REVUFORJ Film-coated tablet Ref.[116086] Active ingredients: Revumenib
Source: FDA, National Drug Code (US) Revision Year: 2025
Product description
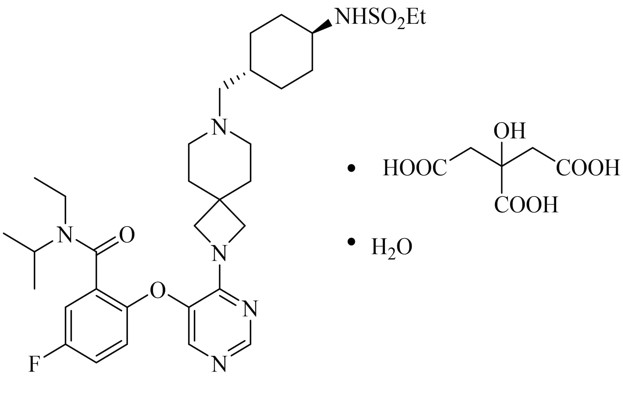
REVUFORJ contains revumenib, a menin inhibitor. Revumenib is present as revumenib citrate hydrate with a chemical name of benzamide, Nethyl-2-[[4-[7-[[trans-4-[(ethylsulfonyl)amino]cyclohexyl]methyl]-2,7-diazaspiro[3.5]non-2-yl]-5-pyrimidinyl]oxy]-5-fluoroN[1 methylethyl]-, 2-hydroxypropane-1,2,3-tricarboxylic acid, hydrate (1:1:1). The molecular formula is C32H47FN6O4S●C6H8O7●H2O with a molecular weight 840.96 g/mol.
Revumenib citrate hydrate is a white to faint pink solid. Revumenib citrate hydrate is soluble at pH 1.2 and 6.8, and sparingly soluble at pH 4.5.
The chemical structure is shown in Figure 1.
Figure 1. Chemical structure of Revumenib Citrate:
REVUFORJ is available as tablets for oral use.
Each 25 mg strength tablet contains 25 mg revumenib, equivalent to 33.4 mg revumenib citrate, and the following inactive ingredients: microcrystalline cellulose, dicalcium phosphate, crospovidone, hypromellose, sodium bicarbonate, hydrophobic colloidal silica, magnesium stearate, polyvinyl alcohol, titanium dioxide, polyethylene glycol, talc, and red iron oxide.
Each 110 mg strength tablet contains 110 mg revumenib, equivalent to 146.5 mg revumenib citrate, and the following inactive ingredients: microcrystalline cellulose, dicalcium phosphate, crospovidone, hypromellose, sodium bicarbonate, hydrophobic colloidal silica, magnesium stearate, polyvinyl alcohol, titanium dioxide, polyethylene glycol, talc, red iron oxide, and yellow iron oxide.
Each 160 mg strength tablet contains 160 mg revumenib equivalent to 213.2 mg revumenib citrate, and the following inactive ingredients: microcrystalline cellulose, dicalcium phosphate, crospovidone, hypromellose, sodium bicarbonate, hydrophobic colloidal silica, magnesium stearate, polyvinyl alcohol, titanium dioxide, polyethylene glycol, talc, red iron oxide, and FD&C blue #2/indigo carmine aluminum lake.
| Dosage Forms and Strengths |
|---|
|
Tablets:
|
| How Supplied |
|---|
|
25 mg: Pink modified oval film-coated tablet debossed with "S" on one side and "25" on the other side. 30-count bottles with a desiccant and child resistant closure (NDC 73555-500-00) 110 mg: Beige modified oval film-coated tablet debossed with "S" on one side and "110" on the other side. 30-count bottles with a desiccant and child resistant closure (NDC 73555-501-00) 160 mg: Purple modified oval film-coated tablet debossed with "S" on one side and "160" on the other side. 30-count bottles with a desiccant and child resistant closure (NDC 73555-502-00) Manufactured for Syndax Pharmaceuticals, Inc., New York, NY 10017 |
Drugs
| Drug | Countries | |
|---|---|---|
| REVUFORJ | United States |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.