ZYNYZ Concentrate for solution for infusion Ref.[110050] Active ingredients: Retifanlimab
Source: European Medicines Agency (EU) Revision Year: 2026 Publisher: Incyte Biosciences Distribution B.V., Paasheuvelweg 25, 1105 BP Amsterdam, Netherlands
5.1. Pharmacodynamic properties
Pharmacotherapeutic group: Antineoplastic agents, PD-1/PD-L1 (Programmed cell death protein 1/death ligand 1) inhibitors
ATC code: L01FF10
Mechanism of action
Retifanlimab is an immunoglobulin G4 (IgG4) monoclonal antibody that binds to the programmed death receptor-1 (PD-1) and blocks its interaction with its ligands PD-L1 and PD-L2. Engagement of PD-1 with its ligands PD-L1 and PD-L2, which are expressed by antigen presenting cells and may be expressed by tumour cells and/or other cells in the tumour microenvironment, results in inhibition of T-cell function such as proliferation, cytokine secretion and cytotoxic activity. Retifanlimab binds to the PD-1 receptor, blocks interaction with its ligands PD-L1 and PD-L2, and potentiates T-cell activity.
Pharmacodynamic effects
Immunogenicity
Anti-drug antibodies (ADA) were uncommonly detected. No evidence of ADA impact on pharmacokinetics, efficacy or safety was observed.
Clinical efficacy and safety
Squamous cell carcinoma of the anal canal (SCAC)
The efficacy and safety of retifanlimab in combination with carboplatin and paclitaxel was studied in the POD1UM-303/InterAACT-2 study, a randomised, multicenter, double-blind phase III study that enrolled patients with chemotherapy-naïve metastatic or inoperable locally recurrent SCAC. Patients with active autoimmune disease or a medical condition that required immunosuppression, severe hepatic or renal impairment, clinically significant cardiac disease, history of organ transplant, Eastern Cooperative Oncology Group (ECOG) performance score (PS) ≥2, or evidence of interstitial lung disease or active noninfectious pneumonitis were ineligible. Patients who had not received prior systemic therapy other than a radiosensitising agent or neoadjuvant or adjuvant therapy completed >6 months prior to study entry were eligible as were patients who were HIV-positive if they had an undetectable viral load, a CD4+ count ≥200 cells/microliter and were receiving antiretroviral therapy. The study did not enroll patients who received prior treatment with PD-(L)1 directed therapies. Randomisation was stratified by PD-L1 expression (<1% versus ≥1%), region, and extent of disease (locally recurrent versus metastatic). Patients were randomised (1:1) to receive either:
- Retifanlimab 500 mg intravenously every 4 weeks on Day 1, carboplatin AUC of 5 mg/mL on Day 1, and paclitaxel 80 mg/m² on Days 1, 8, and 15 for 6 cycles followed by retifanlimab 500 mg intravenously every 4 weeks.
- Placebo intravenously every 4 weeks on Day 1, carboplatin AUC 5 mg/mL on Day 1, and paclitaxel 80 mg/m² on Days 1, 8, and 15 for 6 cycles followed by placebo 500 mg intravenously every 4 weeks.
Treatment with retifanlimab continued until disease progression, unacceptable toxicity, death, or withdrawal of consent, for up to 12 months. Tumour response assessments were performed every 8 weeks throughout the treatment period. Patients who received placebo in combination with carboplatin and paclitaxel and who experienced documented disease progression (verified by blinded independent central review (BICR)), had the option of receiving retifanlimab 500 mg monotherapy in a crossover treatment period.
Among the 308 patients enrolled, the median age was 62 years (range, 29–86 years) with 31 (10.1%) age 75 or older; 27.9% of patients were male, 87.3% of patients were Caucasian and the ECOG performance status was 0 (54.5%) or 1 (45.1%). Seventy-one percent of patients were reported to have had prior radiotherapy and 34.7% had prior surgery. Four percent of patients were HIV-positive. Eighty-three percent of patients had metastatic disease at baseline. PD-L1 expression of ≥1% was present in 91% of tumours.
