ALOXI Solution for injection Ref.[8704] Active ingredients: Palonosetron
Source: European Medicines Agency (EU) Revision Year: 2018 Publisher: Helsinn Birex Pharmaceuticals Ltd., Damastown, Mulhuddart, Dublin 15, Ireland
Pharmacodynamic properties
Pharmacotherapeutic group: Antiemetics and antinauseants, serotonin (5HT3) antagonists
ATC code: A04AA05
Palonosetron is a selective high-affinity receptor antagonist of the 5HT3 receptor.
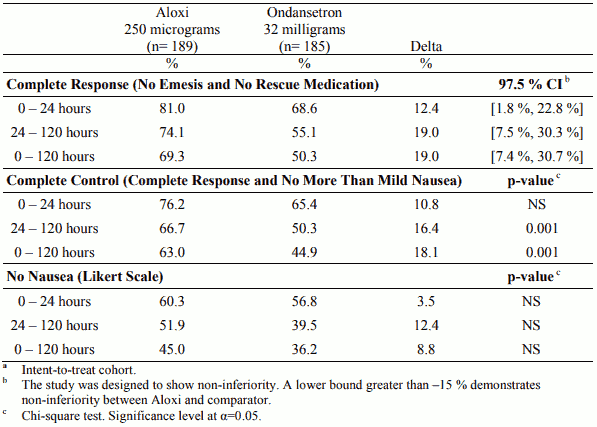
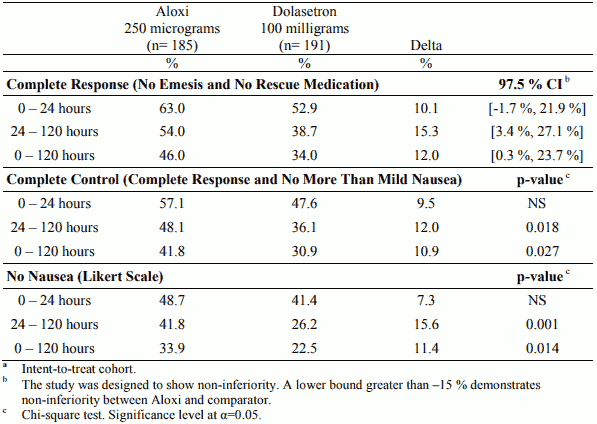
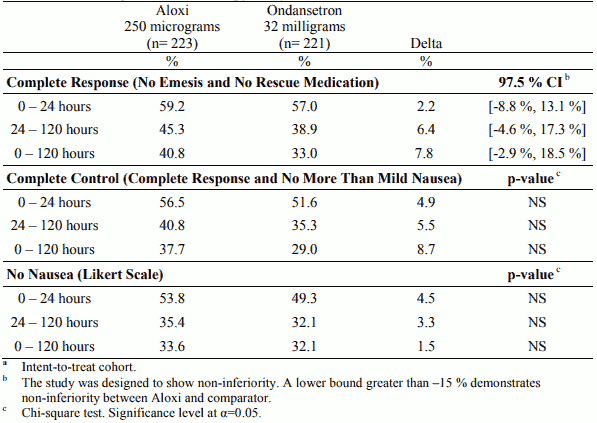
In two randomised, double-blind studies with a total of 1,132 patients receiving moderately emetogenic chemotherapy that included cisplatin ≤50 mg/m², carboplatin, cyclophosphamide ≤1,500 mg/m² and doxorubicin >25 mg/m², palonosetron 250 micrograms and 750 micrograms were compared with ondansetron 32 mg (half-life 4 hours) or dolasetron 100 mg (half-life 7.3 hours) administered intravenously on Day 1, without dexamethasone. In a randomised, double-blind study with a total of 667 patients receiving highly emetogenic chemotherapy that included cisplatin ≥ 60 mg/m², cyclophosphamide >1,500 mg/m² and dacarbazine, palonosetron 250 micrograms and 750 micrograms were compared with ondansetron 32 mg administered intravenously on Day 1. Dexamethasone was administered prophylactically before chemotherapy in 67% of patients.
