ORAVERSE Solution for injection Ref.[10888] Active ingredients: Phentolamine
Source: FDA, National Drug Code (US) Revision Year: 2018
12.1. Mechanism of Action
The mechanism by which OraVerse accelerates reversal of soft-tissue anesthesia and the associated functional deficits is not fully understood. Phentolamine mesylate, the active ingredient in OraVerse, produces an alpha-adrenergic block of relatively short duration resulting in vasodilatation when applied to vascular smooth muscle. In an animal model, OraVerse increased local blood flow in submucosal tissue of the dog when given after an intraoral injection of lidocaine 2% with 1:100,000 epinephrine.
12.3. Pharmacokinetics
Following OraVerse administration, phentolamine is 100% available from the submucosal injection site and peak concentrations are achieved 10-20 minutes after injection. Phentolamine systemic exposure increased linearly after 0.8 mg compared to 0.4 mg OraVerse intraoral submucosal injection. The terminal elimination half-life of phentolamine in the blood was approximately 2-3 hours.
Pediatrics
Following OraVerse administration, the phentolamine Cmax was higher (approximately 3.5-fold) in children who weighed between 15 and 30 kg (33 and 66 lbs) than in children who weighed more than 30 kg. However, phentolamine AUC was similar between the two groups. It is recommended that in children weighing 15-30 kg, the maximum dose of OraVerse should be limited to ½ cartridge (0.2 mg) (see Dosage and Administration section).
The pharmacokinetics of OraVerse in adults and in children who weighed more than 30 kg (66 lbs) are similar after intraoral submucosal injection.
OraVerse has not been studied in children under 3 years of age or weighing less than 15 kg (33 lbs). The pharmacokinetics of OraVerse after administration of more than 1 cartridge (0.4mg) has not been studied in children.
13.1. Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Carcinogenic studies with OraVerse have not been conducted.
Mutagenesis
Phentolamine was not mutagenic in the in-vitro bacterial reverse mutation (Ames) assay. In the in-vitro chromosomal aberration study in Chinese hamster ovary cells, numerical aberrations were slightly increased after a 4-hour exposure to phentolamine without metabolic activation and structural aberrations were slightly increased after a 4-hour exposure to phentolamine with metabolic activation only at the highest concentrations tested, but neither numerical nor structural aberrations were increased after a 20-hour exposure without metabolic activation. Phentolamine was not clastogenic in two in-vivo mouse micronucleus assays.
Impairment of Fertility
The effect of phentolamine on female fertility has not been studied. Male rats treated with oral phentolamine for nine weeks (four weeks prior to mating, 3 weeks during the mating period and 2 weeks after mating) were mated with untreated females. At doses up to 143-times human therapeutic exposure levels at the Cmax, no adverse effects on male fertility parameters or on reproductive parameters in the untreated females mated with the treated males were observed.
14. Clinical Studies
The safety and efficacy of OraVerse when used for reversal of soft-tissue anesthesia (STA), i.e., anesthesia of the lips and tongue following a dental procedure that required local anesthesia containing a vasoconstrictor, were evaluated in the following clinical studies. OraVerse-induced reversal of local anesthetic effects on the teeth, mandible and maxilla has not been assessed.
Two Phase 3, double-blinded, randomized, multi-center, controlled studies were conducted in dental patients who had mandibular (Study 1) or maxillary (Study 2) restorative or periodontal maintenance procedures and who had received a local anesthetic that contained a vasoconstrictor. The primary endpoint was time to normal lip sensation as measured by patient reported responses to lip palpation. The secondary endpoints included patients' perception of altered function, sensation and appearance, and their actual functional deficits in smiling, speaking, drinking and drooling, as assessed by both the patient and an observer blinded to the treatment. In the mandibular study, the time to recovery of tongue sensation was also a secondary endpoint. Patients were stratified by type and amount of anesthetic administered. OraVerse was administered at a cartridge ratio of 1:1 to local anesthetic. The control was a sham injection.