The primary efficacy endpoint was progression-free survival (PFS) as assessed by a BICR according to Response Evaluation Criteria in Solid Tumors (RECIST) v1.1 and the key secondary endpoint was overall survival (OS). Additional secondary efficacy endpoints included objective response rate (ORR) and duration of response (DOR).
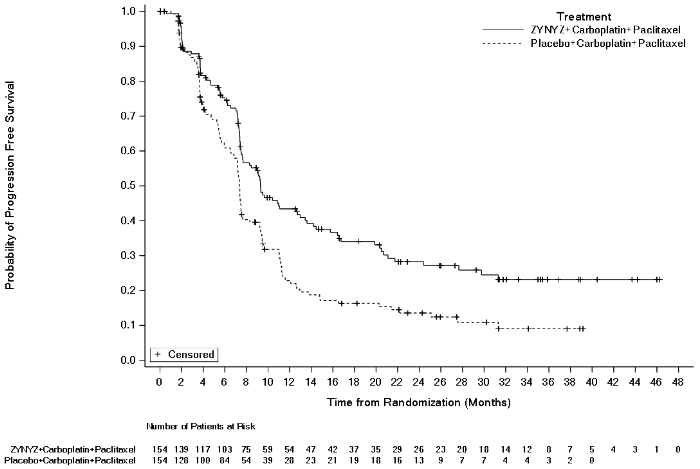
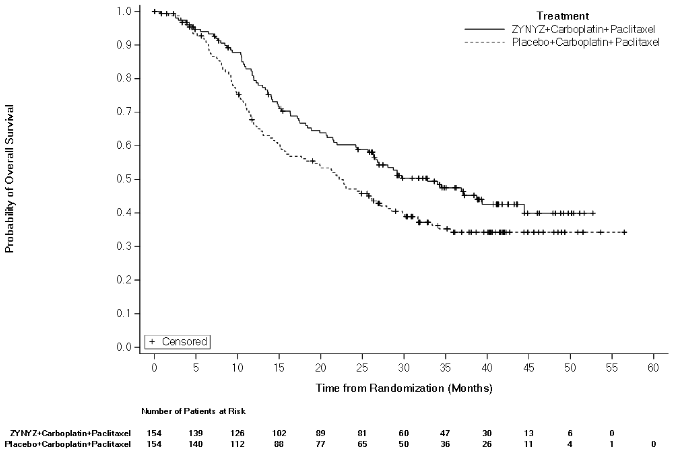
Efficacy results are summarised in Table 3, Figure 1, and Figure 2.
Table 3. Efficacy results in POD1UM-303/InterAACT-2 for patients with metastatic or inoperable locally recurrent SCAC:
| Endpoint | ZYNYZ in combination with carboplatin and paclitaxel (N=154) | Placebo in combination with carboplatin and paclitaxel (N=154) |
| Progression-free survivala,b | ||
| Events, n (%) | 92 (59.7) | 110 (71.4) |
| Median in months (95% CI) | 9.3 (7.5, 11.3) | 7.4 (7.1, 7.7) |
| Hazard ratio (95% CI) | 0.63 (0.47, 0.84) | |
| p-valuec | 0.0013 | |
| Overall survivald | ||
| Deaths, n (%) | 78 (50.6) | 94 (61.0) |
| Median in months (95% CI) | 32.8 (25.7, 44.5) | 22.2 (15.7, 27.2) |
| Hazard ratio (95% CI) | 0.75 (0.55, 1.01) | |
| Objective response ratea | ||
| Objective response rate (95% CI) | 55.8% (47.6, 63.8) | 44.2% (36.2, 52.4) |
CI = confidence interval.
a Based on primary analysis.
b Median duration of follow-up for PFS: ZYNYZ in combination with carboplatin and paclitaxel = 7.6 months (range, 0 – 33.9 months); placebo in combination with carboplatin and paclitaxel = 7.1 months (range, 0 – 27.4 months).
c Two-sided p-value based on the stratified log-rank test.
d Based on final analysis; OS did not reach the prespecified threshold for statistical significance.
