XIIDRA Οphthalmic solution Ref.[10118] Active ingredients: Lifitegrast
Source: FDA, National Drug Code (US) Revision Year: 2020
12.1. Mechanism of Action
Lifitegrast binds to the integrin LFA-1, a cell surface protein found on leukocytes and blocks the interaction of LFA-1 with its cognate ligand intercellular adhesion molecule-1 (ICAM-1). ICAM-1 may be overexpressed in corneal and conjunctival tissues in DED. LFA-1/ICAM-1 interaction can contribute to the formation of an immunological synapse resulting in T-cell activation and migration to target tissues. In vitro studies demonstrated that lifitegrast may inhibit T-cell adhesion to ICAM-1 in a human T-cell line and may inhibit secretion of inflammatory cytokines in human peripheral blood mononuclear cells. The exact mechanism of action of lifitegrast in DED is not known.
12.3. Pharmacokinetics
In a subset of DED patients (n=47) enrolled in a Phase 3 trial, the pre-dose (trough) plasma concentrations of lifitegrast were measured after 180 and 360 days of topical ocular dosing (one drop twice daily) with Xiidra (lifitegrast ophthalmic solution) 5%. A total of nine of the 47 patients (19%) had plasma lifitegrast trough concentrations above 0.5 ng/mL (the lower limit of assay quantitation). Trough plasma concentrations that could be quantitated ranged from 0.55 ng/mL to 3.74 ng/mL.
13.1. Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Animal studies have not been conducted to determine the carcinogenic potential of lifitegrast.
Mutagenesis
Lifitegrast was not mutagenic in the in vitro Ames assay. Lifitegrast was not clastogenic in the in vivo mouse micronucleus assay. In an in vitro chromosomal aberration assay using mammalian cells (Chinese hamster ovary cells), lifitegrast was positive at the highest concentration tested, without metabolic activation.
Impairment of Fertility
Lifitegrast administered at IV doses of up to 30 mg/kg/day (5400-fold the human plasma exposure at the RHOD of lifitegrast ophthalmic solution, 5%) had no effect on fertility and reproductive performance in male and female-treated rats.
14. Clinical Studies
The safety and efficacy of lifitegrast for the treatment of DED were assessed in a total of 1181 patients (1067 of which received lifitegrast 5%) in four 12-week, randomized, multi-center, double-masked, vehicle-controlled studies. Patients were randomized to Xiidra or vehicle (placebo) in a 1:1 ratio and dosed twice a day. Use of artificial tears was not allowed during the studies. The mean age was 59 years (range, 19-97 years). The majority of patients were female (76%). Enrollment criteria included minimal signs (i.e., Corneal Fluorescein Staining and non-anesthetized Schirmer Tear Test) and symptoms (i.e., Eye Dryness Score (EDS) and Ocular Discomfort Score) severity scores at baseline.
Effects on Symptoms of Dry Eye Disease
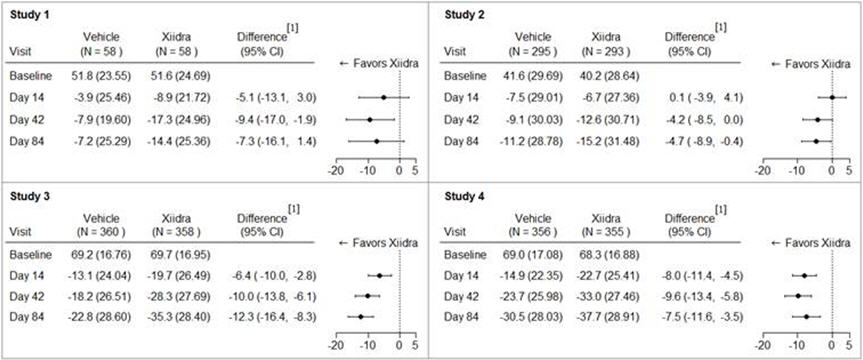
Eye dryness score was rated by patients using a visual analogue scale (0 = no discomfort, 100 = maximal discomfort) at each study visit. The average baseline EDS was between 40 and 70. A larger reduction in EDS favoring Xiidra was observed in all studies at Day 42 and Day 84 (see Figure 1).
Figure 1. Mean Change (SD) From Baseline and Treatment Difference (Xiidra – Vehicle) in Eye Dryness Score in 12-Week Studies in Patients With Dry Eye Disease:
[1] Based on analysis of covariance (ANCOVA) model adjusted for baseline value in Study 1, and ANCOVA model adjusted for baseline value and randomization stratification factors in Studies 2-4. All randomized and treated patients were included in the analysis and missing data were imputed using last-available data. In Study 1, one Xiidra-treated subject who did not have a baseline value was excluded from analysis.
Effects on Signs of Dry Eye Disease
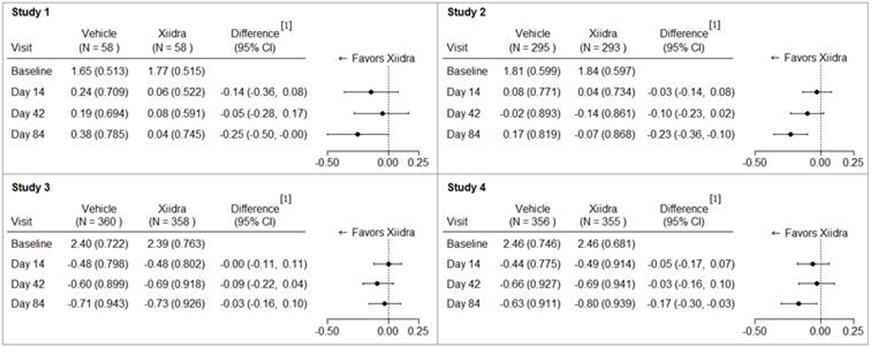
Inferior fluorescein corneal staining score (ICSS) (0 = no staining, 1 = few/rare punctate lesions, 2 = discrete and countable lesions, 3 = lesions too numerous to count but not coalescent, 4 = coalescent) was recorded at each study visit. The average baseline ICSS was approximately 1.8 in Studies 1 and 2, and 2.4 in Studies 3 and 4. At Day 84, a larger reduction in ICSS favoring Xiidra was observed in three of the four studies (see Figure 2).
Figure 2. Mean Change (SD) From Baseline and Treatment Difference (Xiidra – Vehicle) in Inferior Corneal Staining Score in 12-Week Studies in Patients With Dry Eye Disease:
[1] Based on ANCOVA model adjusted for baseline value in Study 1, and ANCOVA model adjusted for baseline value and randomization stratification factors in Studies 2-4. All randomized and treated patients were included in the analysis and missing data were imputed using last-available data. In Study 2, one vehicle-treated subject who did not have a study eye designated was excluded from analysis.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.