LIFYORLI Capsule Ref.[116441] Active ingredients:
Source: FDA, National Drug Code (US) Revision Year: 2026
12.1. Mechanism of Action
Relacorilant is a reversible glucocorticoid receptor (GR) antagonist. In functional in vitro assays with the mineralocorticoid receptor, relacorilant showed no agonist or antagonist activity. In human cell-line derived xenograft models, relacorilant enhanced apoptosis and antitumor activity when administered with paclitaxel.
Cortisol binding to the GR is immunosuppressive, decreasing secretion of pro‑inflammatory cytokines. GR antagonism may indirectly activate the immune system; relacorilant inhibited the cortisol-induced reduction of tumor necrosis factor alpha and interferon gamma in stimulated peripheral blood mononuclear cells.
12.2. Pharmacodynamics
Exposure-Response Relationships
Relacorilant exposure-response relationships and the time course of pharmacodynamic response have not been fully characterized.
Cardiac Electrophysiology
At 3.8 times the mean maximal relacorilant concentration of the recommended dosage, clinically significant QTc interval prolongation was not observed.
12.3. Pharmacokinetics
Relacorilant pharmacokinetics were observed in adult patients at the approved recommended dosage and are presented as mean (CV%), unless otherwise specified. Relacorilant maximum concentration (Cmax) is 720 (68%) ng/mL and systemic exposure (AUC) is 5,686 (84%) ng*h/mL following the third consecutive daily dose.
Absorption
Relacorilant median (min, max) time to maximum plasma concentration (Tmax) is 2.5 hours (1.2, 5.5).
Effect of Food
Relacorilant Cmax increased 1.7-fold, AUC increased 2-fold, and Tmax was delayed 0.5 hours following administration with a low-fat meal (approximately 400 to 500 calories, 25% fat content).
Relacorilant Cmax increased 1.9-fold, AUC increased 2.4-fold, and Tmax was delayed 0.25 hours following administration with a high-fat meal (approximately 800 to 1,000 calories, 50% fat content).
Distribution
Relacorilant apparent volume of distribution (Vz/F) is 2490 (54%) L after a single 150 mg dose under fasted conditions. Protein binding of relacorilant in human plasma is >99% in vitro.
Elimination
Relacorilant terminal half-life is 27 (30%) hours with an apparent total clearance of 52 (69%) L/h under fasted conditions.
Metabolism
Relacorilant is primarily metabolized by cytosolic reductases and CYP3A. An active metabolite, CORT125295, with functional activity approximately 5-fold lower than relacorilant, represents 75% of the parent AUC.
Excretion
After a single oral dose of radiolabeled relacorilant 250 mg to healthy participants, about 73% (<1% unchanged) of the dose was recovered in feces and 17% (<2% unchanged) in urine.
Specific Populations
No clinically significant differences in the pharmacokinetics of relacorilant were observed based on age (26 to 85 years), body weight (35 to 128 kg), race (White [81%], Asian [9%]), ethnicity (Non-Hispanic [93%], Hispanic [7%]), CLcr 30 to <90 mL/min, or mild hepatic impairment (total bilirubin > ULN to ≤1.5 × ULN or AST > ULN and total bilirubin ≤ ULN).
The effect of CLcr <30 mL/min or end-stage renal disease undergoing hemodialysis or severe hepatic impairment (total bilirubin >3 to 10 × ULN and any AST) on the pharmacokinetics of relacorilant is unknown.
Patients with Hepatic Impairment
Relacorilant AUC increased 1.3-fold in participants with moderate hepatic impairment (Child-Pugh Class B).
Drug Interaction Studies
Clinical Studies
Nab-paclitaxel: Dose-normalized paclitaxel (CYP2C8 and CYP3A4 substrate) Cmax increased 2-fold and dose-normalized AUC increased 1.7-fold following coadministration of nab-paclitaxel with LIFYORLI 150 mg.
