RoACTEMRA Solution for injection in pre-filled syringe Ref.[8356] Active ingredients: Tocilizumab
Source: European Medicines Agency (EU) Revision Year: 2019 Publisher: Roche Registration GmbH, Emil-Barell-Strasse 1, 79639, Grenzach-Wyhlen, Germany
Pharmacodynamic properties
Pharmacotherapeutic group: Immunosuppressants, Interleukin inhibitors
ATC code: L04AC07
Mechanism of action
Tocilizumab binds specifically to both soluble and membrane-bound IL-6 receptors (sIL-6R and mIL-6R). Tocilizumab has been shown to inhibit sIL-6R and mIL-6R-mediated signalling. IL-6 is a pleiotropic pro-inflammatory cytokine produced by a variety of cell types including T- and B-cells, monocytes and fibroblasts. IL-6 is involved in diverse physiological processes such as T-cell activation, induction of immunoglobulin secretion, induction of hepatic acute phase protein synthesis and stimulation of haemopoiesis. IL-6 has been implicated in the pathogenesis of diseases including inflammatory diseases, osteoporosis and neoplasia.
Pharmacodynamic effects
In clinical studies with RoActemra, rapid decreases in CRP, erythrocyte sedimentation rate (ESR), serum amyloid A (SAA) and fibrinogen were observed. Consistent with the effect on acute phase reactants, treatment with RoActemra was associated with reduction in platelet count within the normal range. Increases in haemoglobin levels were observed, through RoActemra decreasing the IL-6 driven effects on hepcidin production to increase iron availability. In RoActemra-treated patients, decreases in the levels of CRP to within normal ranges were seen as early as week 2, with decreases maintained while on treatment.
In GCA clinical study WA28119, similar rapid decreases in CRP and ESR were observed along with slight increases in mean corpuscular haemoglobin concentration. In healthy subjects administered RoActemra in doses from 2 to 28 mg/kg intravenously and 81 to 162 mg subcutaneously, absolute neutrophil counts decreased to their lowest 2 to 5 days following administration. Thereafter, neutrophils recovered towards baseline in a dose dependent manner.
Patients demonstrate a comparable (to healthy subjects) decrease of absolute neutrophil counts following RoActemra administration (see section 4.8).
Subcutaneous use
RA
Clinical efficacy
The efficacy of subcutaneous administered RoActemra in alleviating the signs and symptoms of RA and radiographic response, was assessed in two randomised, double-blind, controlled, multi-center studies. For study I (SC-I), patients were required to be >18 years of age with moderate to severe active RA diagnosed according to ACR criteria who had at least 4 tender and 4 swollen joints at baseline. All patients received background non-biologic DMARD. For study II (SC-II), patients were required to be >18 years of age with moderate to severe active RA diagnosed according to ACR criteria who had at least 8 tender and 6 swollen joints at baseline.
Switching from 8 mg/kg intravenous once every 4 weeks to 162 mg subcutaneous once every week, will alter exposure in the patient. The extent varies with the patient’s body weight (increased in light body weight patients and decreased in heavy body weight patients) but clinical outcome is consistent with that observed in intravenous treated patients.
Clinical response
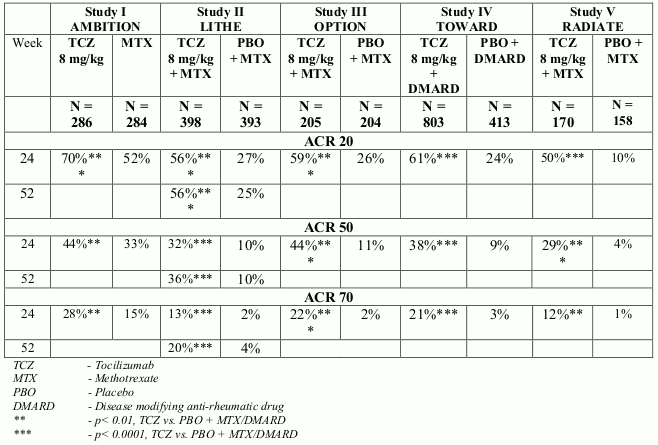
Study SC-I evaluated patients with moderate to severe active RA who had an inadequate clinical response to their existing rheumatologic therapy, including one or more DMARD where approximately 20% had a history of inadequate response to at least one TNF inhibitor. In SC-I, 1262 patients were randomized 1:1 to receive RoActemra subcutaneous 162 mg every week or RoActemra intravenous 8 mg/kg every four weeks in combination with non-biologic DMARD. The primary endpoint in the study was the difference in the proportion of patients who achieved an ACR20 response at week 24. The results from study SC-I is shown in Table 2.
Table 2. ACR responses in study SC-I (% patients) at Week 24:
| SC-Ia | ||
|---|---|---|
| TCZ SC 162 mg every week + DMARD N=558 | TCZ IV 8 mg/kg + DMARD N=537 | |
| ACR20 Week 24 | 69.4% | 73.4% |
| Weighted difference (95% CI) | -4.0 (-9.2, 1.2) | |
| ACR50 Week 24 | 47.0% | 48.6% |
| Weighted difference (95% CI) | -1.8 (-7.5, 4.0) | |
| ACR70 Week 24 | 24.0% | 27.9% |
| Weighted difference (95% CI) | -3.8 (-9.0, 1.3) | |
TCZ = tocilizumab
a = Per Protocol Population
Patients in study SC-I had a mean Disease Activity Score (DAS28) at baseline of 6.6 and 6.7 on the subcutaneous and intravenous arms, respectively. At week 24, a significant reduction in DAS28 from baseline (mean improvement) of 3.5 was observed on both treatment arms, and a comparable proportion of patients had achieved DAS28 clinical remission (DAS28 <2.6) on the subcutaneous (38.4%) and IV (36.9%) arms.
