LIFYORLI Capsule Ref.[116441] Active ingredients: Relacorilant
Source: FDA, National Drug Code (US) Revision Year: 2026
Product description
LIFYORLI (relacorilant) is a glucocorticoid receptor antagonist antineoplastic agent.
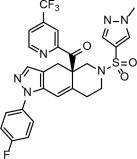
Relacorilant chemical names:
- (R)-(1-(4-fluorophenyl)-6-((1-methyl-1H-pyrazol-4-yl)sulfonyl)-4,4a,5,6,7,8-hexahydro-1H-pyrazolo[3,4-g]isoquinolin-4a-yl)(4-trifluoromethyl)(pyridin-2-yl)methanone
- [(4aR)-1-(4-fluorophenyl)-6-(1-methyl-1H-pyrazole-4-sulfonyl)-1,4,5,6,7,8-hexahydro-4aH-pyrazolo[3,4-g]isoquinolin-4a-yl][4-(trifluoromethyl)pyridin-2-yl]methanone
The molecular formula is C27H22F4N6O3S and the molecular weight is 586.57 g/mol.
The chemical structure of relacorilant is:
Relacorilant is an off-white to light yellow solid that is practically insoluble in aqueous media.
LIFYORLI capsules for oral use are supplied as immediate release, printed, soft gelatin capsules containing 25 mg or 100 mg of relacorilant. Each capsule contains the following excipients: butylated hydroxytoluene, lauroyl polyoxyl-32 glycerides, and propylene glycol caprylate. The capsule shell for the 25 mg strength contains black iron oxide, gelatin, red iron oxide, sorbitol special glycerin blend, titanium dioxide, and yellow iron oxide. The capsule shell for the 100 mg strength contains gelatin, sorbitol special glycerin blend, titanium dioxide, and yellow iron oxide. The printing ink contains ammonium hydroxide, black iron oxide, ethanol, ethyl acetate, isopropyl alcohol, macrogol/polyethylene glycol, polyvinyl acetate phthalate, propylene glycol, and purified water.
| Dosage Forms and Strengths |
|---|
|
Capsules:
|
| How Supplied | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
LIFYORLI is available as soft gelatin capsules containing 25 mg or 100 mg of relacorilant. LIFYORLI 25 mg are opaque dark brown, oval soft gelatin capsules with "CR25" printed in black. LIFYORLI 100 mg are opaque yellow, oblong soft gelatin capsules with "CR100" printed in black. LIFYORLI capsules are supplied as follows:
Manufactured for: Corcept Therapeutics Incorporated, Redwood City, CA 9406 |
Drugs
| Drug | Countries | |
|---|---|---|
| LIFYORLI | United States |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.