ZYCUBO Solution for injection Ref.[116175] Active ingredients: Copper
Source: FDA, National Drug Code (US) Revision Year: 2026
12.1. Mechanism of Action
Menkes disease is an X-linked recessive disorder caused by pathogenic variants in the copper transport ATPase encoded by ATP7A. Patients with Menkes disease have impaired absorption of copper from their diet, impaired transport of copper across the blood-brain barrier, and dysregulation of many copper-dependent enzymes. ZYCUBO is a bioavailable copper replacement therapy that is administered as a subcutaneous injection to bypass the impaired gastrointestinal absorption observed in patients with Menkes disease.
12.2. Pharmacodynamics
Patients with Menkes disease have low serum concentrations of copper and ceruloplasmin. Treatment with ZYCUBO increases serum copper and ceruloplasmin concentrations in patients with Menkes disease.
The exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of ZYCUBO have not been fully characterized.
12.3. Pharmacokinetics
The geometric mean (CV%) maximum serum concentration (Cmax) of copper histidinate was 67 (36%) ng/mL, the geometric mean (CV%) area under the concentration-time curve from time 0 to 24 hours (AUC0-24hr) was 186 (21%) ng•hr/mL, and the geometric mean (CV%) area under the concentrationtime curve from time 0 to infinity (AUC0-inf) was 296 (15%) ng•hr/mL following a single subcutaneous dose of 3 mg copper histidinate (approximately twice the approved recommended dose for Menkes disease patients 1 year old or older) in healthy adult subjects.
At the recommended dosage, the mean (SD) serum copper concentration increased from a baseline concentration of 30 (25) mcg/dL to 114 (38) mcg/dL at 12 months, and gradually decreased over the 36-month treatment period, with a mean (SD) serum copper concentration of 63 (31) mcg/dL at 36 months. The mean (SD) serum ceruloplasmin concentration was 12 (12) mg/dL at baseline, 33 (11) mg/dL at 12 months, and 20 (8) mg/dL at 36 months [see Clinical Studies (14)].
Absorption
The absolute bioavailability of copper histidinate following subcutaneous injection has not been determined. The median time to reach maximum serum concentrations of copper histidinate (Tmax) was 0.75 hours following a single subcutaneous dose of 3 mg copper histidinate (approximately twice the approved recommended dose for Menkes disease patients 1 year old or older) in healthy adult subjects.
Distribution
The mean (SD) apparent volume of distribution (Vz/F) of copper histidinate during the terminal elimination phase was 1034 (588) L following a single subcutaneous dose of 3 mg copper histidinate (approximately twice the approved recommended dose for Menkes disease patients 1 year old or older) in healthy adult subjects.
Serum copper histidinate concentration-time profiles exhibited an initial decrease followed by a terminal elimination phase, consistent with the release of copper from the copper histidinate complex to the carrier proteins ceruloplasmin and albumin, followed by incorporation of copper histidinate and histidine into the endogenous pools of copper histidinate and histidine, respectively.
No binding of copper histidinate to human plasma proteins in vitro has been observed.
Elimination
The mean (SD) serum clearance (CL/F) of copper histidinate was 10.3 (1.6) L/hr, and the mean terminal half-life was 75 hours following a single subcutaneous dose of 3 mg copper histidinate (approximately twice the approved recommended dose for Menkes disease patients 1 year old or older) in healthy adult subjects.
Metabolism
The metabolism of copper histidinate has not been well characterized.
Excretion
Elimination of elemental copper is through biliary excretion.
Drug Interaction Studies
In vitro Studies
Copper histidinate is a weak inhibitor of CYP1A2, CYP2B6, CYP2C9, CYP2C19, and CYP2D6 (IC50 >100 μM). Copper histidinate is not an inducer of CYP1A2, CYP2B6, or CYP3A.
Transporter Systems
Copper histidinate is not a substrate of BCRP, P-gp, MATE1, MATE2-K, OAT1, OAT3, OATP1B1, OAT1B3, or OCT2. Copper histidinate is an inhibitor of BSEP and MATE1. Copper histidinate is a weak inhibitor of BCRP, MATE2K, OCT1, and OCT2. Copper histidinate is not an inhibitor of P-gp, OAT1, OAT3, OATP1B1, or OATP1B3.
13.1. Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Animal studies to evaluate the carcinogenic potential of copper histidinate have not been conducted with ZYCUBO.
