MYQORZO Film-coated tablet Ref.[116119] Active ingredients: Aficamten
Source: FDA, National Drug Code (US) Revision Year: 2025
Product description
MYQORZO tablets for oral use contain aficamten, a cardiac myosin inhibitor.
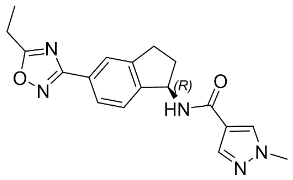
The chemical name of aficamten is (R)-N-(5-(5-ethyl-1,2,4-oxadiazol-3-yl)-2,3-dihydro-1H-inden-1-yl)-1-methyl-1H-pyrazole-4-carboxamide. The molecular formula is C18H19N5O2 and the molecular weight is 337.38 g/mol.
The structural formula of aficamten is:
Aficamten is a white to off-white to yellow to grey solid that is practically insoluble in water and aqueous buffers (pH 2 – 9), sparingly soluble in acetone and ethanol, and soluble in N-methylpyrrolidone (NMP).
MYQORZO is supplied as tablets containing 5 mg, 10 mg, 15 mg, or 20 mg of aficamten per tablet as the active ingredient and the following inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, magnesium stearate (non-bovine), mannitol, microcrystalline cellulose, and sodium lauryl sulfate. The tablet coating contains carmine, FD&C Blue No. 2, glyceryl mono and dicaprylate, polyvinyl alcohol, polyvinyl alcohol graft polyethylene copolymer, talc and titanium dioxide.
| Dosage Forms and Strengths |
|---|
|
MYQORZO is available as tablets in the following strengths: Tablets: 5 mg - purple, round tablet debossed with "5" on one side and "CK" on the other side. Tablets: 10 mg - purple, triangular tablet debossed with "10" on one side and "CK" on the other side. Tablets: 15 mg - purple, pentagonal tablet debossed with "15" on one side and "CK" on the other side. Tablets: 20 mg - purple, oval tablet debossed with "20" on one side and "CK" on the other side. |
| How Supplied | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
MYQORZO is supplied as purple, film-coated tablets containing 5 mg, 10 mg, 15 mg, or 20 mg aficamten. The tablets are debossed on one side with "CK" and the other side with "5", "10", "15" and "20" for the 5, 10, 15, and 20 mg strength, respectively. Tablets are supplied in bottles with child-resistant closure as follows:
Distributed By: Cytokinetics Incorporated, South San Francisco, CA 94080 |
Drugs
| Drug | Countries | |
|---|---|---|
| MYQORZO | Lithuania, United States |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.