ONDEXXYA Powder for solution for infusion Ref.[27699] Active ingredients: Andexanet alfa
Source: European Medicines Agency (EU) Revision Year: 2023 Publisher: AstraZeneca AB, SE-151 85 Södertälje, Sweden
5.1. Pharmacodynamic properties
Pharmacotherapeutic group: All other therapeutic products, antidotes
ATC code: V03AB38
Mechanism of action
Andexanet alfa is a recombinant form of human FXa protein that has been modified to lack FXa enzymatic activity. The active site serine was substituted with alanine, rendering the molecule unable to cleave and activate prothrombin, and the gamma-carboxyglutamic acid (Gla) domain was removed to eliminate the ability of the protein to assemble into the prothrombinase complex, thus removing any anti-coagulant effects.
Andexanet alfa is a specific reversal agent for FXa inhibitors. The predominant mechanism of action is the binding and sequestration of the FXa inhibitor. In addition, andexanet alfa has been observed to bind to, and inhibit tissue factor pathway inhibitor (TFPI). Inhibition of TFPI activity can increase tissue factor-initiated thrombin generation inducing a pro-coagulant effect.
Pharmacodynamic effects
The effects of andexanet alfa can be measured through pharmacodynamic markers, including free fraction of available FXa inhibitor as well as through restoration of thrombin generation. In addition, andexanet alfa has been shown to inhibit TFPI-activity.
Commercial anti-FXa-activity assays are unsuitable for measuring anti-FXa activity following administration of andexanet alfa. Due to the reversible binding of andexanet alfa to the FXa inhibitor, the high sample dilution currently used in these assays leads to dissociation of the inhibitor from andexanet alfa, resulting in detection of erroneously elevated anti-FXa activity levels, thereby causing a substantial underestimation of the reversal activity of andexanet alfa.
In prospective, randomised, placebo-controlled, dose-ranging studies in healthy subjects, the dose and dose regimen of andexanet alfa required to reverse anti-FXa activity and restore thrombin generation for FXa inhibitors (apixaban or rivaroxaban) were determined with modified assays that are not commercially available.
The maximal reversal of anti-FXa activity was achieved within two minutes of completing the bolus administration. Administration of andexanet alfa as a bolus followed by continuous infusion resulted in a sustained decrease in anti-FXa activity. The anti-FXa activity returned to the placebo levels and above approximately two hours after the end of a bolus or infusion dependent on dosage.
When andexanet alfa was administered as a bolus followed by a continuous infusion, the maximum decrease in unbound FXa inhibitors was rapid (within two minutes of the end of the bolus) and was sustained over the course of the infusion then gradually increased over time, reaching a maximum at approximately two hours following the end of infusion.
Restoration of thrombin generation following administration was dose- and dose-regimen-dependent and did not correlate with anti-FXa-activity beyond approximately four hours (see below, "restoration of thrombin generation").
Plasma TFPI activity has been shown to be inhibited completely from 2 minutes to 14.5 hours after andexanet alfa bolus-administration in healthy subjects, and returned to baseline within 3 days. Tissue-factor (TF)-initiated thrombin generation immediately increased above the baseline (prior to anticoagulation) and remained elevated for >20 hours in contrast to placebo. Plausibility of a procoagulant effect of TFPI-inhibition is supported by consecutive and sustained slopes of D-Dimers, TAT, and F1+2.
Population pharmacokinetic/pharmacodynamic (PK/PD) modelling and simulation
PK/PD modelling and simulations rely on the interplay between andexanet alfa and FXa inhibitor PK and on the relationships between biomarkers, here anti FXa-activity, TFPI-activity, and ETP. There remain uncertainties regarding the differing effect of the anticoagulant apixaban or rivaroxaban, duration of the reversal effect dependent on the anti-TFPI-effect, and on the necessity of continuous infusion. Precision of simulations in bleeding patients is less than that within healthy volunteers due to the high inter-individual variability.
Clinical efficacy and safety
The efficacy and safety of andexanet alfa have been evaluated in the following: 1) randomised, placebo-controlled, Phase II dose-ranging trials with healthy volunteers administered FXa inhibitors to establish doses required for reversal; 2) two Phase III studies, one with apixaban and the other with rivaroxaban, to confirm the efficacy of the high and low dose regimens; and 3) a global, multicentre, prospectively defined, open-label Phase IIIb/IV study 14-505 in patients with an acute major bleeding episode requiring urgent reversal of FXa anticoagulation.
