LYNAVOY Film-coated tablet Ref.[116436] Active ingredients: Linerixibat
Source: FDA, National Drug Code (US) Revision Year: 2026
Product description
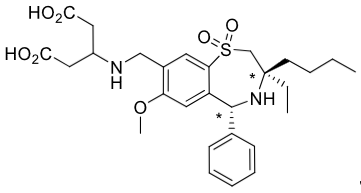
Linerixibat is an orally administered IBAT inhibitor. Linerixibat has the chemical name 3-((((3R,5R)-3-butyl-3-ethyl-7-methyloxy-1,1-dioxido-5-phenyl-2,3,4,5-tetrahydrobenzo[f][1,4]thiazepin-8-yl) methyl)amino)pentanedioic acid. The molecular formula of linerixibat is C28H38N2O7S with a molecular weight of 546.68 g/mol. Linerixibat has the following chemical structure:
* represent chiral centers.
Linerixibat is a white to off-white solid. Its solubility in aqueous solutions is pH-dependent and varies from very slightly to slightly soluble. LYNAVOY is available for oral administration as tablets containing 40 mg linerixibat and the following excipients: croscarmellose sodium, magnesium stearate and microcrystalline cellulose.
The tablet film-coating contains black iron oxide, hypromellose, polyethylene glycol, red iron oxide, and titanium dioxide.
| Dosage Forms and Strengths |
|---|
|
Tablets: 40 mg of linerixibat, purple, biconvex, round, film-coated tablets debossed with "GS 3JG" on one side. |
| How Supplied |
|---|
|
LYNAVOY (linerixibat) tablets, 40 mg, are purple, biconvex, round, film-coated tablets debossed with "GS 3JG" on one side and are supplied in bottles of 60 tablets (NDC 0173-0938-18). LYNAVOY is packaged in white high density polyethylene (HDPE) bottles with polypropylene child-resistant closures and a polyethylene faced induction heat seal liner containing a HDPE canister with silica gel desiccant. Manufactured for: GlaxoSmithKline, Durham, NC 27701 |
Drugs
| Drug | Countries | |
|---|---|---|
| LYNAVOY | United States |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.