The pivotal studies were not designed to assess efficacy of palonosetron in delayed onset nausea and vomiting. The antiemetic activity was observed during 0-24 hours, 24-120 hours and 0-120 hours. Results for the studies on moderately emetogenic chemotherapy and for the study on highly emetogenic chemotherapy are summarised in the following tables.
Palonosetron was non-inferior versus the comparators in the acute phase of emesis both in moderately and highly emetogenic setting.
Although comparative efficacy of palonosetron in multiple cycles has not been demonstrated in controlled clinical studies, 875 patients enrolled in the three phase 3 trials continued in an open label safety study and were treated with palonosetron 750 micrograms for up to 9 additional cycles of chemotherapy. The overall safety was maintained during all cycles.
Table 1. Percentage of patientsa responding by treatment group and phase in the Moderately Emetogenic Chemotherapy study versus ondansetron:
Table 2. Percentage of patientsa responding by treatment group and phase in the Moderately Emetogenic Chemotherapy study versus dolasetron:
Table 3. Percentage of patientsa responding by treatment group and phase in the Highly Emetogenic Chemotherapy study versus ondansetron:
The effect of palonosetron on blood pressure, heart rate, and ECG parameters including QTc were comparable to ondansetron and dolasetron in CINV clinical studies. In non-clinical studies palonosetron possesses the ability to block ion channels involved in ventricular de- and re-polarisation and to prolong action potential duration.
The effect of palonosetron on QTc interval was evaluated in a double blind, randomised, parallel, placebo and positive (moxifloxacin) controlled trial in adult men and women. The objective was to evaluate the ECG effects of IV administered palonosetron at single doses of 0.25, 0.75 or 2.25 mg in 221 healthy subjects. The study demonstrated no effect on QT/QTc interval duration as well as any other ECG interval at doses up to 2.25 mg. No clinically significant changes were shown on heart rate, atrioventricular (AV) conduction and cardiac repolarisation.
Paediatric population
Prevention of Chemotherapy Induced Nausea and Vomiting (CINV)
The safety and efficacy of Palonosetron i.v at single doses of 3 µg/kg and 10 µg/kg was investigated in the first clinical study in 72 patients in the following age groups, >28 days to 23 months (12 patients), 2 to 11 years (31 patients), and 12 to 17 years of age (29 patients), receiving highly or moderately emetogenic chemotherapy. No safety concerns were raised at either dose level. The primary efficacy variable was the proportion of patients with a complete response (CR, defined as no emetic episode and no rescue medication) during the first 24 hours after the start of chemotherapy administration. Efficacy after palonosetron 10 µg/kg compared to palonosetron 3µg/kg was 54.1% and 37.1% respectively.
The efficacy of Aloxi for the prevention of chemotherapy-induced nausea and vomiting in paediatric cancer patients was demonstrated in a second non-inferiority pivotal trial comparing a single intravenous infusion of palonosetron versus an i.v. ondansetron regimen. A total of 493 paediatric patients, aged 64 days to 16.9 years, receiving moderately (69.2%) or highly emetogenic chemotherapy (30.8%) were treated with palonosetron 10 µg/kg (maximum 0.75 mg), palonosetron 20 µg/kg (maximum 1.5 mg) or ondansetron (3 x 0.15 mg/kg, maximum total dose 32 mg) 30 minutes prior to the start of emetogenic chemotherapy during Cycle 1. Most patients were non-naïve to chemotherapy (78.5%) across all treatment groups. Emetogenic chemotherapies administered included doxorubicin, cyclophosphamide (<1500 mg/m²), ifosfamide, cisplatin, dactinomycin, carboplatin, and daunorubicin. Adjuvant corticosteroids, including dexamethasone, were administered with chemotherapy in 55% of patients. The primary efficacy endpoint was Complete Response in the acute phase of the first cycle of chemotherapy, defined as no vomiting, no retching, and no rescue medication in the first 24 hours after starting chemotherapy. Efficacy was based on demonstrating non-inferiority of intravenous palonosetron compared to intravenous ondansetron. Non-inferiority criteria were met if the lower bound of the 97.5% confidence interval for the difference in Complete Response rates of intravenous palonosetron minus intravenous ondansetron was larger than -15%. In the palonosetron 10 µg/kg, 20 µg/kg and ondansetron groups, the proportion of patients with CR0-24h was 54.2%, 59.4% and 58.6%. Since the 97.5% confidence interval (stratum adjusted MantelHaenszel test) of the difference in CR0-24h between palonosetron 20 µg/kg and ondansetron was [-11.7%, 12.4%], the 20 µg/kg palonosetron dose demonstrated non-inferiority to ondansetron.
