DUVYZAT Oral suspension Ref.[115472] Active ingredients: Givinostat
Source: European Medicines Agency (EU) Revision Year: 2025 Publisher: Italfarmaco S.p.A., Viale F. Testi, 330, 20126 Milano, Italy, Tel: +39 02 64431, info@italfarmacogroup.com
5.1. Pharmacodynamic properties
Pharmacotherapeutic group: Other drugs for disorders of the musculoskeletal system
ATC code: M09AX14
Mechanism of action
Givinostat is a class I and II histone deacetylase (HDAC) inhibitor that modulates the uncontrolled HDAC activity in dystrophic muscles, which contributes to the pathology of Duchenne muscular dystrophy (DMD).
Givinostat HDAC inhibition has been shown to reduce muscle fiber damage, chronic muscular inflammation, fibrosis, fat deposition, and to promote mitochondrial biogenesis.
Givinostat mechanism of action is independent of the underlying dystrophin gene mutation causing the disease.
Muscle fat fraction as assessed by MR spectroscopy
The percentage of fat fraction present in the vastus lateralis muscles (VLM) of the thigh was measured in EPIDYS Study using magnetic resonance spectroscopy. At 18 months, for the patients with baseline VLM fat fraction in the range of >5% to ≤30%, a LS mean increase of VLM fat fraction was 7.63% in the Duvyzat-treated patients compared to a 10.56% increase in patients who received placebo.
Clinical efficacy and safety
The safety and efficacy of Duvyzat in DMD patients were assessed in EPIDYS study. EPIDYS was a 18 months, 2:1 randomised, double-blind, placebo-controlled phase 3 study of 179 ambulant DMD patients aged 6 years or older. Givinostat or placebo were administered in addition to a stable dose of corticosteroids throughout the study. Patients were recruited into 2 groups:
- Group A (120 Patients): subjects with a baseline VL MFF in the range >5% and ≤30%, as assessed by MRS.
- Group B (59 Patients): subjects with baseline VL MFF outside of the above range (other criteria were the same).
A weight-based dose treatment regimen was applied. The starting dose was initially 17.7–62 mg oral givinostat twice a day, with a reduced dose of 11.8–41.4 mg twice a day. The protocol was then amended to reduce the starting dose for new participants, to 11.8–41.4 mg twice a day, allowing a further dose reduction of 9.4–33.1 mg twice a day.
The primary endpoint in Group A (pre-specified primary analysis population) was the change of time to complete 4 stairs climb (4SC) at 18 months.
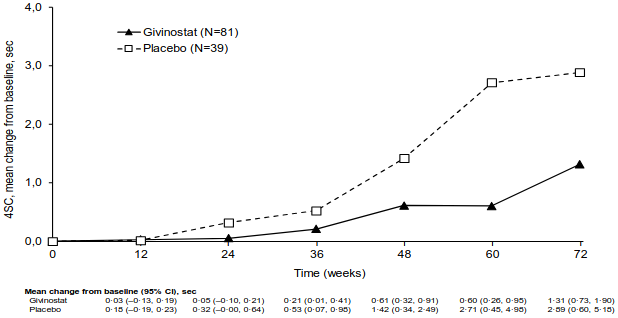
The primary endpoint was met, givinostat significantly (p=0.035) reduced the decline in 4SC compared to placebo based on the prespecified log scale analysis (Table 4). When the results were analysed in non-log scale, the mean 4SC increased by 1.25 seconds in the givinostat group vs 3.03 seconds in the placebo group (see Table 4). Therefore, the treatment effect (change from baseline, givinostat minus placebo) was -1.78 seconds (p=0.037).
Table 4. EPIDYS Study: Time (Seconds) to 4SC, Change from Baseline to 18 Months (Group A):
| Time to 4SC | Givinostat§ (N=81) | Placebo§ (N=39) |
|---|---|---|
| Log Scale Analysis* | ||
| GLS mean (log scale SE) (95% CI) | 1.27 (0.040) (1.172, 1.372) | 1.48 (0.058) (1.317, 1.657) |
| GLS mean ratio (givinostat/placebo) (log scale SE) (95% CI) p-value | 0.86 (0.071) (0.745, 0.989) 0.0345 | |
| No Log Scale Analysis | ||
| LS mean (95% CI) | 1.25 (0.311, 2.181) | 3.03 (1.666, 4.394) |
| Difference in LS means (givinostat-placebo) (95% CI) p-value | -1.78 (-3.462, -0.106) 0.0374 | |
* Log Scale analysis was performed since data were not normally distributed.