Figure 1. Kaplan-Meier curve for PFS in POD1UM-303/InterAACT-2 (Final Analysis):
Figure 2. Kaplan-Meier curve for OS in POD1UM-303/InterAACT-2 (Final Analysis):
Merkel cell carcinoma (MCC)
The efficacy and safety of retifanlimab was studied in the POD1UM-201 study, an open-label, single-arm, multiregional study that enrolled patients with metastatic or recurrent locally advanced MCC who had not received prior systemic therapy for their advanced disease. Patients with active autoimmune disease or a medical condition that required immunosuppression, severe hepatic or renal impairment, clinically significant cardiac disease, history of organ transplant, or Eastern Cooperative Oncology Group (ECOG) performance score (PS) ≥2 were ineligible. Patients who were HIV-positive, with an undetectable viral load, a CD4+ count ≥300 cells/microliter and receiving antiretroviral therapy were eligible.
Patients received retifanlimab 500 mg every 4 weeks until disease progression or unacceptable toxicity for a maximum of 2 years. Assessment of efficacy was performed every 8 weeks for the first year of therapy and 12 weeks thereafter. The major efficacy outcome measure of confirmed objective response rate, and duration of response were assessed by an independent central review committee according to Response Evaluation Criteria in Solid Tumours (RECIST) v1.1. All ongoing responses were followed for a minimum of 12 months.
A total of 101 patients were analysed for efficacy. The median age of enrolled patients was 71.1 years (range, 38 - 90 years) with 39 (39%) age 75 or older; 67.3% of patients were male, all but one patient were Caucasian and the Eastern Cooperative Oncology Group performance status was 0 (73.3%) or 1 (26.7%). Thirty-seven percent of patients were reported to have had prior radiotherapy and 68.3% had prior surgery. Ninety percent of patients had metastatic disease. One patient was HIV-positive. The majority of tumour samples tested (72.3%) were positive for Merkel cell polyomavirus (MCPyV).
Efficacy results are summarized in Table 4. The median duration of treatment was 10.3 months (range, 1 day – 24.8 months).
Table 4. Efficacy results in POD1UM-201 study for patients with metastatic or recurrent locally advanced MCC:
| Endpoint | ZYNYZ (N=101) |
|---|---|
| Objective response rate | |
| Objective response rate (95% CI) | 53.5% (43.3, 63.5) |
| Complete response | 16.8% |
| Partial response | 36.6% |
| Duration of response | |
| Median in months (95% CI) | 25.3 (14.2, NE) |
| Minimum, maximum (months) | 1.1, 38.7+ |
CI = confidence interval; NE = not estimable; + denotes ongoing response. Median duration of follow-up: 17.6 months (range, 1.1 – 38.7 months).
Efficacy and PD-L1/MCPyV status
Clinical activity was observed regardless of PD-L1 or MCPyV status. Table 5 summarises the objective response rates by tumour PD-L1 expression and MCPyV status of chemotherapy-naïve MCC patients with central biomarker results in the POD1UM-201 study.
Table 5. Objective response rates by tumour PD-L1 expression and MCPyV status:
| ZYNYZ Objective response rates (95% CI) N=101 | |
|---|---|
| PD-L1 expressiona at cut-off of ≥1% | |
| Positive (n=83) | 57.8% (46.5, 68.6) |
| Negative or missing (n=18) | 33.3% (13.3, 59.0) |
| MCPyV status | |
| Positive (n=73) | 52.1% (40, 63.9) |
| Negative, equivocal, or missing (n=28) | 57.1% (37.2, 75.5) |
MCPyV = Merkel cell polyomavirus.
a PD-L1 expression was determined by IHC using Combined Positive Score (CPS) interpretation.
Elderly population
Squamous cell carcinoma of the anal canal (SCAC)
Of the 154 patients treated with retifanlimab in combination with carboplatin and paclitaxel in the efficacy population in POD1UM-303/InterAACT-2, 37.7% (58/154) were 65 years or older, and 9.1% (14/154) were 75 years or older. No overall differences in efficacy were observed between elderly patients and younger patients treated with retifanlimab in combination with carboplatin and paclitaxel.