No clinically significant differences in the pharmacokinetics of relacorilant were observed when given in combination with nab-paclitaxel.
Strong CYP3A Inhibitors: Relacorilant (300 mg once daily for 10 days under fasted conditions) steady state Cmax increased 1.2-fold and steady state AUC increased 1.5-fold following coadministration of itraconazole (strong CYP3A inhibitor) 200 mg once daily for 10 days.
CYP3A Substrates: Midazolam (CYP3A substrate) Cmax increased 3.1-fold and AUC increased 8.9-fold following coadministration of LIFYORLI 350 mg under fasted conditions (1.8 times the relacorilant exposure at the recommended dosage) once daily for 10 days.
CYP2C8 Substrates: Pioglitazone (CYP2C8 substrate) Cmax decreased to 78% and AUC to 75% following coadministration of LIFYORLI 350 mg under fasted conditions (1.8 times the relacorilant exposure at the recommended dosage) once daily for 11 days.
Other Drugs: No clinically significant differences in the pharmacokinetics of the following drugs were observed when used concomitantly with relacorilant: metoprolol (CYP2D6), tolbutamide (CYP2C9), omeprazole (CYP2C19), or dabigatran etexilate (P-gp).
In vitro Studies
CYP450 Enzymes: Relacorilant and CORT125295 are inducers of CYP1A2.
Transporter Systems: Relacorilant and CORT125295 are inhibitors of BCRP.
13.1. Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
In a 6-month carcinogenicity study, transgenic rasH2 mice were orally administered up to 100 mg/kg/day relacorilant. In a 2-year carcinogenicity study, rats were orally administered up to 10 mg/kg/day in male rats (1.3 times the human exposure based on AUC at the recommended dose) and 3 mg/kg/day in female rats (0.7 times the human exposure based on AUC at the recommended dose). There was no evidence of relacorilant-induced carcinogenicity in either study.
Mutagenesis
Relacorilant was not genotoxic in the bacterial reverse mutation (Ames) assay, an in vitro human lymphocyte micronucleus assay, or an in vivo micronucleus assay in rats.
Impairment of Fertility
In a fertility study, relacorilant was administered orally to male rats prior to and throughout mating and to female rats prior to mating and up to the implantation day (gestation day 7). Relacorilant had no effect on fertility or reproductive function in male or female rats at doses up to 40 mg/kg/day (≥4.5 times the human exposure based on AUC at the recommended dose).
14. Clinical Studies
The efficacy of LIFYORLI in combination with nab-paclitaxel was evaluated in ROSELLA (NCT05257408), a multicenter, open-label, active-controlled, randomized, two-arm, trial in patients with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer. Patients were permitted to receive up to three prior lines of systemic therapy and prior bevacizumab was required. The trial excluded patients who required chronic or frequent use of glucocorticoids.
A total of 381 patients were randomized (1:1) to receive:
- LIFYORLI 150 mg orally the day before, the day of, and the day after administration of nab-paclitaxel 80 mg/m² intravenous infusion on Days 1, 8 and 15 of each 28-day cycle (188) or
- nab-paclitaxel 100 mg/m² intravenous infusion on Days 1, 8 and 15 of each 28-day cycle (193)
Treatment was administered until disease progression or unacceptable toxicity. Tumor response assessments occurred every 8 weeks for the first 40 weeks and every 12 weeks thereafter. Randomization was stratified by the following factors: number of prior lines of therapy (1 vs. >1) and region (North America vs. Europe vs. Korea/Australia/Latin America).
The major efficacy outcome measures were progression-free survival (PFS) assessed by blinded independent central review (BICR) and overall survival (OS). PFS was evaluated according to Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1.
