ZOLADEX Implant, in pre-filled syringe Ref.[8359] Active ingredients: Goserelin
Source: Medicines & Healthcare Products Regulatory Agency (GB) Revision Year: 2017 Publisher: AstraZeneca UK Limited, 600 Capability Green, Luton, LU1 3LU, UK
Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in section 6.1.
Pregnancy and lactation (see section 4.6).
Special warnings and precautions for use
There is an increased risk of incident depression (which may be severe) in patients undergoing treatment with GnRH agonists, such as Goserelin. Patients should be informed accordingly and treated as appropriate if symptoms occur.
Androgen deprivation therapy may prolong the QT interval.
In patients with a history of or risk factors for QT prolongation and in patients receiving concomitant medicinal products that might prolong the QT interval (see section 4.5) physicians should assess the benefit risk ratio including the potential for Torsade de pointes prior to initiating Zoladex.
Injection site injury has been reported with Zoladex, including events of pain, haematoma, haemorrhage and vascular injury. Monitor affected patients for signs or symptoms of abdominal haemorrhage. In very rare cases, administration error resulted in vascular injury and haemorrhagic shock requiring blood transfusions and surgical intervention. Extra care should be taken when administering Zoladex to patients with a low BMI and/or receiving full anticoagulation medications (see section 4.2).
Males
The use of Zoladex in men at particular risk of developing ureteric obstruction or spinal cord compression should be considered carefully, and the patients monitored closely during the first month of therapy. If spinal cord compression or renal impairment due to ureteric obstruction are present or develop, specific standard treatment of these complications should be instituted.
Consideration should be given to the initial use of an anti-androgen (e.g. cyproterone acetate 300 mg daily for three days before and three weeks after commencement of Zoladex) at the start of LHRH analogue therapy since this has been reported to prevent the possible sequelae of the initial rise in serum testosterone.
The use of LHRH agonists may cause reduction in bone mineral density. In men, preliminary data suggest that the use of a bisphosphonate in combination with an LHRH agonist may reduce bone mineral loss. Particular caution is necessary in patients with additional risk factors for osteoporosis (e.g. chronic alcohol abusers, smokers, long-term therapy with anticonvulsants or corticosteroids, family history of osteoporosis).
Patients with known depression and patients with hypertension should be monitored carefully.
Reduction in glucose tolerance has been observed in men receiving LHRH agonists. This may manifest as diabetes or loss of glycaemic control in patients with pre-existing diabetes mellitus. Thus, monitoring of blood glucose levels should be considered.
Myocardial infarction and cardiac failure were observed in a pharmaco-epidemiology study of LHRH agonists used in the treatment of prostate cancer. The risk appears to be increased when used in combination with anti-androgens.
Females
Breast cancer indication
Reduced bone mineral density:
The use of LHRH agonists may cause reduction in bone mineral density. Following two years treatment for early breast cancer, the average loss of bone mineral density was 6.2% and 11.5% at the femoral neck and lumbar spine respectively. This loss has been shown to be partially reversible at the one year off treatment follow-up with recovery to 3.4% and 6.4% relative to baseline at the femoral neck and lumbar spine respectively, although this recovery is based on very limited data. In the majority of women, currently available data suggest that recovery of bone loss occurs after cessation of therapy.
Preliminary data suggest that the use of Zoladex in combination with tamoxifen in patients with breast cancer may reduce bone mineral loss.
Benign indications
Loss of bone mineral density:
The use of LHRH agonists is likely to cause reduction in bone mineral density averaging 1% per month during a six month treatment period. Every 10% reduction in bone mineral density is linked with about a two to three times increased fracture risk. In the majority of women, currently available data suggest that recovery of bone loss occurs after cessation of therapy.
In patients receiving Zoladex for the treatment of endometriosis, the addition of hormone replacement therapy has been shown to reduce bone mineral density loss and vasomotor symptoms.
No specific data is available for patients with established osteoporosis or with risk factors for osteoporosis (e.g. chronic alcohol abusers, smokers, long-term therapy with drugs that reduce bone mineral density, e.g. anticonvulsants or corticosteroids, family history of osteoporosis, malnutrition, e.g. anorexia nervosa). Since reduction in bone mineral density is likely to be more detrimental in these patients, treatment with Zoladex should be considered on an individual basis and only be initiated if the benefits of treatment outweigh the risks following a very careful appraisal. Consideration should be given to additional measures in order to counteract loss of bone mineral density.