§ Givinostat or placebo were administered in addition to a stable dose of corticosteroids throughout the study.
Note: LS Means, CIs, and p-values are obtained from an analysis of covariance (ANCOVA) model on change from baseline in 4SC at Month 18.
GLSmean change from baseline should be interpreted as a rate change (EOS/baseline).
Figure 1 describes the observed mean time to 4SC during the 72 weeks of treatment in the two groups.
Figure 1. Study 48: Observed Mean Change in Seconds to 4SC by Treatment Over Time (Group A):
* Givinostat or placebo were administered in addition to a stable dose of corticosteroids throughout the study.
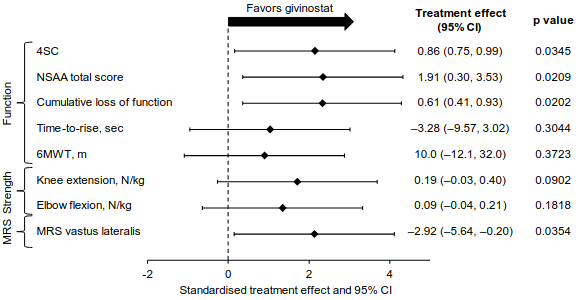
The key secondary efficacy endpoints in Group A were the change from baseline to 18 months in physical function assessed by North Star Ambulatory Assessment (NSAA), time to rise from floor (TTR); distance walked in 6 minutes (6MWT); muscle strength evaluated by knee extension and elbow flexion as measured by hand-held myometry (HHM); and fat fraction of vastus lateralis muscles evaluated by Magnetic Resonance Spectroscopy (MRS) technique. Overall, the results of the key secondary endpoints assessing function, strength and muscle morphology did not reach formal statistical significance based on the pre-specified Hochberg analysis, however all outcomes were in favour of givinostat (Figure 2).
Figure 2. EPIDYS Study: Primary and Key Secondary Efficacy Endpoints of Givinostat Versus Placebo (Group A)§:
§ Givinostat or placebo were administered in addition to a stable dose of corticosteroids throughout the study.
Givinostat long term safety, tolerability and efficacy are evaluated in an ongoing prospective open label, long-term extension (OLE) study named STUDY 51. Patients who completed the givinostat phase 2 trial (STUDY 43) and patients who completed the givinostat phase 3 trials (EPIDYS) were enrolled in STUDY 51. Additionally, 30 givinostat naïve patients were also enrolled in the OLE cohort. In total, 207 male patients were enrolled and receive givinostat with a weight-based dose regimen that ranges from 9.4 mg twice daily to 62 mg twice daily. All patients were on a stable dose of corticosteroids before enrolling and continue corticosteroid treatment throughout the study.
The benefit/risk of givinostat in the absence of concomitant corticosteroid treatment in DMD patients has not been determined.
The benefit/risk of givinostat in non-ambulatory patients has not been determined.
Paediatric population
The European Medicines Agency has deferred the obligation to submit the results of studies with Duvyzat in one or more subsets of the paediatric population in DMD.
This medicinal product has been authorised under a so-called 'conditional approval' scheme. This means that further evidence on this medicinal product is awaited. The European Medicines Agency will review new information on this medicinal product at least every year and this SmPC will be updated as necessary.
5.2. Pharmacokinetic properties
Absorption
Givinostat is well absorbed after oral administration. Mean plasma concentrations increase in a dose-proportional manner, and maximum plasma concentrations are achieved about 2-3 hours after administration. A high fat standard meal resulted in some increase in the exposure (about 30% increase in area under the plasma concentration-time curve [AUC] and about 20% increase in maximum plasma concentration [Cmax]) and a delay in time to maximum concentration (Tmax) from 2 to 3 hours. Steady-state concentrations are achieved within 5 to 7 days after both once a day and twice a day dosing. A moderate accumulation of less than 2-fold was observed after twice daily administration.
A physiologically based pharmacokinetic analysis, including healthy volunteer data, predicted an oral bioavailability in humans ≥50% after single oral administration at the dose range of 44.3 to 177.2 mg.
Distribution
Givinostat is approximately 96% bound to human plasma proteins and is slightly partitioned into red blood cells (blood to plasma ratio = 1.3).