Reversal of anticoagulation in healthy subjects aged 50-75 (Studies 14-503 and 14-504)
In a prospective, randomised, placebo-controlled study, healthy subjects with a median age of 56.5 years on apixaban 5 mg twice daily received andexanet alfa (n=24) administered as a 400 mg IV bolus immediately followed by a 4 mg per minute IV infusion for 120 minutes (480 mg) or placebo (n=8).
In a similar study, subjects with a median age of 57 years on rivaroxaban 20 mg daily received andexanet alfa (n=26) administered as an 800 mg IV bolus immediately followed by an 8 mg per minute IV infusion for 120 minutes (960 mg) or placebo (n=13).
Reduction in anti-FXa activity
The primary endpoint for both Study 14-503 (apixaban) and Study 14-504 (rivaroxaban) was the percent change in anti-FXa activity from baseline to post-infusion nadir.
Among the apixaban-treated subjects in Study 14-503, the percent change [± standard deviation (SD)] in anti-FXa activity was -92.34% (± 2.809%) for the andexanet alfa group and -32.70% (± 5.578%) for the placebo group (p < 0.0001), the latter reflecting the intrinsic clearance of the anticoagulant.
Among the rivaroxaban-treated subjects in Study 14-504, the percent change (± SD) in anti-FXa activity was -96.72% (± 1.838%) for the andexanet alfa group and -44.75% (± 11.749%) for the placebo group (p<0.0001), the latter reflecting the intrinsic clearance of the anticoagulant.
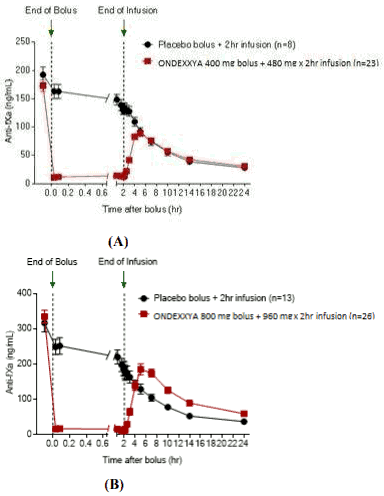
The time courses of anti-FXa activity before and after andexanet alfa administration are shown in Figure 1. Reduction in anti-FXa activity correlates with restoration of thrombin generation. The antiFXa activity thresholds for normalization of thrombin generation (defined by mean ETP and standard deviations) were estimated to be 44.2 ng/mL (within one standard deviation of normal ETP) based on pooled data from Studies 14-503 and 14-504, as indicated in the figure.
Figure 1. Change in anti-FXa activity (ng/mL) in healthy subjects anticoagulated with apixaban (A) and rivaroxaban (B):
Restoration of thrombin generation
In both, Study 14-503 and Study 14-504, treatment with andexanet alfa also resulted in a statistically significant increase in thrombin generation in healthy subjects anticoagulated with apixaban or rivaroxaban versus placebo (p<0.0001). Restoration of thrombin generation to within normal ranges (defined as one standard deviation from baseline levels) within two minutes and maintained for 20 hours was achieved with bolus only and bolus plus infusion for low-dose andexanet alfa in subjects on apixaban. For subjects on rivaroxaban, high-dose andexanet alfa (bolus plus infusion) resulted in increased thrombin generation above two standard deviations. No clinical evaluation for apixabantreated subjects with high-dose andexanet alfa and no evaluation for rivaroxaban-treated subjects with low-dose andexanet alfa was performed in these studies.
Change from baseline in free FXa inhibitor concentration at nadir
The mean unbound concentrations of apixaban and rivaroxaban were <3.5 ng/mL and 4 ng/mL, respectively, after bolus andexanet alfa administration and were maintained throughout the continuous infusion. These levels of unbound FXa inhibitor provide little or no anticoagulant effect.
Reversal of FXa inhibitor anticoagulation in patients with acute major bleeding (study 14-505)
In Study 14-505 (ANNEXA-4), a Phase IIIb/IV multinational, prospective, single-arm, open-label study, Ondexxya was administered to 477 patients on FXa inhibitors, 419 of whom were on apixaban and rivaroxaban, who presented with acute major bleeding. The two co-primary endpoints were: a) percent change in anti-FXa activity from baseline to the nadir between five minutes after the end of the bolus until the end of the infusion, and; b) rate of good or excellent (compared to poor or none) haemostatic efficacy within 12 hours after infusion, as rated by an independent endpoint adjudication committee.