Radiographic response:
The radiographic response of subcutaneous administered RoActemra was assessed in a double-blind, controlled, multicenter study in patients with active RA (SC-II). Study SC-II evaluated patients with moderate to severe active RA who had an inadequate clinical response to their existing rheumatologic therapy, including one or more DMARD where approximately 20% had a history of inadequate response to at least one TNF inhibitor. Patients were required to be >18 years of age with active RA diagnosed according to ACR criteria who had at least 8 tender and 6 swollen joints at baseline. In SC-II, 656 patients were randomized 2:1 to RoActemra subcutaneous 162 mg every other week or placebo, in combination with non-biologic DMARD.
In study SC-II, inhibition of structural joint damage was assessed radiographically and expressed as a change from baseline in the van der Heijde modified mean total Sharp score (mTSS). At week 24, inhibition of structural damage was shown, with significantly less radiographic progression in patients receiving RoActemra subcutaneous compared to placebo (mean mTSS of 0.62 vs. 1.23, p=0.0149 (van Elteren). These results are consistent with those observed in patients treated with intravenous RoActemra.
In study SC-II, at week 24 there was ACR20 of 60.9%, ACR50 of 39.8% and ACR70 of 19.7% for patients treated with RoActemra subcutaneous every other week versus placebo ACR20 of 31.5%, ACR50 of 12.3% and ACR70 of 5.0%. Patients had mean DAS28 at baseline of 6.7 on subcutaneous and 6.6 on placebo arms. At week 24, a significant reduction in DAS28 from baseline of 3.1 was observed on subcutaneous and 1.7 on placebo arm, and for DAS28 <2.6, 32.0% was observed on subcutaneous and 4.0% on placebo arm.
Health-related and quality of life outcomes:
In study SC-I, the mean decrease in HAQ-DI from baseline to week 24 was 0.6 on both the subcutaneous and intravenous arms. The proportion of patients achieving a clinically relevant improvement in HAQ-DI at week 24 (change from baseline of ≥0.3 units) was also comparable on the subcutaneous (65.2%) versus intravenous (67.4%) arms, with a weighted difference in proportions of – 2.3% (95% CI – 8.1, 3.4). For SF-36, the mean change from baseline at week 24 in the mental component score was 6.22 for the subcutaneous arm and 6.54 for the intravenous arm, and for the physical component score was also similar with 9.49 for the subcutaneous arm and 9.65 for the intravenous arm.
In study SC-II, mean decrease in HAQ-DI from baseline to week 24 was significantly greater for patients treated with RoActemra subcutaneous every other week (0.4) versus placebo (0.3). Proportion of patients achieving a clinically relevant improvement in HAQ-DI at week 24 (change from baseline of ≥0.3 units) was higher for RoActemra subcutaneous every other week (58%) versus placebo (46.8%). SF-36 (mean change in mental and physical component scores) was significantly greater with RoActemra subcutaneous group (6.5 and 5.3) versus placebo (3.8 and 2.9).
sJIA (SC)
Clinical Efficacy
A 52-week, open-label, multi-centre, PK/PD and safety study (WA28118) was conducted in paediatric patients with sJIA, aged 1 to 17 years, to determine the appropriate SC dose of RoActemra that achieved comparable PK/PD and safety profiles to the IV regimen.
Eligible patients received RoActemra dosed according to body weight (BW), with patients weighing ≥30 kg (n=26) dosed with 162 mg of RoActemra every week (QW) and patients weighing below 30 kg (n=25) dosed with 162 mg of RoActemra every 10 days (Q10D; n=8) or every 2 weeks (Q2W; n=17) for 52 weeks. Of these 51 patients, 26 (51%) were naive to RoActemra and 25 (49%) had been receiving RoActemra IV and switched to RoActemra SC at baseline.
Exploratory efficacy results showed that RoActemra SC improved all exploratory efficacy parameters including Juvenile Arthritis Disease Activity Score (JADAS)-71, for TCZ naïve patients and maintained all exploratory efficacy parameters for patients who switched from RoActemra IV to RoActemra SC treatment over the entire course of the study for patients in both body weight groups (below 30 kg and ≥30 kg).
pJIA (SC)
A 52-week, open-label, multicenter, PK-PD and safety study was conducted in paediatric patients with pJIA, aged 1 to 17 years old, to determine the appropriate subcutaneous dose of RoActemra that achieved comparable PK/PD and safety profiles to the IV regimen.
Eligible patients received tocilizumab dosed according to body weight (BW), with patients weighing ≥30 kg (n=25) dosed with 162 mg of RoActemra every 2 weeks (Q2W) and patients weighing below 30 kg (n=27) dosed with 162 mg of RoActemra every 3 weeks (Q3W) for 52 weeks. Of these 52 patients, 37 (71%) were naive to RoActemra and 15 (29%) had been receiving RoActemra IV and switched to RoActemra SC at baseline.
The RoActemra SC regimens of 162 mg Q3W for patients weighing below 30 kg and of 162 mg Q2W for patients weighing ≥ 30 kg respectively provide PK exposure and PD responses to support efficacy and safety outcomes similar to those achieved with the approved RoActemra IV regimens for pJIA.
Exploratory efficacy results showed that RoActemra SC improved median Juvenile Arthritis Disease Activity Score (JADAS)-71 for RoActemra naïve patients and maintained the median JADAS-71 for patients who switched from IV to SC RoActemra treatment over the entire course of the study for patients in both body weight groups (below 30 kg and ≥30 kg).
GCA (SC)
Clinical efficacy
Study WA28119 was a randomized, multi-center, double-blind placebo-controlled Phase III superiority study conducted to assess the efficacy and safety of RoActemra in patients with GCA.