Withdrawal bleeding
During early treatment with Zoladex some women may experience vaginal bleeding of variable duration and intensity. If vaginal bleeding occurs it is usually in the first month after starting treatment. Such bleeding probably represents oestrogen withdrawal bleeding and is expected to stop spontaneously. If bleeding continues, the reason should be investigated.
There are no clinical data on the effects of treating benign gynaecological conditions with Zoladex for periods in excess of six months.
The use of Zoladex may cause an increase in cervical resistance and care should be taken when dilating the cervix.
Zoladex should only be administered as part of a regimen for assisted reproduction under the supervision of a specialist experienced in the area.
As with other LHRH agonists, there have been reports of ovarian hyperstimulation syndrome (OHSS), associated with the use of Zoladex 3.6 mg in combination with gonadotrophin. The stimulation cycle should be monitored carefully to identify patients at risk of developing OHSS. If OHSS risk is present, human chorionic gonadotrophin (hCG) should be withheld, if possible.
It is recommended that Zoladex is used with caution in fertilisation treatment of patients with polycystic ovarian syndrome as follicle recruitment may be increased.
Fertile women should use non-hormonal contraceptive methods during treatment with Zoladex and until reset of menstruation following discontinuation of treatment with Zoladex.
Patients with known depression and patients with hypertension should be monitored carefully.
Treatment with Zoladex may lead to positive reactions in anti-doping tests.
Paediatric population
Zoladex is not indicated for use in children, as safety and efficacy have not been established in this patient group.
Interaction with other medicinal products and other forms of interaction
Since androgen deprivation treatment may prolong the QT interval, the concomitant use of Zoladex with medicinal products known to prolong the QT interval or medicinal products able to induce Torsade de pointes such as class IA (e.g. quinidine, disopyramide) or class III (e.g. amiodarone, sotalol, dofetilide, ibutilide) antiarrhythmic medicinal products, methadone, moxifloxacin, antipsychotics, etc. should be carefully evaluated (see section 4.4).
Pregnancy and lactation
Pregnancy
Zoladex should not be used during pregnancy since concurrent use of LHRH agonists is associated with a theoretical risk of abortion or foetal abnormality. Prior to treatment, potentially fertile women should be examined carefully to exclude pregnancy. Non-hormonal methods of contraception should be employed during therapy until menses resume (see also warning concerning the time to return of menses in section 4.4).
Pregnancy should be excluded before Zoladex is used for fertilisation treatment. When Zoladex is used in this setting, there is no clinical evidence to suggest a causal connection between Zoladex and any subsequent abnormalities of oocyte development or pregnancy or outcome.
Breast-feeding
The use of Zoladex during breast-feeding is not recommended.
Effects on ability to drive and use machines
Zoladex has no or negligible influence on the ability to drive and use machines.
