LUCEMYRA Film-coated tablet Ref.[10120] Active ingredients: Lofexidine
Source: FDA, National Drug Code (US) Revision Year: 2019
Product description
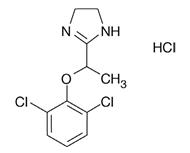
LUCEMYRA tablets contain lofexidine, a central alpha-2 adrenergic agonist, as the hydrochloride salt. Lofexidine hydrochloride is chemically designated as 2-[1-(2,6-dichlorophenoxy)ethyl]-4,5 dihydro-1H-imidazole monohydrochloride with a molecular formula of C11H12Cl2N2O∙HCl.
Its molecular weight is 295.6 g/mole and its structural formula is:
Lofexidine hydrochloride is a white to off-white crystalline powder freely soluble in water, methanol, and ethanol. It is slightly soluble in chloroform and practically insoluble in n-hexane and benzene.
LUCEMYRA is available as round, convex-shaped, peach-colored, film-coated tablets for oral administration. Each tablet contains 0.18 lofexidine, equivalent to 0.2 mg of lofexidine hydrochloride, and the following inactive ingredients: 92.6 mg lactose, 12.3 mg citric acid, 1.1 mg povidone, 5.7 mg microcrystalline cellulose, 1.4 mg calcium stearate, 0.7 mg sodium lauryl sulphate, and Opadry OY S 9480 (contains indigo carmine and sunset yellow).
| Dosage Forms and Strengths |
|---|
|
LUCEMYRA is available as round, peach-colored, film-coated tablets, imprinted with "LFX" on one side and "18" on the other side. Each tablet contains 0.18 mg lofexidine (equivalent to 0.2 mg of lofexidine hydrochloride). |
| How Supplied | ||||
|---|---|---|---|---|
|
Available as 0.18 mg round, convex-shaped, peach colored, film-coated tablets, imprinted with "LFX" on one side and "18" on the other side; approximately 7 mm in diameter.
|
Drugs
| Drug | Countries | |
|---|---|---|
| LUCEMYRA | United States |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.