Two hundred and fifty one (251) patients with new-onset or relapsing GCA were enrolled and assigned to one of four treatment arms. The study consisted of a 52-week blinded period (Part 1), followed by a 104-week open-label extension (Part 2). The purpose of Part 2 was to describe the long-term safety and maintenance of efficacy after 52 weeks of RoActemra therapy, to explore the rate of relapse and the requirement for RoActemra therapy beyond 52 weeks, and to gain insight into the potential long-term steroid-sparing effect of RoActemra.
Two subcutaneous doses of RoActemra (162 mg every week and 162 mg every other week) were compared to two different placebo control groups randomised 2:1:1:1.
All patients received background glucocorticoid (prednisone) therapy. Each of the RoActemra-treated groups and one of the placebo-treated groups followed a pre-specified prednisone-taper regimen over 26 weeks, while the second placebo-treated group followed a pre-specified prednisone-taper regimen over 52 weeks, designed to be more in keeping with standard practice.
The duration of glucocorticoid therapy during screening and before RoActemra (or placebo) was initiated, was similar in all 4 treatment groups (see Table 3).
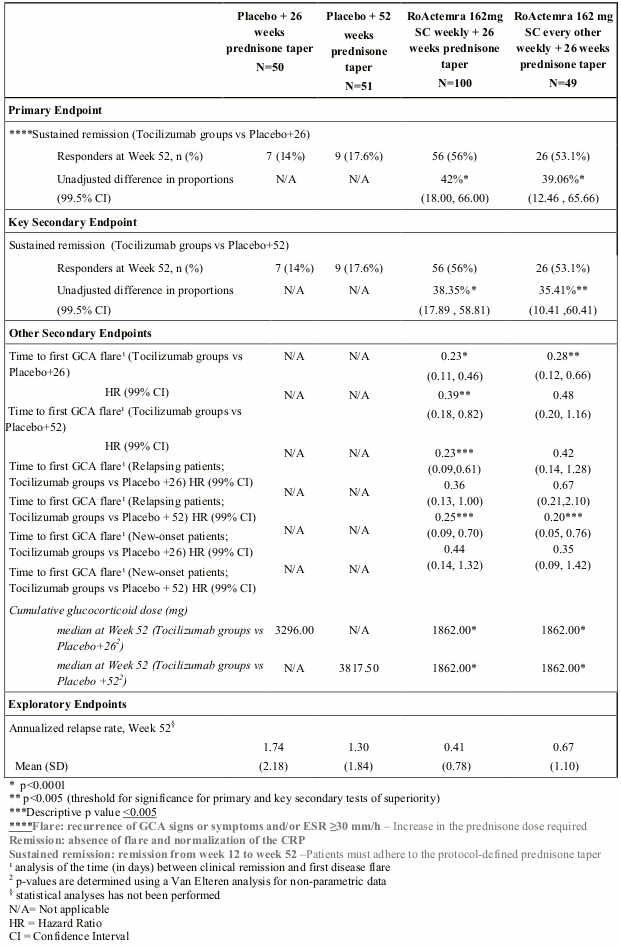
Table 3. Duration of Corticosteroid Therapy During Screening in Study WA28119:
| Placebo + 26 weeks prednisone taper N=50 | Placebo + 52 weeks prednisone taper N=51 | RoActemra 162mg SC weekly + 26 weeks prednisone taper N=100 | RoActemra 162 mg SC every other weekly + 26 weeks prednisone taper N=49 | |
|---|---|---|---|---|
| Duration (days) | ||||
| Mean (SD) | 35.7 (11.5) | 36.3 (12.5) | 35.6 (13.2) | 37.4 (14.4) |
| Median | 42.0 | 41.0 | 41.0 | 42.0 |
| Min-Max | 6-63 | 12–82 | 1-87 | 9-87 |
The primary efficacy endpoint assessed by the proportion of patients achieving steroid free sustained remission at week 52 on RoActemra plus 26 weeks prednisone taper compared with placebo plus 26 weeks prednisone taper, was met (Table 4).
The key secondary efficacy endpoint also based on the proportion of patients achieving sustained remission at week 52, comparing tocilizumab plus 26 weeks prednisone taper with placebo plus 52 weeks prednisone taper, was also met (Table 4).
A statistically significant superior treatment effect was seen in favour of RoActemra over placebo in achieving steroid-free sustained remission at week 52 on RoActemra plus 26 weeks prednisone taper compared with placebo plus 26 weeks prednisone taper and with placebo plus 52 weeks prednisone taper.
The percentage of patients achieving sustained remission at week 52, are shown in the Table 4.
Secondary Endpoints:
The assessment of the time to first GCA flare showed a significantly lower risk of flare for the RoActemra subcutaneous weekly group compared to placebo plus 26 weeks prednisone and placebo plus 52 weeks prednisone taper groups and for the RoActemra subcutaneous every other weekly group compared to placebo plus 26 weeks prednisone (when compared at a 0.01 significance level). RoActemra subcutaneous weekly dose also showed a clinically meaningful decrease in the risk for flare compared to placebo plus 26 weeks prednisone in patients who entered the trial with relapsing GCA as well as those with new-onset disease (Table 4).
Cumulative glucocorticoid dose:
The cumulative prednisone dose at week 52 was significantly lower in the two RoActemra dose groups compared to the two placebo groups (Table 3). In a separate analysis of the patients who received escape prednisone to treat GCA flare during the first 52 weeks, the cumulative prednisone dose varied greatly. The median doses for escape patients in the RoActemra weekly and every other weekly groups were 3129.75 mg and 3847 mg, respectively. Both considerably lower than in the placebo plus 26 weeks and the placebo plus 52 weeks prednisone taper groups, 4023.5 mg and 5389.5 mg respectively.