While this study demonstrated that paediatric patients require a higher palonosetron dose than adults to prevent chemotherapy-induced nausea and vomiting, the safety profile is consistent with the established profile in adults (see section 4.8). Pharmacokinetic information is provided in section 5.2.
Prevention of Post Operative Nausea and Vomiting (PONV)
Two paediatric trials were performed. The safety and efficacy of Palonosetron i.v at single doses of 1µg/kg and 3µg/kg was compared in the first clinical study in 150 patients in the following age groups, >28 days to 23 months (7 patients), 2 to 11 years (96 patients), and 12 to 16 years of age (47 patients) undergoing elective surgery. No safety concerns were raised in either treatment group. The proportion of patients without emesis during 0-72 hours post-operatively was similar after palonosetron 1 µg/kg or 3 µg/kg (88% vs 84%).
The second paediatric,trial was a multicenter, double-blind, double-dummy, randomised, parallel group, active control, single-dose non-inferiority study, comparing i.v. palonosetron (1 µg/kg, max 0.075 mg) versus I.V. ondansetron. A total of 670 paediatric surgical patients participated, age 30 days to 16.9 years. The primary efficacy endpoint, Complete Response (CR: no vomiting, no retching, and no antiemetic rescue medication) during the first 24 hours postoperatively was achieved in 78.2% of patients in the palonosetron group and 82.7% in the ondansetron group. Given the pre-specified non-inferiority margin of -10%, the stratum adjusted Mantel-Haenszel statistical non-inferiority confidence interval for the difference in the primary endpoint, complete response (CR), was [-10.5, 1.7%], therefore non-inferiority was not demonstrated. No new safety concerns were raised in either treatment group.
Please see section 4.2 for information on paediatric use.
Pharmacokinetic properties
Absorption
Following intravenous administration, an initial decline in plasma concentrations is followed by slow elimination from the body with a mean terminal elimination half-life of approximately 40 hours. Mean maximum plasma concentration (Cmax) and area under the concentration-time curve (AUC0-∞) are generally dose-proportional over the dose range of 0.3–90 μg/kg in healthy subjects and in cancer patients.
Following intravenous administration of palonosetron 0.25 mg once every other day for 3 doses in 11 testicular cancer patients, the mean (±SD) increase in plasma concentration from Day 1 to Day 5 was 42±34%. After intravenous administration of palonosetron 0.25 mg once daily for 3 days in 12 healthy subjects, the mean (±SD) increase in plasma palonosetron concentration from Day 1 to Day 3 was 110±45%.
Pharmacokinetic simulations indicate that the overall exposure (AUC0-∞) of 0.25 mg intravenous palonosetron administered once daily for 3 consecutive days was similar to a single intravenous dose of 0.75 mg, although Cmax of the 0.75 mg single dose was higher.
Distribution
Palonosetron at the recommended dose is widely distributed in the body with a volume of distribution of approximately 6.9 to 7.9 l/kg. Approximately 62% of palonosetron is bound to plasma proteins.
Biotransformation
Palonosetron is eliminated by dual route, about 40% eliminated through the kidney and with approximately 50% metabolised to form two primary metabolites, which have less than 1% of the 5HT3 receptor antagonist activity of palonosetron. In vitro metabolism studies have shown that CYP2D6 and to a lesser extent, CYP3A4 and CYP1A2 isoenzymes are involved in the metabolism of palonosetron. However, clinical pharmacokinetic parameters are not significantly different between poor and extensive metabolisers of CYP2D6 substrates. Palonosetron does not inhibit or induce cytochrome P450 isoenzymes at clinically relevant concentrations.
