EDURANT Film-coated tablet Ref.[8790] Active ingredients: Rilpivirine
Source: European Medicines Agency (EU) Revision Year: 2024 Publisher: Janssen-Cilag International NV, Turnhoutseweg 30, B-2340 Beerse, Belgium
Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in section 6.1.
EDURANT should not be co-administered with the following medicinal products, as significant decreases in rilpivirine plasma concentrations may occur (due to CYP3A enzyme induction or gastric pH increase), which may result in loss of therapeutic effect of EDURANT (see section 4.5):
- the anticonvulsants carbamazepine, oxcarbazepine, phenobarbital, phenytoin
- the antimycobacterials rifampicin, rifapentine
- proton pump inhibitors, such as omeprazole, esomeprazole, lansoprazole, pantoprazole, rabeprazole
- the systemic glucocorticoid dexamethasone, except as a single dose treatment
- St John’s wort (Hypericum perforatum).
Special warnings and precautions for use
Virologic failure and development of resistance
EDURANT has not been evaluated in patients with previous virologic failure to any other antiretroviral therapy. The list of rilpivirine resistance-associated mutations presented in section 5.1 should only guide the use of EDURANT in the treatment-naïve population.
In the pooled efficacy analysis from the phase 3 trials TMC278-C209 (ECHO) and TMC278-C215 (THRIVE) in adults through 96 weeks, patients treated with rilpivirine with a baseline viral load >100,000 HIV-1 RNA copies/ml had a greater risk of virologic failure (18.2% with rilpivirine versus 7.9% with efavirenz) compared to patients with a baseline viral load ≤100,000 HIV-1 RNA copies/ml (5.7% with rilpivirine versus 3.6% with efavirenz). The greater risk of virologic failure for patients in the rilpivirine arm was observed in the first 48 weeks of these trials (see section 5.1). Patients with a baseline viral load >100,000 HIV-1 RNA copies/ml who experienced virologic failure exhibited a higher rate of treatment-emergent resistance to the non-nucleoside reverse transcriptase inhibitor (NNRTI) class. More patients who failed virologically on rilpivirine than who failed virologically on efavirenz developed lamivudine/emtricitabine associated resistance (see section 5.1).
Findings in adolescents and paediatric patients in trial TMC278-C213 were generally in line with these data. No virological failures were observed in trial TMC278HTX2002 (for details see section 5.1).
Only patients deemed likely to have good adherence to antiretroviral therapy should be treated with rilpivirine, as suboptimal adherence can lead to development of resistance and the loss of future treatment options.
As with other antiretroviral medicinal products, resistance testing should guide the use of rilpivirine (see section 5.1).
Cardiovascular
At supra-therapeutic doses (75 and 300 mg once daily), rilpivirine has been associated with prolongation of the QTc interval of the electrocardiogram (ECG) (see sections 4.5, 4.8 and 5.2). EDURANT at the recommended dose of 25 mg once daily is not associated with a clinically relevant effect on QTc. EDURANT should be used with caution when co-administered with medicinal products with a known risk of Torsade de Pointes.
Immune reactivation syndrome
In HIV infected patients with severe immune deficiency at the time of initiation of CART, an inflammatory reaction to asymptomatic or residual opportunistic pathogens may arise and cause serious clinical conditions or aggravation of symptoms. Typically, such reactions have been observed within the first weeks or months of initiation of CART. Relevant examples are cytomegalovirus retinitis, generalised and/or focal mycobacterial infections and Pneumocystis jiroveci pneumonia. Any inflammatory symptoms should be evaluated and treatment instituted when necessary.
Autoimmune disorders (such as Graves' disease and autoimmune hepatitis) have also been reported to occur in the setting of immune reactivation; however, the reported time to onset is more variable and these events can occur many months after initiation of treatment (see section 4.8).
Pregnancy
EDURANT should be used during pregnancy only if the potential benefit justifies the potential risk. Lower exposures of rilpivirine were observed when rilpivirine 25 mg once daily was taken during pregnancy. In the phase 3 studies, lower rilpivirine exposure, similar to that seen during pregnancy, has been associated with an increased risk of virological failure, therefore viral load should be monitored closely (see sections 4.6, 5.1 and 5.2). Alternatively, switching to another ART regimen could be considered.