Table 4. Efficacy results from Study WA28119:
Quality of Life Outcomes:
In study WA28119, the SF-36 results were separated into the physical and mental component summary scores (PCS and MCS, respectively). The PCS mean change from baseline to week 52 was higher (showing more improvement) in the RoActemra weekly and every other weekly dose groups [4.10, 2.76, respectively] than in the two placebo groups [placebo plus 26 weeks; -0.28, placebo plus 52 weeks; -1.49], although only the comparison between RoActemra weekly plus 26 weeks prednisone taper group and placebo plus 52 weeks prednisone taper group (5.59, 99% CI: 8.6, 10.32) showed a statistically significant difference (p=0.0024). For MCS, the mean change from baseline to week 52 for both RoActemra weekly and every other weekly dose groups [7.28, 6.12, respectively] were higher than the placebo plus 52 weeks prednisone taper group [2.84] (although the differences were not statistically significant [weekly p=0.0252 for weekly, p=0.1468 for every other weekly]) and similar to the placebo plus 26 weeks prednisone taper group [6.67].
The Patient’s Global Assessment of disease activity was assessed on a 0-100mm Visual Analogue Scale (VAS). The mean change in Patient’s global VAS from baseline at week 52 was lower (showing greater improvement) in the RoActemra weekly and every other weekly dose groups [-19.0, -25.3, respectively] than in both placebo groups [placebo plus 26 weeks -3.4, placebo plus 52 weeks -7.2], although only the RoActemra every other weekly plus 26 weeks prednisone taper group showed a statistically significant difference compared to placebo [placebo plus 26 weeks taper p=0.0059, and placebo plus 52 weeks taper p=0.0081].
FACIT-Fatigue change from baseline to week 52 scores were calculated for all groups. The mean [SD] change scores were as follows: RoActemra weekly plus 26 weeks 5.61 [10.115], RoActemra every other weekly plus 26 weeks 1.81 [8.836], placebo plus 26 weeks 0.26 [10.702], and placebo plus 52 weeks -1.63 [6.753].
Change in EQ5D scores from baseline to week 52 were RoActemra weekly plus 26 weeks 0.10 [0.198], RoActemra every other weekly plus 26 weeks 0.05 [0.215], placebo plus 26 weeks 0.07 [0.293], and placebo plus 52 weeks -0.02 [0.159].
Higher scores signal improvement in both FACIT-Fatigue and EQ5D.
Intravenous use
RA
Clinical efficacy
The efficacy of RoActemra in alleviating the signs and symptoms of RA was assessed in five randomised, double-blind, multi-centre studies. Studies I-V enrolled patients ≥18 years of age with active RA diagnosed according to the American College of Rheumatology (ACR) criteria and who had at least eight tender and six swollen joints at baseline.
In Study I, RoActemra was administered intravenously every four weeks as monotherapy. In Studies II, III and V, RoActemra was administered intravenously every four weeks in combination with MTX vs. placebo and MTX. In Study IV, RoActemra was administered intravenously every 4 weeks in combination with other DMARDs vs. placebo and other DMARDs. The primary endpoint for each of the five studies was the proportion of patients who achieved an ACR 20 response at week 24.
Study I evaluated 673 patients who had not been treated with MTX within six months prior to randomisation and who had not discontinued previous MTX treatment as a result of clinically important toxic effects or lack of response. The majority (67%) of patients were MTX-naïve. Doses of 8 mg/kg of RoActemra were given every four weeks as monotherapy. The comparator group was weekly MTX (dose titrated from 7.5 mg to a maximum of 20 mg weekly over an eight week period).
Study II, a two year study with planned analyses at week 24, week 52 and week 104, evaluated 1,196 patients who had an inadequate clinical response to MTX. Doses of 4 or 8 mg/kg of RoActemra or placebo were given every four weeks as blinded therapy for 52 weeks in combination with stable MTX (10 mg to 25 mg weekly). After week 52, all patients could receive open-label treatment with RoActemra 8 mg/kg. Of the patients who completed the study who were originally randomised to placebo + MTX, 86% received open-label RoActemra 8 mg/kg in year 2. The primary endpoint at week 24 was the proportion of patients who achieved an ACR 20 response. At week 52 and week 104 the co-primary endpoints were prevention of joint damage and improvement in physical function.
Study III evaluated 623 patients who had an inadequate clinical response to MTX. Doses of 4 or 8 mg/kg RoActemra or placebo were given every four weeks, in combination with stable MTX (10 mg to 25 mg weekly).
Study IV evaluated 1,220 patients who had an inadequate response to their existing rheumatologic therapy, including one or more DMARDs. Doses of 8 mg/kg RoActemra or placebo were given every four weeks in combination with stable DMARDs.
Study V evaluated 499 patients who had an inadequate clinical response or were intolerant to one or more TNF antagonist therapies. The TNF antagonist therapy was discontinued prior to randomisation. Doses of 4 or 8 mg/kg RoActemra or placebo were given every four weeks in combination with stable MTX (10 mg to 25 mg weekly).
Clinical response
In all studies, patients treated with RoActemra 8 mg/kg had statistically significant higher ACR 20, 50, 70 response rates at 6 months compared to control (Table 5). In study I, superiority of RoActemra 8 mg/kg was demonstrated against the active comparator MTX.
The treatment effect was similar in patients independent of rheumatoid factor status, age, gender, race, number of prior treatments or disease status. Time to onset was rapid (as early as week 2) and the magnitude of response continued to improve with duration of treatment. Continued durable responses were seen for over 3 years in the open label extension studies I-V.
In patients treated with RoActemra 8 mg/kg, significant improvements were noted on all individual components of the ACR response including: tender and swollen joint counts; patients and physician global assessment; disability index scores; pain assessment and CRP compared to patients receiving placebo plus MTX or other DMARDs in all studies.