Elimination
After a single intravenous dose of 10 micrograms/kg [14C]-palonosetron, approximately 80% of the dose was recovered within 144 hours in the urine with palonosetron representing approximately 40% of the administered dose, as unchanged active substance. After a single intravenous bolus administration in healthy subjects the total body clearance of palonosetron was 173±73 ml/min and renal clearance was 53±29 ml/min. The low total body clearance and large volume of distribution resulted in a terminal elimination half-life in plasma of approximately 40 hours. Ten percent of patients have a mean terminal elimination half-life greater than 100 hours.
Pharmacokinetics in special populations
Elderly people
Age does not affect the pharmacokinetics of palonosetron. No dosage adjustment is necessary in elderly patients.
Gender
Gender does not affect the pharmacokinetics of palonosetron. No dosage adjustment is necessary based on gender.
Paediatric population
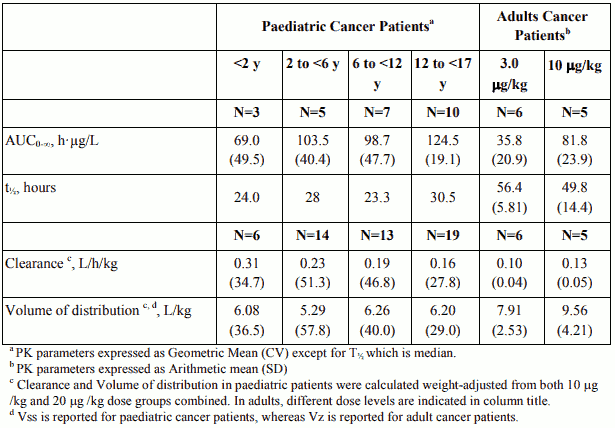
Single-dose i.v. Aloxi pharmacokinetic data was obtained from a subset of paediatric cancer patients (n=280) that received 10 µg/kg or 20 µg/kg. When the dose was increased from10 µg/kg to 20 µg/kg a dose-proportional increase in mean AUC was observed. Following single dose intravenous infusion of Aloxi 20 µg/kg, peak plasma concentrations (CT) reported at the end of the 15 minute infusion were highly variable in all age groups and tended to be lower in patients <6 years than in older paediatric patients. Median half-life was 29.5 hours in overall age groups and ranged from about 20 to 30 hours across age groups after administration of 20 µg/kg.
The total body clearance (L/h/kg) in patients 12 to 17 years old was similar to that in healthy adults. There are no apparent differences in volume of distribution when expressed as L/kg.
Table 4. Pharmacokinetic Parameters in Paediatric Cancer Patients following intravenous infusion of Aloxi at 20 µg/kg over 15 min and in Adult Cancer Patients receiving 3 and 10 µg/kg palonosetron doses via intravenous bolus:
Renal impairment
Mild to moderate renal impairment does not significantly affect palonosetron pharmacokinetic parameters. Severe renal impairment reduces renal clearance, however total body clearance in these patients is similar to healthy subjects. No dosage adjustment is necessary in patients with renal insufficiency. No pharmacokinetic data in haemodialysis patients are available.
Hepatic impairment
Hepatic impairment does not significantly affect total body clearance of palonosetron compared to the healthy subjects. While the terminal elimination half-life and mean systemic exposure of palonosetron is increased in the subjects with severe hepatic impairment, this does not warrant dose reduction.
Preclinical safety data
Effects in non-clinical studies were observed only at exposures considered sufficiently in excess of the maximum human exposure indicating little relevance to clinical use.
Non-clinical studies indicate that palonosetron, only at very high concentrations, may block ion channels involved in ventricular de- and re-polarisation and prolong action potential duration.
Animal studies do not indicate direct or indirect harmful effects with respect to pregnancy, embryonal/foetal development, parturition or postnatal development. Only limited data from animal studies are available regarding the placental transfer (see section 4.6).
Palonosetron is not mutagenic. High doses of palonosetron (each dose causing at least 30 times the human therapeutic exposure) applied daily for two years caused an increased rate of liver tumours, endocrine neoplasms (in thyroid, pituitary, pancreas, adrenal medulla) and skin tumours in rats but not in mice. The underlying mechanisms are not fully understood, but because of the high doses employed and since Aloxi is intended for single application in humans, these findings are not considered relevant for clinical use.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.