The median age was 62 years (range: 26 to 86); 71% were White, 13% were Asian, 1.3% were Black or African American, and 13% did not have race reported. Nine percent of patients were Hispanic or Latino, 76% were non-Hispanic, and 15% had ethnicity not reported or unknown. Nearly all patients had an Eastern Cooperative Oncology Group Performance Status (ECOG PS) of 0 (69%) or 1 (30%). Nine percent of patients had received 1 prior line of systemic therapy, 48% of patients had received 2 prior lines of systemic therapy, and 44% of patients had received 3 prior lines of systemic therapy. Thirty-nine percent of patients received prior systemic therapy for platinum-resistant disease. All patients received prior bevacizumab, 99.5% had received a prior taxane (19% in the last line of therapy and 4% in the platinum-resistant setting) and 61% had received a prior PARP inhibitor. Of patients who had received a prior PARP inhibitor, 78% had radiographic progression while receiving the PARP inhibitor.
LIFYORLI demonstrated a statistically significant improvement in PFS and OS for patients randomized to LIFYORLI in combination with nab-paclitaxel compared to nab-paclitaxel monotherapy.
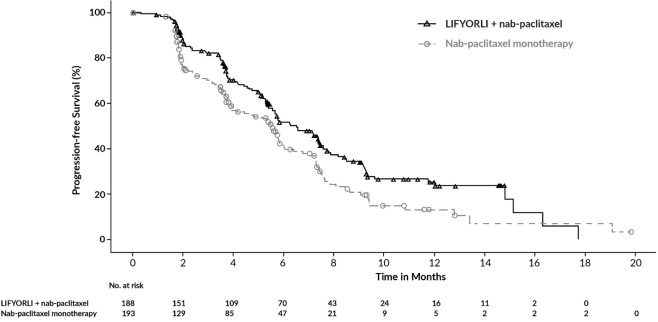
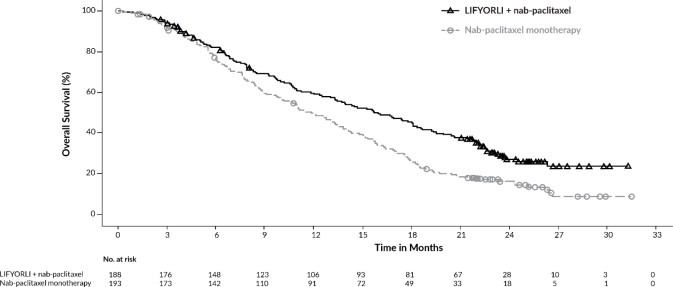
Efficacy results for ROSELLA are summarized in Table 7, Figure 1 and Figure 2.
Table 7. Efficacy Results in ROSELLA:
| LIFYORLI and Nab-Paclitaxel N=188 | Nab-Paclitaxel N=193 | |
| Progression-free Survival (PFS) by BICRa | ||
| Number (%) of patients with events | 113 (60) | 121 (63) |
| Median, months (95% CI) | 6.5 (5.6, 7.4) | 5.5 (3.9, 5.9) |
| Hazard ratio (95% CI)b | 0.70 (0.54, 0.91) | |
| p-valuec | 0.0076 | |
| Overall Survival (OS)d | ||
| Number (%) of patients with events | 129 (69) | 159 (82) |
| Median, months (95% CI) | 16.0 (13.0, 18.3) | 11.9 (10.0, 13.8) |
| Hazard ratio (95% CI)b | 0.65 (0.51, 0.83) | |
| p-valuee | 0.0004 | |
BICR = Blinded Independent Central Review; CI = Confidence Interval
a Results at the pre-specified final PFS analysis.
b Based on the stratified Cox proportional hazard model.
c Two-sided p-value based on stratified log-rank test (compared to a significance level of 0.04).
d Results at the pre-specified final OS analysis.
e Two-sided p-value based on stratified log-rank test (compared to a significance level of 0.0499).
Figure 1. Kaplan-Meier Curve for Progression-Free Survival in ROSELLA:
Figure 2. Kaplan-Meier Curve for Overall Survival in ROSELLA:
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.