Patients in studies I–V had a mean Disease Activity Score (DAS28) of 6.5–6.8 at baseline. Significant reduction in DAS28 from baseline (mean improvement) of 3.1–3.4 were observed in RoActemra-treated patients compared to control patients (1.3-2.1). The proportion of patients achieving a DAS28 clinical remission (DAS28 <2.6) was significantly higher in patients receiving RoActemra (28–34%) compared to 1–12% of control patients at 24 weeks. In study II, 65% of patients achieved a DAS28 <2.6 at week 104 compared to 48% at 52 weeks and 33% of patients at week 24.
In a pooled analysis of studies II, III and IV, the proportion of patients achieving an ACR 20, 50 and 70 response was significantly higher (59% vs. 50%, 37% vs. 27%, 18% vs. 11%, respectively) in the RoActemra 8 mg/kg plus DMARD vs. the RoActemra 4 mg/kg plus DMARD group (p<0.03). Similarly the proportion of patients achieving a DAS28 remission (DAS28 <2.6) was significantly higher (31% vs. 16% respectively) in patients receiving RoActemra 8 mg/kg plus DMARD than in patients receiving RoActemra 4 mg/kg plus DMARD (p<0.0001).
Table 5. ACR responses in placebo-/MTX-/DMARDs-controlled studies (% patients):
Major Clinical Response
After 2 years of treatment with RoActemra plus MTX, 14% of patients achieved a major clinical response (maintenance of an ACR70 response for 24 weeks or more).
Radiographic response
In Study II, in patients with an inadequate response to MTX, inhibition of structural joint damage was assessed radiographically and expressed as change in modified Sharp score and its components, the erosion score and joint space narrowing score. Inhibition of joint structural damage was shown with significantly less radiographic progression in patients receiving RoActemra compared to control (Table 6).
In the open-label extension of Study II the inhibition of progression of structural joint damage in RoActemra plus MTX-treated patients was maintained in the second year of treatment. The mean change from baseline at week 104 in total Sharp-Genant score was significantly lower for patients randomised to RoActemra 8 mg/kg plus MTX (p<0.0001) compared with patients who were randomised to placebo plus MTX.
Table 6. Radiographic mean changes over 52 weeks in Study II:
| PBO + MTX (+ TCZ from week 24) N=393 | TCZ 8 mg/kg + MTX N=398 | |
|---|---|---|
| Total Sharp-Genant score | 1.13 | 0.29* |
| Erosion score | 0.71 | 0.17* |
| JSN score | 0.42 | 0.12** |
PBO – Placebo
MTX – Methotrexate
TCZ – Tocilizumab
JSN – Joint space narrowing
* - p≤0.0001, TCZ vs. PBO + MTX
** - p<0.005, TCZ vs. PBO + MTX
Following 1 year of treatment with RoActemra plus MTX, 85% of patients (n=348) had no progression of structural joint damage, as defined by a change in the Total Sharp Score of zero or less, compared with 67% of placebo plus MTX-treated patients (n=290) (p≤0.001). This remained consistent following 2 years of treatment (83%; n=353). Ninety three percent (93%; n=271) of patients had no progression between week 52 and week 104.
Health-related and quality of life outcomes
RoActemra-treated patients reported an improvement in all patient-reported outcomes (Health Assessment Questionnaire Disability Index – HAQ-DI), Short Form-36 and Functional Assessment of Chronic Illness Therapy questionnaires. Statistically significant improvements in HAQ-DI scores were observed in patients treated with RoActemra compared with patients treated with DMARDs. During the open-label period of Study II, the improvement in physical function has been maintained for up to 2 years. At Week 52, the mean change in HAQ-DI was -0.58 in the RoActemra 8 mg/kg plus MTX group compared with -0.39 in the placebo + MTX group. The mean change in HAQ-DI was maintained at Week 104 in the RoActemra 8 mg/kg plus MTX group (-0.61).
Haemoglobin levels
Statistically significant improvements in haemoglobin levels were observed with RoActemra compared with DMARDs (p<0.0001) at week 24. Mean haemoglobin levels increased by week 2 and remained within normal range through to week 24.
RoActemra versus adalimumab in monotherapy
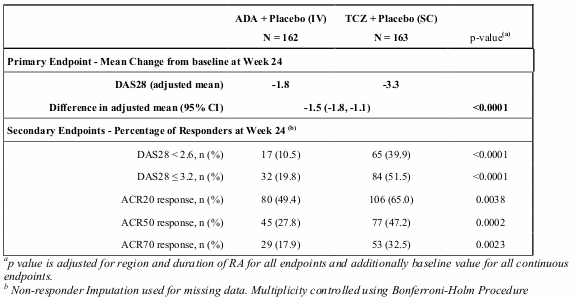
Study VI (WA19924), a 24 week double-blinded study that compared RoActemra monotherapy with adalimumab monotherapy, evaluated 326 patients with RA who were intolerant of MTX or where continued treatment with MTX was considered inappropriate (including MTX inadequate responders). Patients in the RoActemra arm received an intravenous (IV) infusion of RoActemra (8 mg/kg) every 4 weeks (q4w) and a subcutaneous (SC) placebo injection every 2 weeks (q2w). Patients in the adalimumab arm received an adalimumab SC injection (40 mg) q2w plus an IV placebo infusion q4w. A statistically significant superior treatment effect was seen in favour of RoActemra over adalimumab in control of disease activity from baseline to week 24 for the primary endpoint of change in DAS28 and for all secondary endpoints (Table 7).