Merkel cell carcinoma (MCC)
Of the 101 patients treated with retifanlimab in the efficacy population in POD1UM-201, 76.2% (77/101) were 65 years or older, and 38.6% (39/101) were 75 years or older. Objective response rates in these age groups were 55.8% (95% CI: 44.1, 67.2) and 48.7% (95% CI: 32.4, 65.2), respectively.
Paediatric population
The European Medicines Agency has waived the obligation to submit the results of studies with ZYNYZ in all subsets of the paediatric population for the treatment of MCC and SCAC. See 4.2 for information on paediatric use.
5.2. Pharmacokinetic properties
The pharmacokinetics (PK) of retifanlimab were characterised using a population pharmacokinetics analysis with concentration data collected from 788 patients with various cancers who received retifanlimab doses of 1, 3, 10 mg/kg every 2 weeks, 375 mg every 3 weeks, or 3 mg/kg, 10 mg/kg, 500 mg and 750 mg every 4 weeks. The AUC was dose proportional in the studied dose range. The geometric mean (CV%) of Cmax and AUC at steady state for the recommended 500 mg every 4 weeks dose were 197 mg/L (25.4%) and 2270 day*mg/L (35.1%).
Distribution
The geometric mean value (CV%) for volume of distribution at steady state is 6 L (19.8%).
Biotransformation
The metabolic route of retifanlimab has not been characterised. Retifanlimab is expected to be catabolised through protein degradation processes.
Elimination
The geometric mean (CV%) clearance of retifanlimab after the first dose was 0.301 L/day (38.3%) and decreased over time by 22.9%, resulting in a steady-state clearance of 0.232 L/day (35.7%). For the recommended 500 mg every 4 weeks dose, half-life is 15.6 days (31.5%) and 19.8 days (29.9%) after first-dose and at steady-state, respectively.
Special Populations
The following factors are not expected to have clinically important effects on the pharmacokinetics of retifanlimab: age (range: 18 to 94 years), weight (33 to 133 kg), sex, race, or tumour burden.
Renal impairment
The effect of renal impairment on the clearance of retifanlimab was evaluated by population pharmacokinetic analyses in patients with mild (n=354) or moderate (n=151) renal impairment (eGFR between 89 and 30 mL/min/1.73m²; n=505) compared to patients with normal renal function (eGFR ≥ 90 mL/min/1.73m²; n=263). No clinically important differences were found in the clearance of retifanlimab. There are limited data in patients with severe renal impairment (n=4, lowest eGFR 26.0 mL/min/1.73m²). Retifanlimab has not been studied in patients with end-stage renal disease.
Hepatic impairment
The effect of hepatic impairment on the clearance of retifanlimab was evaluated by population pharmacokinetic analyses in patients with mild (n=93; TB > ULN to 1.5 ULN or AST > ULN) hepatic impairment compared to patients with normal (n=692; TB and AST ≤ ULN) hepatic function. No clinically important differences were found in the clearance of retifanlimab. There are limited data in patients with moderate (n=1; TB between 1.5 and 3.0 times ULN and any AST) hepatic impairment. Retifanlimab has not been studied in patients with severe (TB between 3.0 and 10 times ULN and any AST) hepatic impairment.
5.3. Preclinical safety data
No findings of toxicological significance were observed in monkeys in studies of up to 13 weeks duration at exposures sufficiently in excess compared to the clinical exposure at the recommended dose of 500 mg retifanlimab every 4 weeks.
No studies have been performed to assess the potential of retifanlimab for carcinogenicity or genotoxicity.
Animal reproduction and development toxicity studies have not been conducted with retifanlimab. A central function of the PD-1/PD-L1 pathway is to preserve pregnancy by maintaining maternal immune tolerance to the foetus. In murine models of pregnancy, blockade of PD-L1 signaling has been shown to disrupt tolerance to the foetus and to result in an increase in foetal loss; therefore, potential risks of administering retifanlimab during pregnancy include increased rates of abortion or stillbirth. As reported in the literature, there were no malformations related to the blockade of PD-1/PD-L1 signaling in the offspring of these animals; however, immune-mediated disorders occurred in PD-1 and PD-L1 knockout mice. Based on its mechanism of action, foetal exposure to retifanlimab may increase the risk of developing immune-mediated disorders or altering the normal immune response.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.