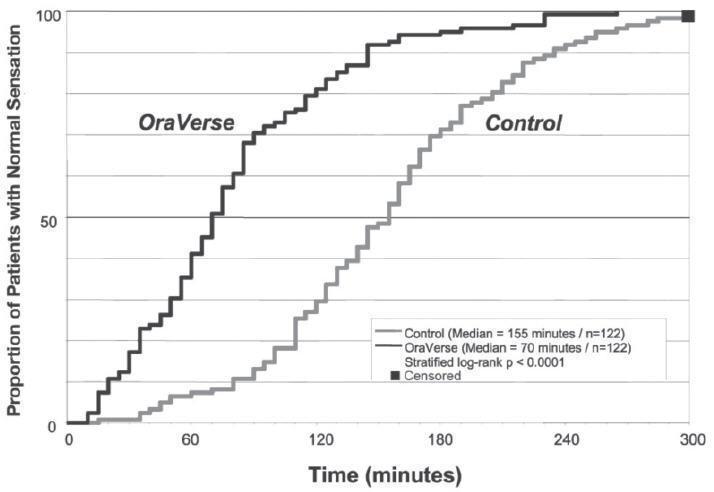
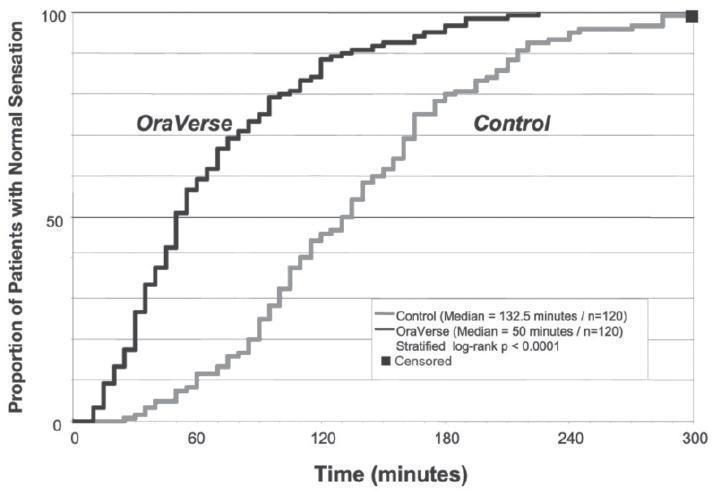
OraVerse reduced the median time to recovery of normal sensation in the lower lip by 85 minutes (55%) compared to control. The median time to recovery of normal sensation in the upper lip was reduced by 83 minutes (62%). The differences between these times for both studies are depicted in Kaplan-Meier plots for time to normal lip sensation in
Figures 1 and 2. Within 1 hour after administration of OraVerse, 41% of patients reported normal lower lip sensation as compared to 7% in the control group, and 59% of patients in the OraVerse group reported normal upper lip sensation as compared to 12% in the control group.
Figure 1. Kaplan-Meier Plot of Time to Recovery of Normal Sensation in the Lower Lip (ITT Analysis Data Set):
Figure 2. Kaplan-Meier Plot of Time to Recovery of Normal Sensation in the Upper Lip (ITT Analysis Data Set):
In Study 1 (mandibular), OraVerse accelerated: a) the recovery of the perception of normal appearance and function by 60 minutes (40%), b) the recovery of normal function by 60 minutes (50%), and c) the recovery of normal sensation in the tongue by 65 minutes (52%). In Study 2 (maxillary), the recovery of the perception of normal appearance and function was reduced by 60 minutes (50%) and the recovery of normal function was reduced by 45 minutes (43%).
Study 3, a pediatric, Phase 2, double-blinded, randomized, multi-center, controlled study was conducted in dental patients who had received 2% lidocaine with 1:100,000 epinephrine. Dental patients (n=152, ages 4-11 years) received ½ cartridge of OraVerse if they weighed ≥15 kg but <30 kg, and one-half or one full cartridge if they weighed ≥30 kg at a cartridge ratio of 1:1 to local anesthetic.
The median time to normal lip sensation in patients 6 to 11years of age who were trainable in the lip-palpation procedures, for mandibular and maxillary procedures combined, was reduced by 75 minutes (56%). Within 1 hour after administration of OraVerse, 44 patients (61%) reported normal lip sensation, while 9 patients (21%) randomized to the control group reported normal lip sensation. In this study, neither the patients' perception of their appearance or ability to function nor their actual ability to function was evaluated.
Study 4, a pediatric, Phase 4, double-blinded, randomized, multi-center, controlled study was conducted in dental patients undergoing mandibular and maxillary procedures after receiving 2% lidocaine with 1:100,000 epinephrine. Patients 2-5 years of age received sham injection (n=51) or ¼ cartridge of OraVerse if they weighed ≥10 kg but <15kg (n=5), ½ cartridge if they weighed ≥15 kg but <30kg (n=91), and a full cartridge if they weighed >30kg (n=3). This study was not designed to demonstrate efficacy.
The median time to normal lip sensation in patients 4 and 5 years of age who were trainable in the lip-palpation procedure, for mandibular and maxillary procedures combined, was reduced by 48 minutes (44%). Within 2 hours after administration of OraVerse, 57 patients (80%) reported normal lip sensation, while 19 patients (51%) randomized to the sham injection group reported normal lip sensation. There were no significant differences between OraVerse and sham injection for time to return of normal function in pediatric functional assessment battery and time to recovery of normal tongue sensation (for mandibular procedures only).
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.