Table 7. Efficacy Results for Study VI (WA19924):
The overall clinical adverse event profile was similar between RoActemra and adalimumab. The proportion of patients with serious adverse events was balanced between the treatment groups (RoActemra 11.7% vs. adalimumab 9.9%). The types of adverse drug reactions in the RoActemra arm were consistent with the known safety profile of RoActemra and adverse drug reactions were reported at a similar frequency compared with Table 1. A higher incidence of infections and infestations was reported in the RoActemra arm (48% vs. 42%), with no difference in the incidence of serious infections (3.1%). Both study treatments induced the same pattern of changes in laboratory safety parameters (decreases in neutrophil and platelet counts, increases in ALT, AST and lipids), however, the magnitude of change and the frequency of marked abnormalities was higher with RoActemra compared with adalimumab. Four (2.5%) patients in the RoActemra arm and two (1.2%) patients in the adalimumab arm experienced CTC grade 3 or 4 neutrophil count decreases. Eleven (6.8%) patients in the RoActemra arm and five (3.1%) patients in the adalimumab arm experienced ALT increases of CTC grade 2 or higher. The mean LDL increase from baseline was 0.64 mmol/L (25 mg/dL) for patients in the RoActemra arm and 0.19 mmol/L (7 mg/dL) for patients in the adalimumab arm. The safety observed in the RoActemra arm was consistent with the known safety profile of RoActemra and no new or unexpected adverse drug reactions were observed (see Table 1).
Pharmacokinetic properties
The pharmacokinetics of RoActemra is characterized by nonlinear elimination which is a combination of linear clearance and Michaelis-Menten elimination. The nonlinear part of RoActemra elimination leads to an increase in exposure that is more than dose-proportional. The pharmacokinetic parameters of RoActemra do not change with time. Due to the dependence of total clearance on RoActemra serum concentrations, the half-life of RoActemra is also concentration-dependent and varies depending on the serum concentration level. Population pharmacokinetic analyses in any patient population tested so far indicate no relationship between apparent clearance and the presence of anti-drug antibodies.
RA
Intravenous use
The pharmacokinetics of RoActemra were determined using a population pharmacokinetic analysis on a database composed of 3552 RA patients treated with a one-hour infusion of 4 or 8 mg/kg RoActemra every 4 weeks for 24 weeks or with 162 mg tocilizumab given subcutaneously either once a week or every other week for 24 weeks.
The following parameters (predicted mean ± SD) were estimated for a dose of 8 mg/kg RoActemra given every 4 weeks: steady-state area under curve (AUC) = 38000 ± 13000 h•μg/mL, trough concentration (Cmin) = 15.9 ± 13.1 μg/mL and maximum concentration (Cmax) = 182 ± 50.4 μg/mL, and. the accumulation ratios for AUC and Cmax were small, 1.32 and 1.09, respectively. The accumulation ratio was higher for Cmin (2.49), which was expected based on the non-linear clearance contribution at lower concentrations. Steady-state was reached following the first administration for Cmax and after 8 and 20 weeks for AUC and Cmin, respectively. RoActemra AUC, Cmin and Cmax increased with increase of body weight. At body weight ≥100 kg, the predicted mean (± SD) steady-state AUC, Cmin and Cmax of RoActemra were 50000 ± 16800 μg•h/mL, 24.4 ± 17.5 μg/mL, and 226 ± 50.3 μg/mL, respectively, which are higher than mean exposure values for the patient population (i.e. all body weights) reported above. The dose-response curve for tocilizumab flattens at higher exposure, resulting in smaller efficacy gains for each incremental increase in RoActemra concentration such that clinically meaningful increases in efficacy were not demonstrated in patients treated with >800 mg of RoActemra. Therefore, RoActemra doses exceeding 800 mg per infusion are not recommended (see section 4.2).
Distribution
In RA patients the central volume of distribution was 3.72 L, the peripheral volume of distribution was 3.35 L resulting in a volume of distribution at steady state of 7.07 L.
Elimination
Following intravenous administration, RoActemra undergoes biphasic elimination from the circulation. The total clearance of RoActemra was concentration-dependent and is the sum of the linear and non-linear clearance. The linear clearance was estimated as a parameter in the population pharmacokinetic analysis and was 9.5 mL/h. The concentration-dependent non-linear clearance plays a major role at low RoActemra concentrations. Once the non-linear clearance pathway is saturated, at higher RoActemra concentrations, clearance is mainly determined by the linear clearance.
The t½ of RoActemra was concentration-dependent. At steady-state following a dose of 8 mg/kg every 4 weeks, the effective t½ decreased with decreasing concentrations within a dosing interval from 18 days to 6 days.
Linearity
Pharmacokinetic parameters of RoActemra did not change with time. A more than dose-proportional increase in the AUC and Cmin was observed for doses of 4 and 8 mg/kg every 4 weeks. Cmax increased dose-proportionally. At steady-state, predicted AUC and Cmin were 3.2 and 30 fold higher at 8 mg/kg as compared to 4 mg/kg, respectively.
Subcutaneous use
The pharmacokinetics of RoActemra were determined using a population pharmacokinetic analysis on a database composed of 3552 RA patients treated with 162 mg subcutaneous every week, 162 mg subcutaneous every other week, and or 4 or 8 mg/kg intravenous every 4 weeks for 24 weeks.
The pharmacokinetic parameters of RoActemra did not change with time. For the 162 mg every week dose, the predicted mean (±SD) steady-state AUC1week, Cmin and Cmax of RoActemra were 7970 ± 3432 μg•h/mL, 43.0 ± 19.8 μg/mL, and 49.8 ± 21.0 μg/mL, respectively. The accumulation ratios for AUC, Cmin , and Cmax were 6.32, 6.30, and 5.27, respectively. Steady state was reached after 12 weeks for AUC, Cmin , and Cmax.
For the 162 every other week dose, the predicted mean (±SD) steady-state AUC2week, Cmin, and Cmax of RoActemra were 3430 ± 2660 μg•h/mL, 5.7 ± 6.8 μg/mL, and 13.2 ± 8.8 μg/mL, respectively. The accumulation ratios for AUC, Cmin, and Cmax were 2.67, 6.02, and 2.12, respectively. Steady state was reached after 12 weeks for AUC and Cmin, and after 10 weeks for Cmax.