Approximately half of the patients were male, and the mean age was 77.9 years. Most patients had previously received either apixaban (245/477; 51.4%) or rivaroxaban (174/477; 36.5%), or edoxaban (36/477; 7.5%) or enoxaparin (22/477; 4.6%) and experienced either an ICH (329/477; 69%) or a gastrointestinal (GI) bleed (109/477; 22.9 %).
381/477 (79.9%) received the low-dose regimen of andexanet, while 96/477 patients (20.1%) received the high-dose regimen, accordingly to section 4.2.
Of 477 enrolled patients, 347 (73%) were evaluable for efficacy as they were dosed with andexanet for a confirmed major bleed and had a baseline anti-FXa activity above 75 ng/mL. For these patients, median anti-FXa activity at baseline was 147 ng/mL for patients taking apixaban, and 214 ng/mL for patients taking rivaroxaban. For anti-FXa activity, the median (95% CI) decrease from baseline to nadir in anti-FXa activity for apixaban was -93.3% (-94.2%, -92.5%); and rivaroxaban was -94.1% (-95.1%; -93.0%).
Haemostatic efficacy was good or excellent in 79% of 169 patients taking apixaban and in 80% of 127 patients taking rivaroxaban.
Anti-TFPI-effect
Immediate and sustained (for about 3 days post infusion) pro-coagulant anti-TFPI-effect was documented in patients with major bleeding – consistent with respective results from studies in healthy volunteers (14-503, 14-504, 16-508, 19-514).
Analysis of study 14-505 demonstrated that the change in anti-FXa activity (surrogate) was not predictive for achievement of haemostatic efficacy.
Deaths
In the safety population (n=419), 75 patients (18%) died. Of the 75 subjects who died, the bleeding type was intracranial bleeding in 55 (73%), gastrointestinal bleeding in 14 (19%), and other bleeding types in 6 (8%) subjects. The mortality rates were 19.0% (55/289) in patients presenting with ICH, 14.7% (14/95) with GI bleeding, and 17.1% (6/35) with other types of bleeding. The mortality rates were 23.0% (64/278) in patients aged > 75 years old and 7.8% (11/141) in patients aged ≤ 75 years. According to region, death rates were 24.9% (53/213) in patients recruited in the European Union and 11.3% (22/194) in patients recruited in North America. The higher mortality rate in Europe is only present in older patients or patients with heart failure. Compared with patients recruited in North America, EU patients were significantly older (81.0 years vs. 79.0 years), more frequently had ICH as index event (75.1% vs. 60.3%) and more ICHs were intraparenchymal (69.3% vs. 42.7%). Cardiovascular causes of death (n=36) included: haemorrhagic stroke (n=6), ischaemic stroke (n=10), sudden cardiac death (including unwitnessed) (n=6), cardiomechanical/pump failure (n=4), myocardial infarction (n=2), bleeding other than haemorrhagic stroke (n=2), and other cardiovascular causes (n=6). Non-cardiovascular deaths (n=39) included: infection/sepsis (n=11), respiratory failure (n=6), accident/trauma (n=2), cancer (n=2), and other/non-vascular cause (n=18). The average time to death was 15 days after treatment. All deaths occurred before Day 44.
Thromboembolic events
In study 14-505, 45/419 (11%) patients experienced one or more of the following thromboembolic events: cerebrovascular accident (CVA) (19/45; 42%), deep venous thrombosis (11/45; 24%), myocardial infarction (MI) including acute myocardial infarction and myocardial ischaemia (9/45; 20%), pulmonary embolism (PE) (5/45; 11%), and transient ischaemic attack (TIA) (1/45; 2%). The median time to first thromboembolic event was 10 days. A total of 38% of patients with thromboembolic events (17/45) experienced the thromboembolic event during the first three days. Of the 419 subjects who received andexanet alfa, 266 received at least one anticoagulation dose within 30 days after treatment as a prophylactic measure. Of these 266, 14 subjects (5%) had a thrombotic event after resumption of anticoagulation; while of the 153 subjects who did not receive anticoagulation as a prophylactic, 31 (20.3%) had a thrombotic event (see section 4.4).