Mutagenesis
ZYCUBO was not mutagenic or clastogenic in a standard battery of genotoxicity tests [bacterial mutagenicity (Ames), micronucleus assay in TK6 cells (in vitro), and rat bone marrow micronucleus assay (in vivo)].
Impairment of Fertility
Animal studies to evaluate effects of copp
14. Clinical Studies
The efficacy of ZYCUBO was evaluated in pediatric patients with Menkes disease (age at treatment initiation ranges 0.1 to 31.4 months) receiving 3 years of copper histidinate treatment in two openlabel, single-arm clinical trials (Trial 1, NCT00001262 and Trial 2, NCT00811785). Data from ZYCUBO-treated patients in these two trials were compared to data from an untreated contemporaneous external control cohort as collected under a protocol amendment of Trial 2. In both trials, pediatric patients:
- Less than 1 year of age received 1.45 mg of ZYCUBO administered subcutaneously twice daily until 1 year of age.
- Equal to or greater than 1 year of age received 1.45 mg of ZYCUBO subcutaneously once daily for up to 3 years.
Overall survival was evaluated in a subset of the pooled population from Trial 1 and Trial 2, referred to as the pooled efficacy population. This efficacy population included only patients with Menkes disease who carried a severe pathogenic variant of the ATP7A gene (duplication/deletion, nonsense, or a canonical splice junction variant) and were born after 1999. There were 83 pediatric patients (66 ZYCUBO; 17 external control) in this pooled efficacy population: 21 patients (21 ZYCUBO) from Trial 1 and 62 patients (45 ZYCUBO; 17 external control) from Trial 2.
Patients in the pooled efficacy population were assigned to 1 of 4 cohorts as described in Table 2.
Table 2. Patient Cohorts in the Pooled Efficacy Population:
| Treated Cohorts | Untreated Cohorts | |||
| ZYCUBO-Early Treatment (ZYCUBO-ET) n=31 | ZYCUBO-Late Treatment (ZYCUBO-LT) n=35 | External Control-Early Treatment (EC-ET) n=17 | External Control-Late Treatment (EC-LT) n=16 | |
| Eligibility | Started ZYCUBO treatment within 4 weeks of birth1 | Started ZYCUBO treatment after 4 weeks of birth2 | • No prior ZYCUBO or copper treatment • Asymptomatic for significant neurological signs and symptoms approximately 4 weeks after birth • Survived at least 4 weeks after diagnosis | • Subset of the EC-ET cohort • Diagnosed with Menkes disease after 4 weeks of birth • Survived at least 2 weeks after diagnosis |
| Age at diagnosis (months) | 0.1 (-4.5 – 1.9) | 4.8 (0.4 – 29.4) | 4.7 (2.1 – 22.2) | 5.6 (2.1 – 22.2) |
| Age at treatment initiation (months) | 0.4 (0.1 – 1.9) | 7.1 (1.3 – 31.4) | NA | NA |
| Treatment duration (months) | 34.1 (1.1 – 36) | 20 (1.3 – 36) | NA | NA |
All values are in median (range)
1 Within 4 weeks of birth or within 4 weeks of birth corrected for prematurity (i.e., <40 weeks' gestation)
2 After 4 weeks of birth or after 4 weeks of birth corrected for prematurity (i.e., <40 weeks' gestation)
In the 4 cohorts, 81 patients were male (98%) except for 2 female (2%) patients in ZYCUBO-LT. The pooled efficacy population included patients with the following race and ethnicity: 52 White (63%), 11 Hispanic (13%), 8 Black or African American (10%), 6 Unknown (7%), 4 Other (5%), and 2 Asian or Pacific Islander (2%). The majority of patients in all 4 cohorts were born prematurely: ZYCUBO-ET = 77%, ZYCUBO-LT = 66%, EC-ET = 82%, and EC-LT = 81%.