Absorption
Following subcutaneous dosing in RA patients, the time to peak serum RoActemra concentrations tmax was 2.8 days. The bioavailability for the subcutaneous formulation was 79%.
Elimination
For subcutaneous administration, the effective t½ is up to 13 days for 162 mg every week and 5 days for 162 mg every other week in patients with RA at steady-state.
sJIA
Subcutaneous Use
The pharmacokinetics of RoActemra in sJIA patients was characterized by a population pharmacokinetic analysis which included 140 patients who were treated with 8 mg/kg IV every 2 weeks (patients weighing ≥30 kg), 12 mg/kg IV every 2 weeks (patients weighing below 30 kg), 162 mg SC every week (patients weighing ≥30 kg), 162 mg SC every 10 days or every 2 weeks (patients weighing below 30 kg).
Limited data are available regarding exposures following subcutaneous administration of RoActemra in sJIA patients below 2 years of age with a body weight less than 10 kg. Patients with sJIA must have a minimum body weight of 10 kg when receiving RoActemra subcutaneously (see section 4.2).
Table 8. Predicted mean ± SD PK parameters at steady-state after SC dosing in sJIA:
| RoActemra PK Parameter | 162 mg QW ≥30 kg | 162 mg Q2W below 30 kg |
|---|---|---|
| Cmax (µg/mL) | 99.8 ± 46.2 | 134 ± 58.6 |
| Cmin (µg/mL) | 79.2 ± 35.6 | 65.9 ± 31.3 |
| Cmean (µg/mL) | 91.3 ± 40.4 | 101 ± 43.2 |
| Accumulation Cmax | 3.66 | 1.88 |
| Accumulation Cmin | 4.39 | 3.21 |
| Accumulation Cmean or AUCτ* | 4.28 | 2.27 |
*τ = 1 week or 2 weeks for the two SC regimens
After SC dosing, approximately 90% of the steady-state was reached by week 12 for both the 162 mg QW and Q2W regimens.
Absorption
Following SC dosing in sJIA patients, the absorption half-life was around 2 days, and the bioavailability for the SC formulation in sJIA patients was 95%.
Distribution
In paediatric patients with sJIA, the central volume of distribution was 1.87 L, the peripheral volume of distribution was 2.14 L resulting in a volume of distribution at steady state of 4.01 L.
Elimination
The total clearance of tocilizumab was concentration-dependent and is the sum of the linear clearance and the nonlinear clearance. The linear clearance was estimated as a parameter in the population pharmacokinetic analysis and was 5.7 mL/h in paediatric patients with systemic juvenile idiopathic arthritis. Following subcutaneous administration, the effective t½ of RoActemra in sJIA patients is up to 14 days for both the 162 mg QW and Q2W regimens during a dosing interval at steady state.
pJIA
Subcutaneous use
The pharmacokinetics of RoActemra in pJIA patients was characterized by a population pharmacokinetic analysis which included 237 patients who were treated with 8 mg/kg IV every 4 weeks (patients weighing ≥ 30 kg), 10 mg/kg IV every 4 weeks (patients weighing below 30 kg), 162 mg SC every 2 weeks (patients weighing ≥30 kg), or 162 mg SC every 3 weeks (patients weighing below 30 kg).
Table 9. Predicted mean ± SD PK parameters at steady-state after SC dosing in pJIA:
| RoActemra PK Parameter | 162 mg Q2W ≥30 kg | 162 mg Q3W below 30 kg |
|---|---|---|
| Cmax (µg/mL) | 29.4 ± 13.5 | 75.5 ± 24.1 |
| Cmin (µg/mL) | 11.8 ± 7.08 | 18.4 ± 12.9 |
| Cavg (µg/mL) | 21.7 ± 10.4 | 45.5 ± 19.8 |
| Accumulation Cmax | 1.72 | 1.32 |
| Accumulation Cmin | 3.58 | 2.08 |
| Accumulation Cmean or AUCτ* | 2.04 | 1.46 |
* τ = 2 week or 3 week for the two SC regimens
After IV dosing, approximately 90% of the steady-state was reached by Week 12 for the 10 mg/kg (BW<30 kg), and by Week 16 for the 8 mg/kg (BW≥30 kg) dose. After SC dosing, approximately 90% of the steady-state was reached by Week 12 for both the 162 mg SC Q2W and Q3W regimens.
Absorption
Following SC dosing in pJIA patients, the absorption half-life was around 2 days, and the bioavailability for the SC formulation in pJIA patients was 96%.
Distribution
In paediatric patients with pJIA, the central volume of distribution was 1.97 L, the peripheral volume of distribution was 2.03 L, resulting in a volume of distribution at steady state of 4.0 L.
Elimination
Population pharmacokinetic analysis for pJIA patients showed body size related impact on linear clearance so that body-weight based dosing should be taken into consideration (see Table 9).
After subcutaneous administration, the effective t½ of RoActemra in pJIA patients is up to 10 days for patients <30 kg (162 mg SC Q3W) and up to 7 days for patients ≥30 kg (162 mg SC Q2W) during a dosing interval at steady state. Following intravenous administration, tocilizumab undergoes biphasic elimination from the circulation. The total clearance of tocilizumab was concentration-dependent and is the sum of the linear and non-linear clearance. The linear clearance was estimated as a parameter in the population pharmacokinetic analysis and was 6.25 mL/h. The concentration-dependent non-linear clearance plays a major role at low tocilizumab concentrations. Once the non- linear clearance pathway is saturated, at higher tocilizumab concentrations, clearance is mainly determined by the linear clearance.
