single-dose vial for reconstitution. |
| How Supplied | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
YUVIWEL (navepegritide) for injection is supplied as a lyophilized white to off-white powder in a vial, in three strengths: 1.3 mg, 2.8 mg, and 5.5 mg (see Table 5). Each YUVIWEL carton contains 4 Kits with 1 Prescribing Information, and 1 Instructions for Use. Table 5. YUVIWEL Strengths and Contents of Each Kit:
Only use the vial, needles, and syringes that are in the Kits for handling of YUVIWEL [see Dosage and Administration (2.3)] and do not use the components for other medicinal products. Manufactured for: Ascendis Pharma Growth Disorders A/S, Tuborg Boulevard 12, DK-2900 Hellerup, Denmark |
Drugs
| Drug | Countries | |
|---|---|---|
| YUVIWEL | United States |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.