Pro- thrombotic laboratory markers
Dose-dependent increases in coagulation markers F1+2, TAT, and D-dimers after administration of andexanet alfa were observed, in 223 healthy volunteers who received FXa inhibitors and were treated with andexanet alfa; no thromboembolic events occurred in these healthy volunteers. F1+2, TAT and D-dimers were not measured in patients enrolled in the study 14-505; their relevance in bleeding patients is not known.
Immunogenicity
345 andexanet alfa-treated healthy subjects were tested for antibodies cross reacting with andexanet alfa and antibodies to factor X and FXa. Treatment-emergent, non-neutralizing antibodies to andexanet alfa were detected in approximately 10% (35/345). These antibodies were generally low titre, and no clinical consequences were observed. No neutralising antibodies or antibodies to factor X or FXa were detected. The occurrence of positive, non-neutralizing antibodies to andexanet alfa following treatment in patients in the study 14-505 (8% or 22/276 patients) has been similar to that observed in healthy subjects.
Paediatric population
The European Medicines Agency has deferred the obligation to submit the results of studies with andexanet alfa in one or more subsets of the paediatric population in treatment and prevention of FXa inhibitor-associated haemorrhages (see section 4.2 for information on paediatric use).
Conditional approval
This medicinal product has been authorised under a so-called 'conditional approval' scheme. This means that further evidence on this medicinal product is awaited. The European Medicines Agency will review new information on this medicinal product at least every year, and this SmPC will be updated as necessary.
5.2. Pharmacokinetic properties
Studies of andexanet alfa in the presence of direct FXa inhibitors in healthy subjects demonstrated dose proportional pharmacokinetics over the intended therapeutic dose range evaluated for both Cmax and area under the curve (AUC). The pharmacokinetics of andexanet alfa has not been studied in bleeding patients due to feasibility reasons.
Table 5. Pharmacokinetic parameters for Andexanet bolus-injection of 400 and 800mg:
| PK Parameter | 400 mg Bolus | 800 mg Bolus |
|---|---|---|
| AUC0-∞ (hr*μg/mL) | 61.3 [43.8, 94.9] | 127 [57.5, 209] |
| Cmax (μg/mL) | 61.0 [40.3, 98.5] | 118 [50.2, 191] |
| Clearance (L/hr) | 6.52 [4.21, 9.13] | 6.29 [3.83, 13.9] |
| T1/2 (hr) | 3.78 [2.59, 6.39] | 4.24 [2.47, 6.52] |
| Vss (L) | 9.47 [6.08, 15.3] | 8.94 [5.36, 23.1] |
Source: Study 19-514
Data presented are geometric mean [min, max]
Pharmacokinetics in special populations
Elderly population
In a study comparing andexanet alfa pharmacokinetics in elderly (65-69 years) and younger (26-42 years) healthy subjects who had received apixaban, the pharmacokinetics of andexanet alfa in the elderly subjects were not statistically different than those in the younger subjects.
Renal impairment
No trials have been conducted to investigate the pharmacokinetics of andexanet alfa in renally impaired patients. Based on the available PK data, andexanet alfa has little to no renal clearance, and thus would not require dose adjustment for patients with renal impairment.
Hepatic impairment
No trials have been conducted to investigate the pharmacokinetics of andexanet alfa in patients with hepatic impairment. Biliary and/or faeces elimination of protein therapeutics is not a known route of protein elimination. Therefore, dose adjustment is not considered needed for patients with hepatic impairment.
Gender
Based on population pharmacokinetics analysis, gender does not have a clinically meaningful effect on the pharmacokinetics of andexanet alfa.
Paediatric population
The pharmacokinetics of andexanet alfa has not been studied in paediatric patients.
5.3. Preclinical safety data
Non-clinical data reveal no special hazard for humans based on conventional studies of safety pharmacology and repeated dose toxicity studies up to two weeks in rats and monkeys.
Studies to evaluate the mutagenic and carcinogenic potential of andexanet alfa have not been performed. Based on its mechanism of action and on the characteristics of proteins, no carcinogenic or genotoxic effects are anticipated.
Animal reproductive and developmental studies have not been conducted with andexanet alfa.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.