GCA
Subcutaneous use
The PK of RoActemra in GCA patients were determined using a population PK model from an analysis dataset composed of 149 GCA patients treated with 162 mg subcutaneous every week or 162 mg subcutaneous every other week. The developed model had the same structure as the population PK model developed earlier based on data from RA patients (see Table 10).
Table 10. Predicted mean ± SD PK parameters at steady-state after subcutaneous dosing in GCA:
| Subcutaneous | ||
|---|---|---|
| Tocilizumab PK Parameter | 162 mg every other weekly | 162 mg weekly |
| Cmax (µg/mL) | 19.3 ± 12.8 | 73 ± 30.4 |
| Cmin (µg/mL) | 11.1 ± 10.3 | 68.1± 29.5 |
| Cmean (µg/mL) | 16.2 ± 11.8 | 71.3 ± 30.1 |
| Accumulation Cmax | 2.18 | 8.88 |
| Accumulation Cmin | 5.61 | 9.59 |
| Accumulation Cmean or AUCτ* | 2.81 | 10.91 |
* τ = 2 week or 1 week for the two SC regimens
The steady-state profile following the RoActemra weekly dose was almost flat, with very little fluctuations between trough and peak values, while there were substantial fluctuations for the RoActemra every other weekly dose. Approximately 90% of the steady-state (AUCτ) was reached by week 14 in the every other weekly and week 17 in the weekly dose groups.
Based on the current characterization of PK, RoActemra trough concentration at steady state are 50% higher in this population relative to average concentrations in a large dataset from the RA population. These differences occur due to unknown reasons. PK differences are not accompanied by marked differences in PD parameters and so the clinical relevance is unknown.
In GCA patients, higher exposure was observed in patients with lower body weight. For the 162 mg every week dosing regimen, the steady-state Cavg was 51% higher in patients with body weight less than 60 kg compared to patients weighing between 60 to 100 kg. For the 162 mg every other week regimen, the steady-state Cavg was 129% higher in patients with body weight less than 60 kg compared to patients weighing between 60 to 100 kg. There is limited data for patients above 100 kg (n=7).
Absorption
Following subcutaneous dosing in GCA patients, the absorption t1⁄2 was around 4 days. The bioavailability for the SC formulation was 0.8. The median values of Tmax were 3 days after the RoActemra weekly dose and 4.5 days after the tocilizumab every other week dose.
Distribution
In GCA patients, the central volume of distribution was 4.09 L, the peripheral volume of distribution was 3.37 L, resulting in a volume of distribution at steady state of 7.46 L.
Elimination
The total clearance of RoActemra was concentration-dependent and is the sum of the linear clearance and the nonlinear clearance. The linear clearance was estimated as a parameter in the population pharmacokinetic analysis and was 6.7 mL/h in GCA patients,
In GCA patients, at steady state, the effective t1⁄2 of RoActemra varied between 18.3 and 18.9 days for 162 mg weekly regimen, and between 4.2 and 7.9 days for 162 mg every other weekly regimen. At high serum concentrations, when total clearance of RoActemra is dominated by linear clearance, an effective t1⁄2 of approximately 32 days was derived from the population parameter estimates.
Special populations
Renal impairment: No formal study of the effect of renal impairment on the pharmacokinetics of RoActemra has been conducted. Most of the patients in the RA and GCA studies population pharmacokinetic analysis had normal renal function or mild renal impairment. Mild renal impairment (estimated creatinine clearance based on Cockcroft-Gault formula) did not impact the pharmacokinetics of RoActemra.
Approximately one-third of the patients in the GCA study had moderate renal impairment at baseline (estimated creatinine clearance of 30-59 mL/min). No impact on RoActemra exposure was noted in these patients.
No dose adjustment is required in patients with mild or moderate renal impairment.
Hepatic impairment: No formal study of the effect of hepatic impairment on the pharmacokinetics of RoActemra has been conducted.
Age, gender and ethnicity: Population pharmacokinetic analyses in RA and GCA patients, showed that age, gender and ethnic origin did not affect the pharmacokinetics of RoActemra.
Results of the population PK analysis for sJIA and pJIA patients confirmed that body size is the only covariate which has an appreciable impact on the pharmacokinetics of RoActemra including elimination and absorption so that body-weight based dosing should be taken into consideration (see Tables 8 and 9).
Preclinical safety data
Non-clinical data reveal no special hazard for humans based on conventional studies of safety pharmacology, repeated dose toxicity, genotoxicity and toxicity to reproduction and development.
Carcinogenicity studies were not performed because IgG1 monoclonal antibodies are not deemed to have intrinsic carcinogenic potential.
Available non-clinical data demonstrated the effect of IL-6 on malignant progression and apoptosis resistance to various cancer types. This data does not suggest a relevant risk for cancer initiation and progression under RoActemra treatment. Additionally, proliferative lesions were not observed in a 6-month chronic toxicity study in cynomolgus monkeys or in IL-6 deficient mice.
Available non-clinical data do not suggest an effect on fertility under RoActemra treatment. Effects on endocrine active and reproductive system organs were not observed in a chronic cynomolgus monkey toxicity study and reproductive performance was not affected in IL-6 deficient mice. RoActemra administered to cynomolgus monkeys during early gestation, was observed to have no direct or indirect harmful effect on pregnancy or embryonal-foetal development. However, a slight increase in abortion/embryonal-foetal death was observed with high systemic exposure (>100 x human exposure) in the 50 mg/kg/day high-dose group compared to placebo and other low-dose groups. Although IL-6 does not seem to be a critical cytokine for foetal growth or the immunological control of the maternal/foetal interface, a relation of this finding to RoActemra cannot be excluded.
Treatment with a murine analogue did not exert toxicity in juvenile mice. In particular, there was no impairment of skeletal growth, immune function and sexual maturation.
The non-clinical safety profile of RoActemra in the cynomolgus monkey does not suggest a difference between intravenous and subcutaneous routes of administration.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.