Biotransformation
In vitro studies with human enzymatic preparations together with animal metabolism in vitro and in vivo showed that givinostat is extensively metabolised forming several metabolites. CYP450 and UGTs are not involved in the main metabolic reactions. The enzymes forming the primary metabolites have only been partially identified. Four major metabolites, which are inactive, have been characterized in humans and animal species, although with differences in quantitative amounts.
Elimination
In plasma, givinostat displays a bi-phasic elimination profile with a mean apparent terminal elimination phase (half-life) of about 6 hours. The elimination of givinostat is likely dependent on metabolism followed by renal and biliary excretion. Urinary excretion of givinostat and the main metabolites in humans has been evaluated in healthy volunteers after single and repeated doses of givinostat. The percentage of unchanged givinostat recovered in urine was very low after both single and repeated twice daily administration (<3% of the dose).
Linearity/non-linearity
The pharmacokinetics of givinostat is linear, since the AUC∞ obtained after single administration is comparable to that with repeated once daily administration, with a possible minimal apparent accumulation of active substance over time (range of accumulation ratios found 1.0-1.7). Linearity was tested after single administration of doses 44.3 to 354.4 mg and multiple administration of doses 44.3 to 177.2 mg.
Weight
Based on the population PK analyses, weight resulted to significantly affect givinostat clearance. The effect is not linear, i.e., the effect is larger at smaller weights and smaller in weights 30 kg and above. Thus, a weight-based dose is recommended.
Characteristics in specific groups
The population PK analyses show that age or co-administration with corticosteroids has no effects on the pharmacokinetics of givinostat. The pharmacokinetics of givinostat have been evaluated in male paediatric DMD patients from 6 years old.
Hepatic impairment
Givinostat has not been studied in patients with hepatic impairment. Caution should be exercised in the administration and monitoring of the product in these patients.
Renal impairment
Givinostat has not been studied in patients with renal impairment. However, renal impairment is not expected to impact the exposure of givinostat because renal excretion is not a significant route of givinostat elimination.
5.3. Preclinical safety data
In repeated dose oral toxicity studies in rats and monkeys, a dose dependent decrease in white blood counts with related atrophy of lymphoid organs (thymus, lymph nodes and spleen), in red blood cell and platelet counts, and in cellularity in the bone marrow was seen with givinostat. An increase in liver enzymes was also observed. In monkeys, bile duct hyperplasia was additionally induced. These toxicities were generally reversible upon drug discontinuation, but developed at lower givinostat exposures in animals than achieved at the maximum recommended human dose (MRHD).
Genotoxicity and carcinogenicity
Givinostat was positive for frameshift mutations at high doses in vitro in bacteria (Ames test), negative in mammalian cells (TK+/- in mouse lymphoma cells), and negative in vivo in transgenic BigBlue rats and in the Pig-a locus.
In conclusion givinostat does not pose a relevant genotoxic potential in vivo.
No data from carcinogenicity studies with givinostat are currently available.
Reproductive and developmental toxicity
Givinostat caused dose-dependent decreases in size and weight of male accessory organs already starting at the lowest dose. Mid and high dose animals showed an increase of the pre-coital interval and lower amounts of copulation plugs probably resulting from disturbance on ejaculate formation. However, sperm parameters and number of pregnant females were not affected.
Maternal adverse effects were observed at the high dose levels in the embryo-foetal and in the pre- and postnatal development toxicity studies. Effects on gestation, embryo-foetal development and litter parameters were considered secondary to maternal toxicity. However, effects on embryo-fetal development and litter parameters were already observed at mid dose levels in the rat and rabbit embryo-fetal development study as well as in the low dose group of the pre-/postnatal development study. There was no adverse effect on offspring behaviour, neurological growth, sexual maturation and reproductive function.
Overall, effects on reproductive toxicity were observed at lower givinostat exposures in animals than achieved at the MRHD, except for the embryo-foetal development study in rabbits with a safety margin of about 10 towards human exposure at the MRHD.
Juvenile toxicity
In rats, some effects were observed on haematological parameters and lymphoid organs at the high dose levels, which were fully or partially reversible. These effects were observed at lower givinostat exposures in animals than achieved at the MRHD. No treatment-related effects on animal growth, sexual maturation, reproductive performances, and in neurobehavioural development were observed.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.