BYSANTI Tablet Ref.[116258] Active ingredients: Milsaperidone
Source: FDA, National Drug Code (US) Revision Year: 2026
Product description
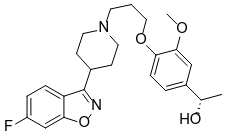
The active ingredient in BYSANTI is milsaperidone, an atypical antipsychotic that is in the piperidinyl-benzisoxazole derivative chemical class. Its chemical name is benzenemethanol, 4-[3-[4-(6-fluoro-1,2-benzisoxazol-3-yl)-1-piperidinyl]propoxy]-3-methoxy-α- methyl-, (αS)-. Its molecular formula is C24H29FN2O4 and its molecular weight is 428.50. Milsaperidone is a white to off-white finely crystalline powder. The structural formula of milsaperidone is:
Milsaperidone is a white to off-white finely crystalline powder.
BYSANTI (milsaperidone) tablets are for oral administration only. Coated tablets contain 1 mg, 2 mg, 4 mg, 6 mg, 8 mg, 10 mg, or 12 mg of milsaperidone. Inactive ingredients are the following: colloidal silicon dioxide, crospovidone, hydroxypropylmethylcellulose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and water (removed during processing).
| Dosage Forms and Strengths |
|---|
|
Tablets:
|
| How Supplied | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
BYSANTI (milsaperidone) tablets are supplied as follows:
Table 9 displays the package configurations for the single strength packages of BYSANTI (milsaperidone) tablets and Table 10 displays the package configuration for the titration packs. Table 9. Package Configurations for the Single Strength Packages of BYSANTI (milsaperidone) Tablets:
Table 10. Package Configurations for the Titration Packs:
Distributed by: Vanda Pharmaceuticals Inc., Washington, D.C. 20037 USA |
Drugs
| Drug | Countries | |
|---|---|---|
| BYSANTI | United States |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.
 " logo on other side
" logo on other side