Important information about some of the ingredients of EDURANT
EDURANT contains lactose. Patients with rare hereditary problems of galactose intolerance, total lactase deficiency, or glucose-galactose malabsorption should not take this medicinal product.
Interaction with other medicinal products and other forms of interaction
Medicinal products that affect rilpivirine exposure
Rilpivirine is primarily metabolised by cytochrome P450 (CYP)3A. Medicinal products that induce or inhibit CYP3A may thus affect the clearance of rilpivirine (see section 5.2). Co-administration of rilpivirine and medicinal products that induce CYP3A has been observed to decrease the plasma concentrations of rilpivirine, which could reduce the therapeutic effect of rilpivirine.
Co-administration of rilpivirine and medicinal products that inhibit CYP3A has been observed to increase the plasma concentrations of rilpivirine.
Co-administration of rilpivirine with medicinal products that increase gastric pH may result in decreased plasma concentrations of rilpivirine which could potentially reduce the therapeutic effect of EDURANT.
Medicinal products that are affected by the use of rilpivirine
Rilpivirine at a dose of 25 mg once daily is not likely to have a clinically relevant effect on the exposure of medicinal products metabolised by CYP enzymes. Rilpivirine inhibits P-glycoprotein in vitro (IC50 is 9.2 μM). In a clinical study, rilpivirine did not significantly affect the