Insulin glargine
Chemical formula: C₂₆₇H₄₀₄N₇₂O₇₈S₆ Molecular mass: 6,060.825 g/mol
Mechanism of action
Insulin glargine is a human insulin analogue designed to have a low solubility at neutral pH. It is completely soluble at the acidic pH of the insulin glargine injection solution (pH 4). After injection into the subcutaneous tissue, the acidic solution is neutralised leading to formation of micro-precipitates from which small amounts of insulin glargine are continuously released, providing a smooth, peakless, predictable concentration/time profile with a prolonged duration of action.
Insulin glargine is metabolised into 2 active metabolites M1 and M2.
Pharmacodynamic properties
Insulin receptor binding
In vitro studies indicate that the affinity of insulin glargine and its metabolites M1 and M2 for the human insulin receptor is similar to the one of human insulin.
IGF-1 receptor binding: The affinity of insulin glargine for the human IGF-1 receptor is approximately 5 to 8-fold greater than that of human insulin (but approximately 70 to 80-fold lower than the one of IGF-1), whereas M1 and M2 bind the IGF-1 receptor with slightly lower affinity compared to human insulin.
The total therapeutic insulin concentration (insulin glargine and its metabolites) found in type 1 diabetic patients was markedly lower than what would be required for a half maximal occupation of the IGF-1 receptor and the subsequent activation of the mitogenic-proliferative pathway initiated by the IGF-1 receptor. Physiological concentrations of endogenous IGF-1 may activate the mitogenic-proliferative pathway; however, the therapeutic concentrations found in insulin therapy, including in insulin glargine therapy, are considerably lower than the pharmacological concentrations required to activate the IGF-1 pathway.
Pharmacodynamic effects
The primary activity of insulin, including insulin glargine, is regulation of glucose metabolism. Insulin and its analogues lower blood glucose levels by stimulating peripheral glucose uptake, especially by skeletal muscle and fat, and by inhibiting hepatic glucose production. Insulin inhibits lipolysis in the adipocyte, inhibits proteolysis and enhances protein synthesis.
In clinical pharmacology studies, intravenous insulin glargine and human insulin have been shown to be equipotent when given at the same doses. As with all insulins, the time course of action of insulin glargine may be affected by physical activity and other variables.
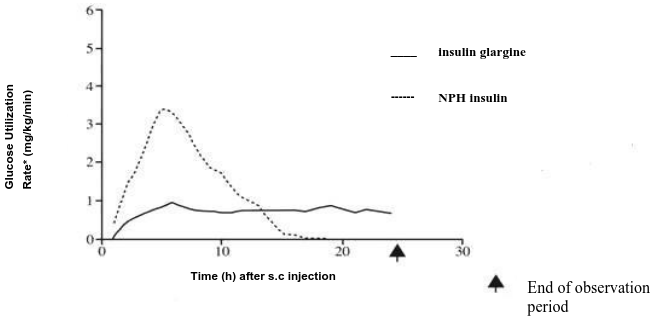
In euglycaemic clamp studies in healthy subjects or in patients with type 1 diabetes, the onset of action of subcutaneous insulin glargine was slower than with human NPH insulin, its effect profile was smooth and peakless, and the duration of its effect was prolonged.
The following graph shows the results from a study in patients:
Figure 1. Activity profile in patients with type 1 diabetes:
* Determined as amount of glucose infused to maintain constant plasma glucose levels (hourly mean values)
The longer duration of action of subcutaneous insulin glargine is directly related to its slower rate of absorption and supports once daily administration. The time course of action of insulin and insulin analogues such as insulin glargine may vary considerably in different individuals or within the same individual
In a clinical study, symptoms of hypoglycaemia or counter-regulatory hormone responses were similar after intravenous insulin glargine and human insulin both in healthy volunteers and patients with type 1 diabetes.
Pharmacokinetic properties
Absorption
In healthy subjects and diabetic patients, insulin serum concentrations indicated a slower and much more prolonged absorption and showed a lack of a peak after subcutaneous injection of insulin glargine in comparison to human NPH insulin. Concentrations were thus consistent with the time profile of the pharmacodynamic activity of insulin glargine. Figure 1 shows the activity profiles over time of insulin glargine and NPH insulin.
Insulin glargine injected once daily will reach steady state levels in 2-4 days after the first dose.
Figure 1. Activity profile in patients with type 1 diabetes:
* Determined as amount of glucose infused to maintain constant plasma glucose levels (hourly mean values)
Biotransformation
After subcutaneous injection in diabetic patients, insulin glargine is rapidly metabolised at the carboxyl terminus of the Beta chain with formation of two active metabolites M1 (21A-Gly-insulin) and M2 (21A-Gly-des-30B-Thr-insulin). In plasma, the principal circulating compound is the metabolite M1. The exposure to M1 increases with the administered dose of insulin glargine.
The pharmacokinetic and pharmacodynamic findings indicate that the effect of the subcutaneous injection with insulin glargine is principally based on exposure to M1. Insulin glargine and the metabolite M2 were not detectable in the vast majority of subjects and, when they were detectable their concentration was independent of the administered dose of insulin glargine.
Elimination
When given intravenously the elimination half-life of insulin glargine and human insulin were comparable.
Special populations
In clinical studies, subgroup analyses based on age and gender did not indicate any difference in safety and efficacy in insulin glargine-treated patients compared to the entire study population.
Paediatric population
Pharmacokinetics in children aged 2 to less than 6 years with type 1 diabetes mellitus was assessed in one clinical study. Plasma trough levels of insulin glargine and its main M1 and M2 metabolites were measured in children treated with insulin glargine, revealing plasma concentration patterns similar to adults, and providing no evidence for accumulation of insulin glargine or its metabolites with chronic dosing.
Preclinical safety data
Non-clinical data reveal no special hazard for humans based on conventional studies of safety pharmacology, repeated dose toxicity, genotoxicity, carcinogenic potential, toxicity to reproduction.
Related medicines
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.