Undesirable effects
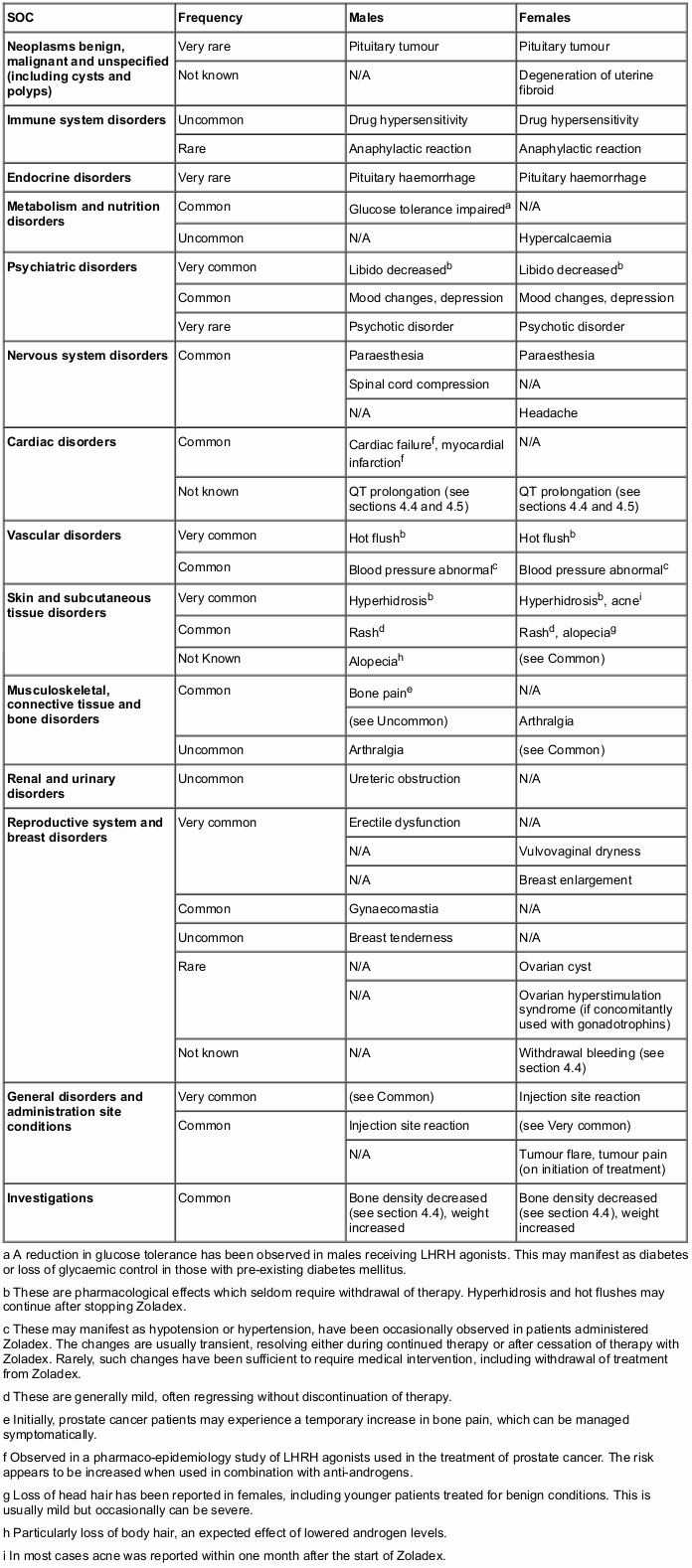
The following frequency categories for adverse drug reactions (ADRs) were calculated based on reports from Zoladex clinical trials and post-marketing sources. The most commonly observed adverse reactions include hot flushes, sweating and injection site reactions.
The following convention has been used for classification of frequency: Very common (≥1/10), Common (≥1/100 to <1/10), Uncommon (≥1/1,000 to <1/100), Rare (≥1/10,000 to <1/1,000), Very rare (<1/10,000) and Not known (cannot be estimated from the available data).
Table. Zoladex 3.6 mg adverse drug reactions presented by MedDRA System Organ Class:
Post-marketing experience
A small number of cases of changes in blood count, hepatic dysfunction, pulmonary embolism and interstitial pneumonia have been reported in connection with Zoladex.
In addition, the following adverse drug reactions have been reported in women treated for benign gynaecological indications:
Acne, change of body hairs, dry skin, weight gain, increase in serum cholesterol, ovarian hyperstimulation syndrome (if concomitantly used with gonadotropines), vaginitis, vaginal discharge, nervousness, sleep disorder, tiredness, peripheral oedema, myalgias, cramp in the calves, nausea, vomiting, diarrhoea, constipation, abdominal complaints, alterations of voice.
Initially, breast cancer patients may experience a temporary increase in signs and symptoms, which can be managed symptomatically.
Rarely, breast cancer patients with metastases have developed hypercalcaemia on initiation of therapy. In the presence of symptoms indicative of hypercalcaemia (e.g. thirst), hypercalcaemia should be excluded.
Rarely, some women may enter the menopause during treatment with LHRH analogues and not resume menses on cessation of therapy. Whether this is an effect of Zoladex treatment or a reflection of their gynaecological condition is not known.
Reporting of suspected adverse reactions
Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the Yellow Card Scheme, Website: www.mhra.gov.uk/yellowcard.
Incompatibilities
None known.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.