pharmacokinetics of digoxin. However, it may not be completely excluded that rilpivirine can increase the exposure to other medicines transported by P-glycoprotein that are more sensitive to intestinal P-gp inhibition, e.g. dabigatran etexilate.
Rilpivirine is an in vitro inhibitor of the transporter MATE-2K with an IC50 of <2.7 nM. The clinical implications of this finding are currently unknown.
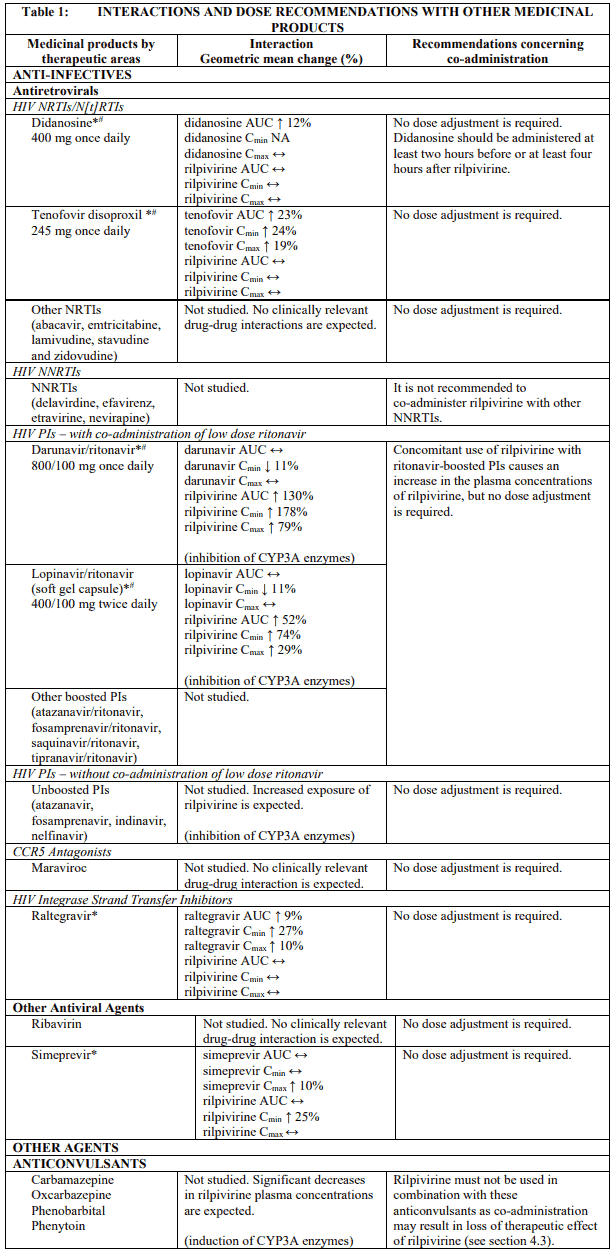
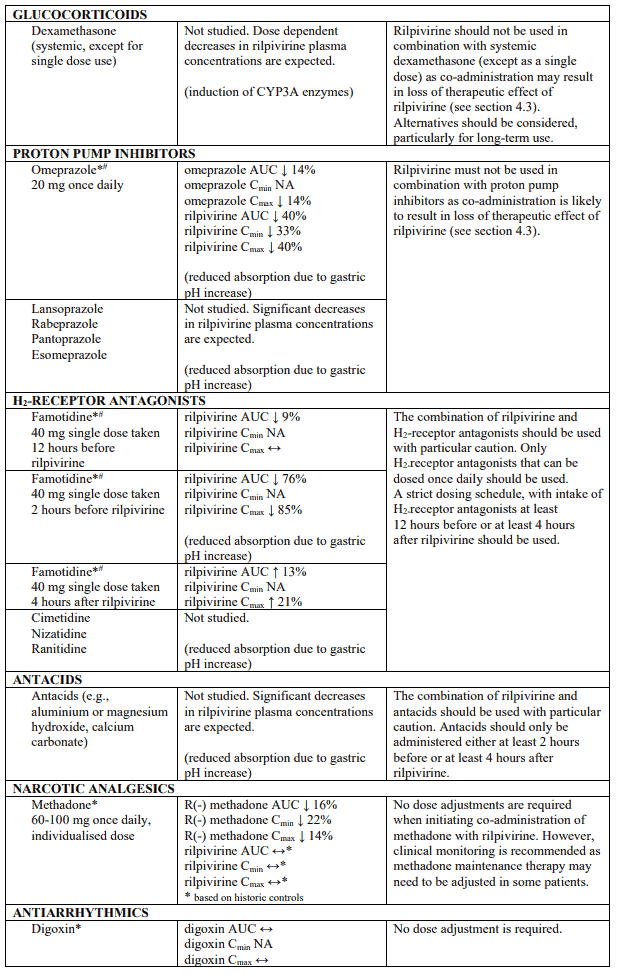
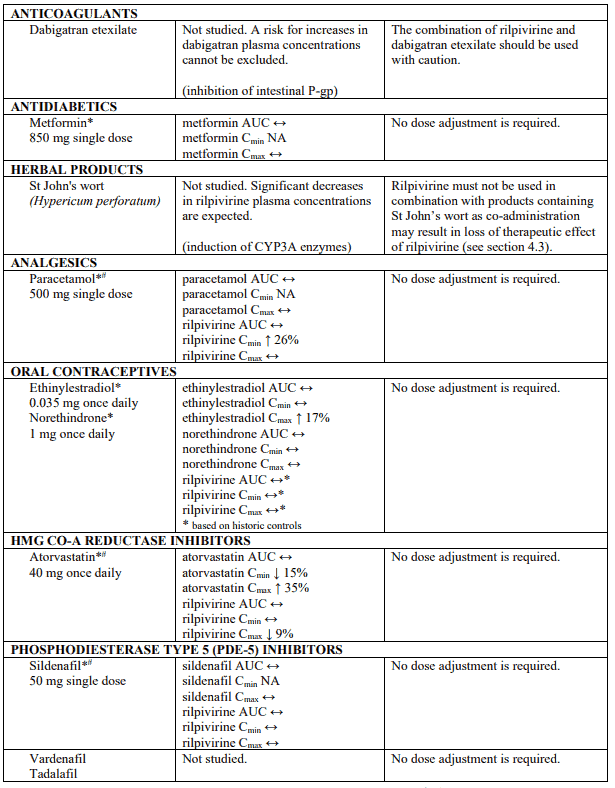
Established and theoretical interactions with selected antiretrovirals and non-antiretroviral medicinal products are listed in table 1.
Interaction table
Interaction studies have only been performed in adults.
Interactions between rilpivirine and co-administered medicinal products are listed in table 1 (increase is indicated as "↑", decrease as "↓", no change as "↔", not applicable as "NA", confidence interval as "CI").
* The interaction between rilpivirine and the medicinal product was evaluated in a clinical study. All other drug-drug interactions shown are predicted.
# This interaction study has been performed with a dose higher than the recommended dose for rilpivirine assessing the maximal effect on the co-administered medicinal product. The dosing recommendation is applicable to the recommended dose of rilpivirine of 25 mg once daily.
† This interaction study has been performed with a dose higher than the recommended dose for rilpivirine.
