ORAVERSE Solution for injection Ref.[10888] Active ingredients: Phentolamine
Source: FDA, National Drug Code (US) Revision Year: 2018
Product description
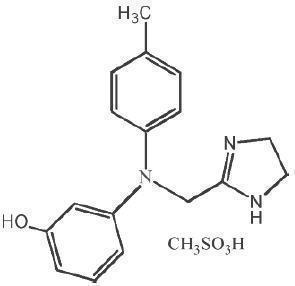
Phentolamine mesylate is phenol,3-[[(4,5-dihydro-1H-imidazol-2-yl)methyl](4-methyl-phenyl)amino]-,methanesulfonate (salt), a non-specific alpha adrenergic blocker.
Phentolamine mesylate USP is a white to off-white, odorless crystalline powder with a molecular weight of 377.46. It is sparingly soluble in water, soluble in alcohol, and slightly soluble in chloroform. The empirical formulation is C17H19N3O•CH4O3S, and the chemical structure is:
OraVerse (phentolamine mesylate) Injection is a clear, colorless, sterile, non pyrogenic, isotonic, preservative free solution. Each 1.7 mL cartridge contains 0.4 mg phentolamine mesylate, D-mannitol, edetate disodium, and sodium acetate. Either acetic acid or sodium hydroxide is used as necessary to adjust the pH.
| Dosage Forms and Strengths |
|---|
|
0.4 mg/1.7 mL solution per cartridge. |
| How Supplied |
|---|
|
OraVerse (phentolamine mesylate) Injection 0.4 mg/1.7 mL is supplied in a dental cartridge, in cartons of 10 and 50 cartridges. Each cartridge is individually packaged in a separate compartment of a 10 cartridge blister pack. NDC 0362-0101-50 Manufactured by Novocol Pharmaceutical of Canada, Inc., Cambridge, Ontario, Canada for Septodont, Inc, Louisville, CO 80027 |
Drugs
| Drug | Countries | |
|---|---|---|
| ORAVERSE | Canada, France, United States |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.