Efficacy Results
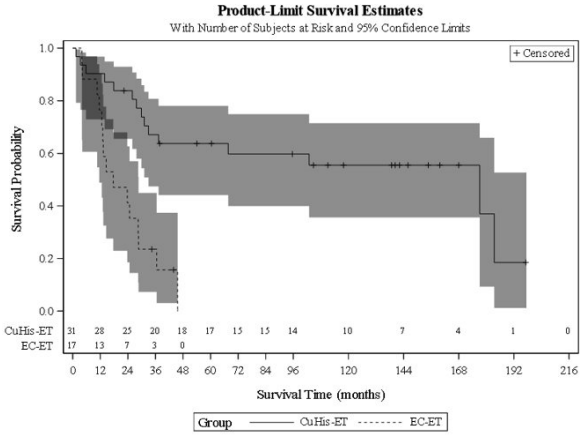
Primary Efficacy Results (Overall Survival) in the ZYCUBO-ET and EC-ET Cohorts The primary efficacy analysis compared the overall survival in patients in the ZYCUBO-ET and EC-ET cohorts. Patients in the ZYCUBO-ET cohort (patients treated with ZYCUBO) had a significant improvement in overall survival compared to patients in the EC-ET cohort, with a 78% reduction in the risk of death (Table 3 and Figure 1).
In the ZYCUBO-ET cohort, 15 (48%) patients survived >6 years, including 7 (23%) patients who survived >12 years. In the EC-ET cohort, no patients survived >6 years.
Table 3. Primary Efficacy Results: Overall Survival in ZYCUBO Early Treatment and External Control Early Treatment Cohorts with Menkes Disease:
| ZYCUBO-Early Treatment (n=31) | External Control-Early Treatment (n=17) | |
| Number (%) of Patients Alive | 16 (52%) | 2 (12%) |
| Median survival time (months) (95% CI) | 177.1 (33, NE) | 17.6 (11.5, 28.6) |
| Hazard Ratio (95% CI) | 0.22 (0.10, 0.49) | |
CI=Confidence Interval; NE=Not estimable
Note: If death dates were unknown, patients were censored at the last known date alive.
Figure 1. Kaplan-Meier Overall Survival Curve for the ZYCUBO Early Treatment and External Control Early Treatment Cohorts with Menkes Disease:
CuHis=copper histidinate; ET=early treatment; EC=external control
Secondary Efficacy Results (Overall Survival) in the ZYCUBO-LT and EC-LT Cohorts
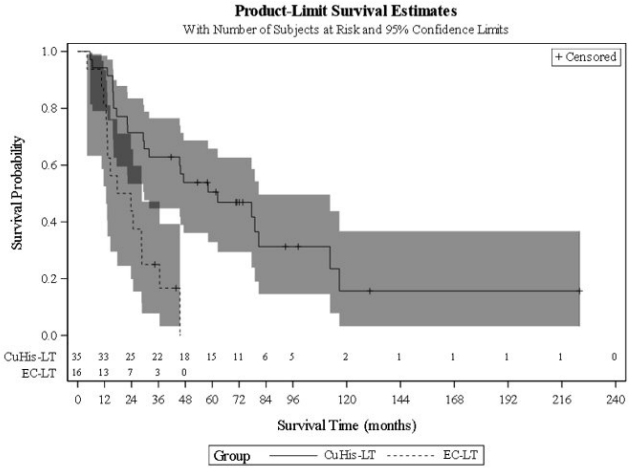
The secondary efficacy analysis compared the overall survival in patients in the ZYCUBO-LT cohort with patients in the EC-LT cohort. Patients in the ZYCUBO-LT cohort (patients treated with ZYCUBO) had a significant improvement in overall survival compared to patients in the EC-LT cohort, with a 73% reduction in the risk of death (Table 4 and Figure 2).
Table 4. Secondary Efficacy Results: Overall Survival in ZYCUBO Late Treatment and External Control Late Treatment Cohorts with Menkes Disease:
| ZYCUBO Late- Treatment (LT) (n=35) | External Control-Late Treatment (EC-LT) (n=16) | |
| Number of Patients Alive (%) | 12 (34%) | 2 (12%) |
| Median survival time (months) (95% CI) | 62.4 (29.6, 80.7) | 20.7 (12.6, 28.6) |
| Hazard Ratio (95% CI) | 0.27 (0.12, 0.57) | |
CI=Confidence Interval
Note: If death dates were unknown, patients were censored at the last known date alive.
Figure 2. Kaplan-Meier Overall Survival Curve for ZYCUBO Late Treatment and External Control Late Treatment Cohorts with Menkes Disease:
CuHis=copper histidinate; LT=late treatment; EC=external control
In the ZYCUBO-LT cohort, 11 (31.4%) patients survived >6 years, including 1 patient (2.9%) who survived >12 years. In the EC-LT cohort, no patients survived >6 years.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.