QT prolonging medicinal products
There is limited information available on the potential for a pharmacodynamic interaction between rilpivirine and medicinal products that prolong the QTc interval of the ECG. In a study of healthy subjects, supratherapeutic doses of rilpivirine (75 mg once daily and 300 mg once daily) have been shown to prolong the QTc interval of the ECG (see section 5.1). EDURANT should be used with caution when co-administered with a medicinal product with a known risk of Torsade de Pointes.
Fertility, pregnancy and lactation
Pregnancy
A moderate amount of data on pregnant women (between 300-1000 pregnancy outcomes) indicate no malformative or feto/neonatal toxicity of rilpivirine (see sections 4.4, 5.1 and 5.2). Lower exposures of rilpivirine were observed during pregnancy, therefore viral load should be monitored closely.
Animal studies do not indicate reproductive toxicity (see section 5.3).
The use of rilpivirine may be considered during pregnancy, if necessary.
Breast-feeding
It is not known whether rilpivirine is excreted in human milk. Rilpivirine is excreted in the milk of rats. Because of the potential for adverse reactions in breastfed infants, mothers should be instructed not to breast-feed if they are receiving rilpivirine.
In order to avoid transmission of HIV to the infant it is recommended that women living with HIV do not breast-feed.
Fertility
No human data on the effect of rilpivirine on fertility are available. No clinically relevant effects on fertility were seen in animal studies (see section 5.3).
Effects on ability to drive and use machines
EDURANT has no or negligible influence on the ability to drive and use machines. However, fatigue, dizziness and somnolence have been reported in some patients taking EDURANT and should be considered when assessing a patient's ability to drive or operate machinery.
Undesirable effects
Summary of the safety profile
During the clinical development program (1,368 patients in the Phase III controlled trials TMC278-C209 (ECHO) and TMC278-C215 (THRIVE)), 55.7% of subjects experienced at least one adverse drug reaction (see section 5.1). The most frequently reported adverse drug reactions (ADRs) (≥2%) that were at least of moderate intensity were depression (4.1%), headache (3.5%), insomnia (3.5%), rash (2.3%), and abdominal pain (2.0%). The most frequent serious treatment-related ADRs were reported in 7 (1.0%) patients receiving rilpivirine. The median duration of exposure for patients in the rilpivirine arm and efavirenz arm was 104.3 and 104.1 weeks, respectively. Most ADRs occurred in the first 48 weeks of treatment.
Selected treatment emergent clinical laboratory abnormalities (grade 3 or grade 4), considered as ADRs, reported in EDURANT treated patients were increased pancreatic amylase (3.8%), increased AST (2.3%), increased ALT (1.6%), increased LDL cholesterol (fasted, 1.5%), decreased white blood cell count (1.2%), increased lipase (0.9%), increased bilirubin (0.7%), increased triglycerides (fasted, 0.6%), decreased haemoglobin (0.1%), decreased platelet count (0.1%), and increased total cholesterol (fasted, 0.1%).
Tabulated summary of adverse reactions
ADRs reported in adult patients treated with rilpivirine are summarised in Table 2. The ADRs are listed by system organ class (SOC) and frequency. Frequencies are defined as very common (≥1/10), common (≥1/100 to <1/10) and uncommon (≥1/1,000 to <1/100). Within each frequency grouping, ADRs are presented in order of decreasing frequency.
Table 2. ADRs reported in antiretroviral treatment-naïve HIV-1 infected adult patients treated with Rilpivirine (pooled data from the week 96 analysis of the phase 3 ECHO and THRIVE trials) N=686:
| System Organ Class (SOC) | Frequency Category | ADRs (Rilpivirine + BR) |
|---|---|---|
| Blood and lymphatic system disorders | common | decreased white blood cell count decreased haemoglobin decreased platelet count |
| Immune system disorders | uncommon | immune reactivation syndrome |
| Metabolism and nutrition disorders | very common | increased total cholesterol (fasted) increased LDL cholesterol (fasted) |
| common | decreased appetite increased triglycerides (fasted) | |
| Psychiatric disorders | very common | insomnia |
| common | abnormal dreams depression sleep disorders depressed mood | |
| Nervous system disorders | very common | headache dizziness |
| common | somnolence | |
| Gastrointestinal disorders | very common | nausea increased pancreatic amylase |
| common | abdominal pain vomiting increased lipase abdominal discomfort dry mouth | |
| Hepatobiliary disorders | very common | increased transaminases |
| common | increased bilirubin | |
| Skin and subcutaneous tissue disorders | common | rash |
| General disorders and administration site conditions | common | fatigue |
BR = background regimen
N = number of subjects
Laboratory abnormalities
In the rilpivirine arm in the week 96 analysis of the phase 3 ECHO and THRIVE trials, mean change from baseline in total cholesterol (fasted) was 5 mg/dl, in HDL cholesterol (fasted) 4 mg/dl, in LDL cholesterol (fasted) 1 mg/dl, and in triglycerides (fasted) -7 mg/dl.
Description of selected adverse reactions
Immune reactivation syndrome
In HIV infected patients with severe immune deficiency at the time of initiation of combination antiretroviral therapy (CART), an inflammatory reaction to asymptomatic or residual opportunistic infections may arise. Autoimmune disorders (such as Graves' disease and autoimmune hepatitis) have also been reported; however, the reported time to onset is more variable and these events can occur many months after initiation of treatment (see section 4.4).
Paediatric population (12 to less than 18 years of age)
TMC278-C213 Cohort 1
The safety assessment is based on the week 48 analysis of the single-arm, open-label, phase 2 trial, TMC278-C213 Cohort 1, in which 36 antiretroviral treatment-naïve HIV-1 infected adolescent patients weighing at least 32 kg received rilpivirine (25 mg once daily) in combination with other antiretroviral agents (see section 5.1). The median duration of exposure for patients was 63.5 weeks. There were no patients who discontinued treatment due to ADRs. No new ADRs were identified compared to those seen in adults.
Most ADRs were grade 1 or 2. The most common ADRs reported in Study TMC278-C213 Cohort 1 (all grades, greater than or equal to 10%) were headache (19.4%), depression (19.4%), somnolence (13.9%), and nausea (11.1%). No grade 3-4 laboratory abnormalities for AST/ALT or grade 3-4 ADRs of transaminase increased were reported.
There were no new safety concerns identified in the week 240 analysis of the TMC278-C213 Cohort 1 trial in adolescents.
Paediatric population (2 to less than 12 years of age)
TMC278-C213 Cohort 2
Cohort 2 of the single-arm, open-label phase 2 trial, TMC278-C213 was designed to evaluate the safety of the rilpivirine weight adjusted doses 12.5, 15, and 25 mg once daily in antiretroviral treatment-naïve HIV-1 infected patients (6 to less than 12 years of age and weighing at least 17 kg) (see section 5.1). The median duration of exposure for patients in the week 48 analysis (including post-week 48 extension) was 69.5 (range 35 to 218) weeks.
All ADRs were mild or moderate. ADRs reported in at least 2 participants, regardless of severity, were: decreased appetite (3/18, 16.7%), vomiting (2/18, 11.1%), ALT increased (2/18, 11.1%), AST increased (2/18, 11.1%), and rash (2/18, 11.1%). There were no patients who discontinued treatment due to ADRs. No new ADRs were identified compared to those seen in adults.
TMC278HTX2002
The single arm, open-label phase 2 trial, TMC278HTX2002, was designed to evaluate the safety of rilpivirine weight-adjusted doses 12.5, 15, and 25 mg once daily in virologically suppressed HIV-1 infected patients (2 to less than 12 years of age and weighing at least 10 kg) (see section 5.1). The median duration of exposure for patients in the week 48 analysis was 48.4 (range 47 to 52) weeks.
All ADRs were mild or moderate. ADRs reported in at least 2 participants, regardless of severity, were: vomiting (4/26, 15.4%), abdominal pain (3/26, 11.5%), nausea (2/26, 7.7%), ALT increased (3/26, 11.5%), AST increased (2/26, 7.7%), and decreased appetite (2/26, 7.7%). There were no patients who discontinued treatment due to ADRs. No new ADRs were identified compared to those seen in adults.
The safety and efficacy of rilpivirine in children less than 2 years or weighing less than 14 kg have not been established.
Other special populations
Patients co-infected with hepatitis B and/or hepatitis C virus
In patients co-infected with hepatitis B or C virus receiving rilpivirine, the incidence of hepatic enzyme elevation was higher than in patients receiving rilpivirine who were not co-infected. This observation was the same in the efavirenz arm. The pharmacokinetic exposure of rilpivirine in co-infected patients was comparable to that in patients without co-infection.
Reporting of suspected adverse reactions
Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the national reporting system listed in Appendix V.
Incompatibilities
Not applicable.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.